- Davisson and Germer tested de Broglie’s idea by firing electrons at a nickel crystal.
- Electrons were accelerated through a known voltage and allowed to scatter from the crystal.
- The scattered electrons formed a diffraction pattern, showing wave behaviour.
- The measured wavelength matched the value calculated from de Broglie’s formula.
- The experiment demonstrated that electrons exhibit wave-like behaviour and confirmed wave–particle duality.
Definitions [31]
Definition: Electron Emission
The phenomenon of emission of electrons from the metal surface is called "Electron Emission".
Define the work function of a metal. Give its unit.
The minimum energy needed for an electron to escape from the metal surface is called the work function of that metal. Its unit is electron volt (eV).
Definition: Photoelectrons
It is a phenomenon where light falling on a material (usually a metal) causes it to emit electrons, generally called photoelectrons.
Definition: Work Function
The minimum value of energy required for the emission of photoelectrons from a metal surface is called the Work Function.
Definition: Stopping Potential
The retarding potential (–V₀) for which the photocurrent becomes zero is called the Stopping Potential.
Definition: Saturation Current
The limit of photocurrent at which the increase in photocurrent stops even if the collector plate potential (V) is increased is called the Saturation Current.
Define the term: stopping potential in the photoelectric effect.
The stopping potential is defined as the potential necessary to stop any electron from reaching the other side.
Define the term: threshold frequency
Threshold frequency is the lowest frequency of electromagnetic radiation that will result in the emission of electrons from a specified metal surface.
Definition: Threshold Frequency
The minimum value of frequency of incident radiation required for the emission of photoelectrons from a metal surface is called the Threshold Frequency.
Definition: Photons
The photoelectric effect demonstrates that light behaves as if it consists of energy packets called quanta or photons.
Definition: de Broglie Wavelength
The wavelength associated with a moving material particle is called the de Broglie wavelength.
Definition: Thermionic Emission
The emission of electrons from a metal surface by heating it to high temperature is called thermionic emission.
Definition: Field Emission
The emission of electrons from a metal surface by applying a strong electric field is called field emission.
Definition: Electron Microscope
A microscope that uses accelerated electron beams instead of visible light to obtain high-resolution images is called an electron microscope.
Definition: Photocell
A device that uses the photoelectric effect to convert light energy into electrical energy is called a photocell.
Definition: Photon
The quantum (bundle) of electromagnetic radiation having energy E = hν is called a photon.
Definition: Wave–particle Duality of Electromagnetic Radiation
The property of electromagnetic radiation to exhibit both wave nature and particle nature is called wave–particle duality of electromagnetic radiation.
Definition: Compton Shift
The change in wavelength of X-rays after scattering from electrons is called the Compton shift.
Definition: de Broglie Hypothesis
The hypothesis that matter, like radiation, exhibits both wave and particle nature is called the de Broglie hypothesis.
Definition: Matter Waves
The waves associated with moving material particles are called matter waves.
Definition: Threshold Frequency
The minimum frequency of incident radiation required to eject electrons from a metal surface (ν0 = ϕ0/h) is called the threshold frequency.
Definition: Einstein’s Photoelectric Equation
The maximum kinetic energy of emitted photoelectrons given by KEmax = hν − ϕ0 is called Einstein’s photoelectric equation.
Definition: Work Function
The minimum energy required to remove an electron from the surface of a metal is called the work function of the metal.
It is denoted by ϕ0.
Definition: Electromagnetic Radiation
Electromagnetic radiation consists of mutually perpendicular oscillating electric and magnetic fields, both perpendicular to the direction of propagation of the wave.
Definition: Photoelectric Effect
The phenomenon of emission of electrons from a metal surface when radiation of appropriate frequency is incident on it is called the photoelectric effect.
Definition: Photosensitive Surface
A surface that emits electrons when illuminated with suitable radiation is called a photosensitive surface.
Definition: Photoelectrons
Electrons emitted from a metal surface due to incident light are called photoelectrons.
Definition: Accelerating Potential
When the anode is at positive potential with respect to the cathode, it accelerates the emitted electrons. This potential is called accelerating potential.
Definition: Retarding Potential
When the anode is at negative potential with respect to the cathode, it opposes the motion of electrons. This is called retarding potential.
