Advertisements
Advertisements
Question
A electron of mass me revolves around a nucleus of charge +Ze. Show that it behaves like a tiny magnetic dipole. Hence prove that the magnetic moment associated wit it is expressed as `vecμ =−e/(2 m_e)vecL `, where `vec L` is the orbital angular momentum of the electron. Give the significance of negative sign.
Advertisements
Solution 1
Electrons revolve around the nucleus. A revolving electron is like a loop of current. which has a definite dipole moment.When electron revolves in anticlockwise direction, the equivalent current is clockwise. Therefore, upper face of the electron loop acts as south pole and lower face acts as north pole. Hence, an atom behaves as a magnetic dipole.
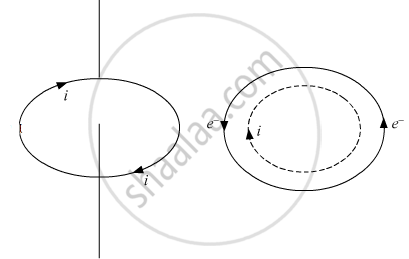
If e is the charge on an electron revolving in an orbit of radius r with a uniform angular velocity ω, then equivalent current `i="charge"/"time"=e/T`
T=the period of revolution of electron
`i=e/((2π)/ω)=(ωe)/(2π)`
`A=πr^2`
magnetic moment of the atom is given by
`μ=iA=(ωe)/(2π)πr^2`
`μ=1/2eω^2`
According to Bohr's theory
`mvr=(nh)/(2π)` where n=1,2,3.... denotes the number of the orbit.
`v=rω`
`m(rω)r=(nh)/(2π)`
`ωr^2=(nh)/(2πm_e)`
`vecμ =1/2 e nh/(2πme)`
`vecmu=n(eh)/(4πm_e) (vecL=(nh)/(2π))`
`μ⃗ =−e/(2m_e)vecL`
The negative sign has been included for the reason that electron has negative charge.
Solution 2
Electrons revolve around the nucleus. A revolving electron is like a loop of current. which has a definite dipole moment.When electron revolves in anticlockwise direction, the equivalent current is clockwise. Therefore, upper face of the electron loop acts as south pole and lower face acts as north pole. Hence, an atom behaves as a magnetic dipole.

If e is the charge on an electron revolving in an orbit of radius r with a uniform angular velocity ω, then equivalent current `i="charge"/"time"=e/T`
T=the period of revolution of electron
`i=e/((2π)/ω)=(ωe)/(2π)`
`A=πr^2`
magnetic moment of the atom is given by
`μ=iA=(ωe)/(2π)πr^2`
`μ=1/2eω^2`
According to Bohr's theory
`mvr=(nh)/(2π)` where n=1,2,3.... denotes the number of the orbit.
`v=rω`
`m(rω)r=(nh)/(2π)`
`ωr^2=(nh)/(2πm_e)`
`vecμ =1/2 e nh/(2πme)`
`vecmu=n(eh)/(4πm_e) (vecL=(nh)/(2π))`
`μ⃗ =−e/(2m_e)vecL`
The negative sign has been included for the reason that electron has negative charge.
APPEARS IN
RELATED QUESTIONS
Calculate the de Broglie wavelength of the electrons accelerated through a potential difference of 56 V.
Find the de Broglie wavelength of a neutron, in thermal equilibrium with matter, having an average kinetic energy of `(3/2)` kT at 300 K.
Sodium and copper have work function 2.3 eV and 4.5 eV respectively. Then, the ratio of the wavelengths is nearest to ______.
An electron is moving with an initial velocity `v = v_0hati` and is in a magnetic field `B = B_0hatj`. Then it’s de Broglie wavelength ______.
The de Broglie wavelength of a photon is twice the de Broglie wavelength of an electron. The speed of the electron is `v_e = c/100`. Then ______.
- `E_e/E_p = 10^-4`
- `E_e/E_p = 10^-2`
- `p_e/(m_ec) = 10^-2`
- `p_e/(m_ec) = 10^-4`
Assuming an electron is confined to a 1 nm wide region, find the uncertainty in momentum using Heisenberg Uncertainty principle (∆x∆p ≃ h). You can assume the uncertainty in position ∆x as 1 nm. Assuming p ≃ ∆p, find the energy of the electron in electron volts.
Given below are two statements:
Statement - I: Two photons having equal linear momenta have equal wavelengths.
Statement - II: If the wavelength of photon is decreased, then the momentum and energy of a photon will also decrease.
In the light of the above statements, choose the correct answer from the options given below.
The De-Broglie wavelength of electron in the third Bohr orbit of hydrogen is ______ × 10-11 m (given radius of first Bohr orbit is 5.3 × 10-11 m):
An electron of mass me, and a proton of mass mp = 1836 me are moving with the same speed. The ratio of the de Broglie wavelength `lambda_"electron"/lambda_"proton"` will be:
In a Frank-Hertz experiment, an electron of energy 5.6 eV passes through mercury vapour and emerges with an energy 0.7 eV. The minimum wavelength of photons emitted by mercury atoms is close to ______.