Definition: Stopping Potential
The minimum negative potential applied to the collector to reduce the photocurrent to zero is called the stopping potential (cut-off potential).
Definition: Einstein’s Relation
The relation E = hν, which connects the energy of a photon with its frequency, is called Einstein’s relation.
Formulae [10]
Formula: Stopping Potential
\[V_0=\frac{hv}{e}-\frac{\phi_0}{e}=\frac{K.E_{max}}{e}\]
Formula: Work Function
\[\phi_0=hv_0=h\frac{c}{\lambda_0}\]
Formula: de Broglie Relation
λ = \[\frac {h}{p}\]
\[
\lambda = \frac{h}{mv}
\]
Formula: Value of Charge of Electron
\[
e = 1.602 \times 10^{-19}\,\text{C}
\]
Formula: Electron Volt (eV)
\[
1\,\text{eV} = 1.602 \times 10^{-19}\,\text{J}
\]
Formula: Specific Charge of Electron
\[
\frac{e}{m} = 1.76 \times 10^{11}\,\text{C/kg}
\]
Formula: Einstein’s Photoelectric Equation
\[
K_{\text{max}} = eV_0
\]
or
\[
K_{\text{max}} = h\nu - \phi_0
\]
Linear Form:
\[
V_0 = \frac{h}{e}\nu - \frac{\phi_0}{e}
\]
Formula: Photon Momentum
\[
p = \frac{h\nu}{c} = \frac{h}{\lambda}
\]
Formula: Threshold Frequency
\[
\nu_0 = \frac{\phi_0}{h}
\]
Formula: Compton Shift
\[\Delta\lambda=\lambda^{\prime}-\lambda=\frac{h}{m_ec}(1-\cos\theta)\]
Key Points
Key Points: Dual Nature of Radiation
- Light (and all radiation) behaves both as a wave and as a particle, depending on circumstances.
- Wave properties: Interference, diffraction, polarisation — explained by wave theory.
- Particle properties: Photoelectric effect, Compton effect — explained by quantum/photon theory.
- Key idea: No single model (wave or particle) explains all phenomena; both are needed.
- Radiation properties of light: spreads as waves, cannot be touched, but can be experienced.
- Material properties of light: moves in straight rays, has momentum, carries definite energy.
Key Points: Electron Emission
Three Types of Electron Emission:
| Type | Method | How |
|---|---|---|
| Thermionic Emission | Heat | Thermal energy is imparted to free electrons by heating. |
| Field Emission | Strong Electric Field (~10⁸ V/m) | Free electrons experience an electric force and are pulled out. |
| Photoelectric Emission | Electromagnetic Radiation | Light of suitable frequency falls on metal, electrons are emitted. |
| Secondary Emission | High-speed electrons/particles | Metal bombarded by fast particles; electrons are emitted. |
Key Points: Photoelectric Effect — Hertz's Observations
- Hertz (1887) observed that UV light falling on a metal cathode caused sparks to jump more easily across the gap of his oscillator.
- He noticed high voltage sparks were enhanced when the emitter plate was illuminated with UV light from an arc lamp.
- The phenomenon was later identified as the Photoelectric Effect — emission of electrons when light strikes a metal.
- Hertz also found that maximum spark length was produced when the apparatus was kept in a dark box, confirming light-induced emission.
- Hertz Experiment Setup: Oscillator with brass knobs joined by an induction coil; spark balls separated by a micrometre air gap and a ring receiver.
Key Points: Photoelectric Effect — Hallwachs' and Lenard's Observations
Hallwachs' Observation:
Hallwachs confirmed that UV light incident on a negatively charged zinc plate caused it to lose charge (emit electrons).
Lenard's Observations:
- Lenard measured electron kinetic energy vs light frequency.
- Found: Maximum KE of emitted electrons is directly proportional to the frequency of incident light.
- Changing the intensity of light had no effect on kinetic energy — only on the number of electrons emitted.
- Below a certain threshold frequency (ν₀), no electrons are emitted regardless of intensity.
- The photocurrent was directly proportional to the intensity of the incident light.
- Setup: cathode illuminated with light → electrons travel through vacuum → reach anode → current measured via ammeter.
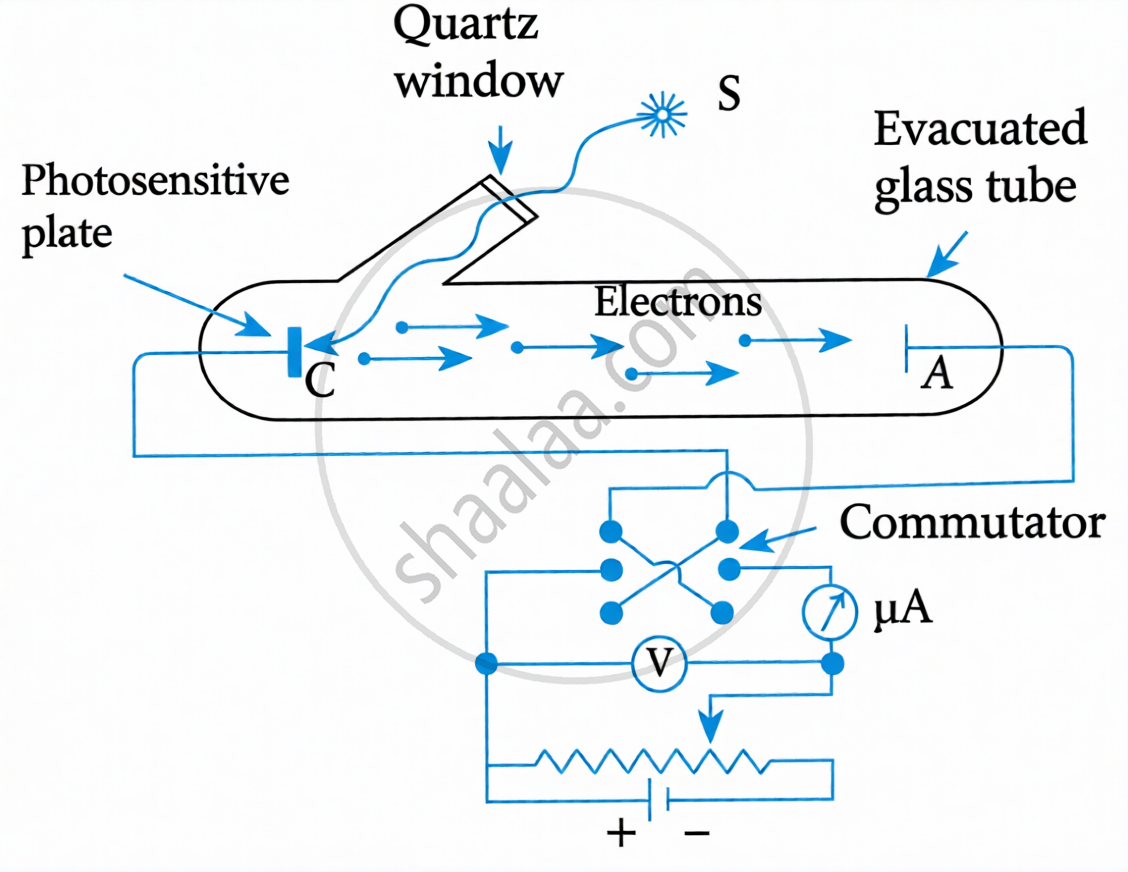
Key Points: Experimental Study of Photoelectric Effect
Setup:
- Monochromatic radiation of suitable frequency from source S falls on photosensitive plate C (cathode).
- Electrons emitted from C are collected at plate A (anode/collector), kept at positive potential.
- Photoelectrons flow in the outer circuit → microammeter shows deflection (measures photoelectric current).
- Quartz window allows UV light to enter the evacuated glass tube.
Effect of Intensity:
- Photoelectric current varies directly with intensity of incident light (more photons → more electrons).
- Graph: Linear relationship between photoelectric current and light intensity.
Effect of Potential:
- On increasing positive collector potential → photocurrent increases and reaches saturation current.
- Higher intensity → higher saturation current (more electrons).
- Even at zero potential, some current flows (electrons have kinetic energy).
- Applying negative potential reduces the current.
Stopping Potential (V₀)
- The minimum negative potential given to the collector to stop all photoelectrons is Stopping Potential.
- At stopping potential: \[K_{\max}=eV_{0}=\frac{1}{2}mv^{2}\]
- Stopping potential is independent of intensity (but depends on frequency).
- Higher frequency → larger stopping potential.
Effect of Frequency on Stopping Potential:
- Graph shows: V₀ varies linearly with frequency ν (for a given photosensitive material).
- There exists a threshold frequency ν₀ below which V₀ = 0 (no emission).
- Threshold frequency is a characteristic of the metal.
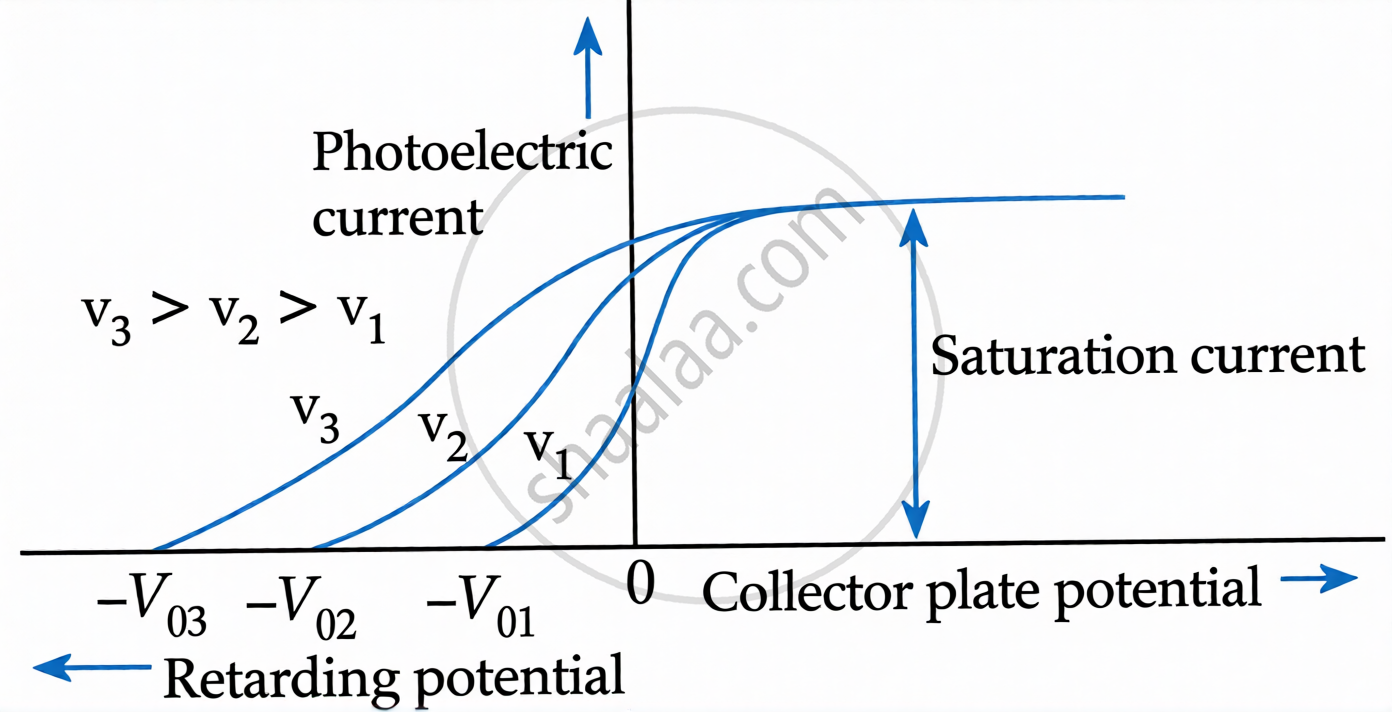
Laws of Photoelectric Emission:
- If frequency of radiation < threshold frequency → no emission (regardless of intensity).
- Maximum KE of photoelectrons depends on frequency of radiation, not intensity.
- Saturation photocurrent increases with intensity, but is independent of frequency.
- There is no time lag between incidence of radiation and emission of electrons.
Key Points: Einstein's Photoelectric Equation: Energy Quantum of Radiation
- Einstein proposed that light consists of quanta (photons), each with energy E = hν.
- When a photon of energy hν falls on a metal, it is completely absorbed by one electron.
- The electron uses energy Φ (work function) to escape, and the rest appears as kinetic energy.
Einstein's Photoelectric Equation: Kmax = hν − ϕ0
or equivalently: \[\frac{1}{2}mv_{max}^2=h\nu-h\nu_0=h(\nu-\nu_0)\]
- If ν < ν0: v2 is negative → imaginary velocity → no photoelectric emission possible.
- If ν > ν0: v2 is positive → emission occurs.
- Increasing intensity → more photons → more electrons, but same KE per electron.
- Increasing frequency → more energy per photon → higher KE of emitted electrons.
- Photoelectric emission is a 'knock-out' process: one photon knocks out one electron with KE = ½mv².
Important Note on Photocurrent vs Frequency:
- Increasing frequency does NOT increase the number of photoelectrons → photocurrent does not increase with frequency.
- Photocurrent depends only on the number of photons (i.e., intensity).
Key Points: Particle Nature of Light: The Photon
- Interaction with Matter: Radiation behaves as particles when interacting with matter.
- Energy and Momentum: Each photon has energy \[E={\frac{hc}{\lambda}}=h\nu\] and momentum \[p=\frac{hv}{c}=\frac{h}{\lambda}\].
- Intensity and Energy: All photons of a given frequency/wavelength carry the same energy, regardless of radiation intensity.
- Effect of Intensity: More intensity → more photons, but each photon has the same energy.
- Speed of Photons: All photons travel at the speed of light c.
- Frequency–Energy Relationship: A photon's frequency determines its energy; its frequency remains constant across different media.
- Photon Velocity in Different Media: Velocity varies with wavelength change in different media.
- Rest Mass of Photon: m0 = 0 (zero rest mass), because it travels at the speed of light.
- Electric and Magnetic Field Interaction: Photons are not deflected by electric/magnetic fields (they are electrically neutral).
Photon-Particle Collision:
- During collisions (like the photoelectric effect), energy and momentum are conserved.
- No new photons are created or destroyed — they are either absorbed or new photons are emitted.
Key Points: Wave Nature of Matter
- de Broglie (1924) Hypothesis: If radiation (waves) shows particle behaviour, then particles of matter should also show wave behaviour. This concept is called Matter Waves or de Broglie Waves.
- Nature's symmetry: electrons, protons, and neutrons can behave as waves under suitable conditions.
de Broglie Wave Equation:
For a particle of mass m moving with velocity v: \[\lambda=\frac{h}{p}=\frac{h}{mv}\]
Also written as: \[\lambda=\frac{h}{\sqrt{2mK.E}}\]
- Larger mass or velocity → smaller wavelength → harder to detect wave nature.
- For large (macroscopic) bodies, wavelengths are so tiny they cannot be measured — hence, no observable wave nature.
Experimental Proof of Matter Waves:
- Davisson and Germer experiment: Electrons showed diffraction patterns — direct proof of wave nature.
- G.P. Thomson's experiment also confirmed electron diffraction.
- Electrons have mass and move with definite velocity → can display wave-like behaviour.
Acceptance of Duality:
- Bohr's Law of Complementarity: Matter can be observed as either a particle or a wave, but not both simultaneously.
- Particle and wave aspects are complementary.
Key Points: Failure of Wove Theory
- Wave theory could not explain the instant emission of electrons; it predicted a time delay.
- It said higher intensity should give higher kinetic energy, but actually, kinetic energy depends on frequency, not intensity.
- Wave theory predicts emission at any frequency when intensity is high, but emission occurs only when the frequency is above the threshold frequency (ν₀).
- Even very low intensity light causes immediate emission, which contradicts wave theory.
- Hence, the photoelectric effect supported the particle (quantum) nature of light rather than the wave theory.
Key Points: Photoelectric Effect – Key Observations
- Electrons are emitted only if the light frequency is greater than a minimum value, the threshold frequency (ν₀), which differs for different metals.
- Emission of electrons is instantaneous; there is no time delay between light falling and electrons coming out.
- At a fixed frequency, photocurrent increases with increasing light intensity.
- Photocurrent increases with accelerating potential and then becomes constant; this maximum value is called the saturation current.
- Saturation current depends on light intensity, not on its frequency (if ν > ν₀).
- The maximum kinetic energy of emitted electrons depends only on the frequency of light, not on its intensity.
- Stopping potential is the minimum negative potential needed to stop the photocurrent; it depends on frequency, not on intensity.
Key Points: Einstein's Postulates
- Einstein extended Planck’s idea and proposed that light behaves as particles called photons, each carrying energy hνh\nuhν.
- A photon gives all its energy to a single electron; emission occurs only if this energy is equal to or greater than the work function of the metal.
- Photoelectric emission is instantaneous because energy transfer from photon to electron occurs in a single interaction.
- The intensity of light controls the number of emitted electrons (photocurrent), while the frequency controls the maximum kinetic energy of the electrons.
- Einstein’s photon theory successfully explained threshold frequency, stopping potential, saturation current, and all experimental observations of the photoelectric effect.
Key Points: Davisson and Germer Experiment
Key Points: Wave-Particle Duality of Matter
Important Questions [83]
- Calculate the Wavelength of Radiation Emitted When Electron in a Hydrogen Atom Jumps from N=Infinity to N=1
- Define the Term ‘Stopping Potential’ in Relation to Photo-electric Effect.
- State Clearly How Photoelectric Equation is Obtained Using the Photon Pictu.Re of Electromagnetic Radiation.
- Write the Three Salient Features Observed in Photoelectric Effect Which Can Be Explained Using this Equation.
- Answer the following question. Define the term "Threshold frequency", in the context of photoelectric emission.
- Give an example each of a metal from which photoelectric emission takes place when irradiated by UV light visible light.
- The work function of a metal is 2.31 eV. Photoelectric emission occurs when the light of frequency 6.4 × 1014 Hz is incident on the metal surface. Calculate the energy of the incident radiation
- Define the Term "Threshold Frequency", in the Context of Photoelectric Emission.
- The work function of aluminum is 4⋅2 eV. If two photons each of energy 2⋅5 eV are incident on its surface, will the emission of electrons take place? Justify your answer.
- How does one explain the emission of electrons from a photosensitive surface with the help of Einstein's photoelectric equation?
- The work function of the following metals is given : Na 2.75 ev, K = 2.3 eV, Mo = 4.17 eV and Ni = 5.15 eV. Which of these metals will not cause photoelectric emission for radiation of wavelength 3300 Å from a laser source placed 1 m away from these metals? What happens if the laser source is brought nearer and placed 50 cm away?
- Name the factors on which photoelectric emission from a surface depends.
- Light of Intensity ‘I’ and Frequency ‘V’ is Incident on a Photosensitive Surface and Causes Photoelectric Emission. What Will Be the Effect on Anode Current When the Intensity of Light is Gradually Increased
- Light of Intensity ‘I’ and Frequency ‘V’ is Incident on a Photosensitive Surface and Causes Photoelectric Emission. What Will Be the Effect on Anode Current
- Light of Intensity ‘I’ and Frequency ‘V’ is Incident on a Photosensitive Surface and Causes Photoelectric Emission. What Will Be the Effect on Anode Current When the Anode Potential is Increased? in Each Case, All Other Factors Remain the Same. Explain, Giving Justification in Each Case.
- Sketch the Graphs Showing Variation of Stopping Potential with Frequency of Incident Radiations for Two Photosensitive Materials
- Wo Monochromatic Beams, One Red and the Other Blue, Have the Same Intensity
- Draw a Plot Showing the Variation of Photoelectric Current Versus the Intensity of Incident Radiation on a Given Photosensitive Surface.
- Which One of the The Variation of Stopping Potential with Frequency of Incident Radiation for Two Photosensitive Metals a and B Has Higher Value of Work-function?
- A Photosensitive Surface Emits Photoelectrons When Red Light Falls on It. Will the Surface Emit Photoelectrons When Blue Light is Incident on It? Give Reason.
- Draw a Plot Showing the Variation of Photoelectric Current with Collector Potential for Different Frequencies but Same Intensity of Incident Radiation ?
- Use Einstein'S Photoelectric Equation to Explain the Observations from this Graph ?
- What Change Will You Observe If Intensity of Incident Radiation is Changed but the Frequency Remains the Same?
- A Beam of Monochromatic Radiation is Incident on a Photosensitive Surface. Answer the Following Question Giving Reason : Does the Kinetic Energy of the Emitted Electrons Depend on the Intensity
- A Beam of Monochromatic Radiation is Incident on a Photosensitive Surface. Answer the Following Question Giving Reason : on What Factors Does the Number of Emitted Photoelectrons Depend?
- In Photoelectric Effect, Why Should the Photoelectric Current Increase as the Intensity of Monochromatic Radiation Incident on a Photosensitive Surface is Increased? Explain.
- Draw a Plot Showing the Variation of Photoelectric Current with Collector Plate Potential for Two Different Frequencies, V1 > V2, of Incident Radiation Having the Same Intensity. in Which Case Will
- Write Three Characteristic Features in Photoelectric Effect That Cannot Be Explained on the Basis of Wave Theory of Light,
- A Beam of Monochromatic Radiation is Incident on a Photosensitive Surface. Answer the Following Question Giving Reason : Do the Emitted Photoelectrons Have the Same Kinetic Energy?
- Plot a Graph Showing the Variation of Photoelectric Current with Intensity of Light. the Work Function for the Following Metals is Given:
- Write Two Characteristic Features Observed is Photoelectric Effect Which Supports the Photon Pictures of Electromagnetic Radiation ?
- Draw a Graph Between the Frequency of Incident Radiation (υ) and the Maximum Kinetic Energy of the Electrons Emitted from the Surface of a Photosensitive Material State Clearly How this Graph Can Be
- Which one of the following metals does not exhibit emission of electrons from its surface when irradiated by visible light?
- Define the Term: Threshold Frequency and
- Define the Term 'Intensity of Radiation' in Terms of Photon Picture of Light.
- The Following Graph Shows the Variation of Photocurrent for a Photosensitive Metal
- Draw Graphs Showing Variation of Photoelectric Current with Applied Voltage for Two Incident Radiations of Equal Frequency and Different Intensities. Mark the Graph for the Radiation of Higher Intensity.
- Answer the Following Question. Plot a Graph of Photocurrent Versus Anode Potential for Radiation of Frequency ν and Intensities I1 And I2 (I1 < I2).
- On the basis of the graphs shown in the figure, answer the following questions : (a) Which physical parameter is kept constant for the three curves? (b) Which is the highest frequency
- Define the Terms "Stopping Potential' and 'Threshold Frequency' in Relation to Photoelectric Effect. How Does One Determine These Physical Quantities Using Einstein'S Equation?
- In Case of Photo Electric Effect Experiment, Explain the Following Facts, Giving Reasons. the Photo Electric Current Increases with Increase of Intensity of Incident Light.
- Define the Term: Stopping Potential in the Photoelectric Effect.
- Why it is the frequency and not the intensity of the light source that determines whether the emission of photoelectrons will occur or not? Explain.
- Read the following paragraph The figure shows the variation of photoelectric current measured Which light beam has the highest frequency and why?
- How would the stopping potential for a given photosensitive surface change if the intensity of incident radiation was decreased? Justify your answer.
- How would the stopping potential for a given photosensitive surface change if the frequency of the incident radiation were increased? Justify your answer.
- The figure shows a plot of stopping potential (V0) versus 1λ, where λ is the wavelength of the radiation causing photoelectric emission from a surface. The slope of the line is equal to ______.
- Plot a graph showing the variation of photoelectric current, as a function of anode potential for two light beams having the same frequency but different intensities I1 and I2 (I1 > I2).
- What is the effect of threshold frequency and stopping potential on increasing the frequency of the incident beam of light? Justify your answer.
- Plot a Graph to Show the Variation of Stopping Potential with Frequency of Incident Radiation in Relation to Photoelectric Effect.
- Answer the Following Question. Why is the Wave Theory of Electromagnetic Radiation Not Able to Explain the Photoelectric Effect? How Does a Photon Picture Resolve this Problem?
- Work Function of Aluminium is 4.2 Ev. If Two Photons Each of Energy 2.5 Ev Are Incident on Its Surface, Will the Emission of Electrons Take Place? Justify Your Answer.
- If Light of Wavelength 412.5 Nm is Incident on Each of the Metals Given Below, Which Ones Will Show Photoelectric Emission and Why?
- Define the Term "Cut off Frequency" in Photoelectric Emission. the Threshod Frequency of a Metal is F. When the Light of Frequency 2f is Incident on the Metal Plate, the Maximum Velocity of
- An increase in the intensity of the radiation causing photo-electric emission from a surface does not affect the maximum K.E. of the photoelectrons. Explain.
- The Stopping Potential in an Experiment on Photoelectric Effect is 1.5v. What is the Maximum Kinetic Energy of the Photoelectrons Emitted? Calculate in Joules.
- In Case of Photo Electric Effect Experiment, Explain the Following Facts, Giving Reasons. the Wave Theory of Light Could Not Explain the Existence of the Threshold Frequency.
- Plot a Graph Showing the Variation of Photoelectric Current with Collector Plate Potential at a Given Frequency but for Two Different Intensities I1 And I2, Where I2 > I1.
- Use Einstein's photoelectric equation to show how from this graph, (i) Threshold frequency, and (ii) Planck's constant can be determined.
- Choose the correct answer from given options Photons of frequency v are incident on the surface of two metals A and B of threshold frequency 3/4 v and 2/3 v, respectively.
- A photon of wavelength 663 nm is incident on a metal surface. The work function of the metal is 1.50 eV. The maximum kinetic energy of the emitted photoelectrons is ______.
- Define the Terms (I) ‘Cut-off Voltage’ and (Ii) ‘Threshold Frequency’ in Relation to the Phenomenon of Photoelectric Effect.
- Briefly Explain the Three Observed Features Which Can Be Explained by Einstein’S Photoelectric Equation.
- Point Out Any Two Characteristic Properties of Photons on Which Einstein’S Photoelectric Equation is Based ?
- Write Einstein’S Photoelectric Equation?
- The photon emitted during the de-excitation from the first excited level to the ground state of a hydrogen atom is used to irradiate a photocathode in which the stopping potential is 5 V.
- Use Einstein’S Photoelectric Equation to Show How from this Graph, (I) Threshold Frequency, and (Ii) Planck’S Constant Can Be Determined.
- How Does One Explain the Emission of Electrons from a Photosensitive Surface with the Help of Einstein’S Photoelectric Equation?
- The energy of a photon of wavelength λ is ______.
- The energy of a photon of wavelength 663 nm is ______.
- Describe Briefly How the Davisson-germer Experiment Demonstrated the Wave Nature of Electrons.
- Which of the following graphs correctly represents the variation of a particle momentum with its associated de-Broglie wavelength?
- The Wavelength λ of a Photon and the De-broglie Wavelength of an Electron Have the Same Value. Show that Energy of a Photon in (2λMc/H) Times the Kinetic Energy of Electron; Where
- How will the de-Broglie wavelength associated with an electron be affected when the velocity of the electron decreases? Justify your answer.
- How will the de-Broglie wavelength associated with an electron be affected when the accelerating potential is increased? Justify your answer.
- An alpha particle is accelerated through a potential difference of 100 V. Calculate: (i) The speed acquired by the alpha particle, and (ii) The de-Broglie wavelength is associated with it.
- A electron of mass me revolves around a nucleus of charge +Ze. Show that it behaves like a tiny magnetic dipole. Hence prove that the magnetic moment associated wit it is expressed as
- An electron is accelerated from rest through a potential difference of 100 V. Find: the wavelength associated with the momentum and the velocity required by the electron.
- Show that the wavelength of electromagnetic radiation is equal to the de Broglie wavelength of its quantum (photon).
- A Proton and an α-particle Have the Same De-broglie Wavelength Determine the Ratio of Their Speeds.
- E, c and v represent the energy, velocity and frequency of a photon. Which of the following represents its wavelength?
- Why Photoelectric Effect Cannot Be Explained on the Basis of Wave Nature of Light? Give Reasons.
- What are matter waves?
Concepts [11]
- Dual Nature of Radiation
- Electron Emission
- Photoelectric Effect - Hertz’s Observations
- Photoelectric Effect - Hallwachs’ and Lenard’s Observations
- Experimental Study of Photoelectric Effect
- Effects of Intensity and Frequency on Photocurrent
- Photoelectric Effect and Wave Theory of Light
- Einstein’s Photoelectric Equation: Energy Quantum of Radiation
- Particle Nature of Light: The Photon
- Wave Nature of Matter
- Overview: Dual Nature of Radiation and Matter
