Advertisements
Advertisements
प्रश्न
A electron of mass me revolves around a nucleus of charge +Ze. Show that it behaves like a tiny magnetic dipole. Hence prove that the magnetic moment associated wit it is expressed as `vecμ =−e/(2 m_e)vecL `, where `vec L` is the orbital angular momentum of the electron. Give the significance of negative sign.
Advertisements
उत्तर १
Electrons revolve around the nucleus. A revolving electron is like a loop of current. which has a definite dipole moment.When electron revolves in anticlockwise direction, the equivalent current is clockwise. Therefore, upper face of the electron loop acts as south pole and lower face acts as north pole. Hence, an atom behaves as a magnetic dipole.
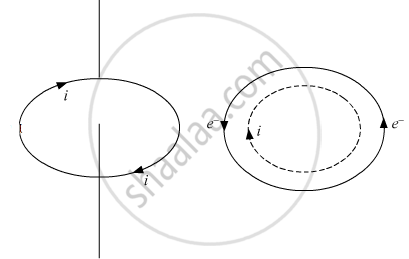
If e is the charge on an electron revolving in an orbit of radius r with a uniform angular velocity ω, then equivalent current `i="charge"/"time"=e/T`
T=the period of revolution of electron
`i=e/((2π)/ω)=(ωe)/(2π)`
`A=πr^2`
magnetic moment of the atom is given by
`μ=iA=(ωe)/(2π)πr^2`
`μ=1/2eω^2`
According to Bohr's theory
`mvr=(nh)/(2π)` where n=1,2,3.... denotes the number of the orbit.
`v=rω`
`m(rω)r=(nh)/(2π)`
`ωr^2=(nh)/(2πm_e)`
`vecμ =1/2 e nh/(2πme)`
`vecmu=n(eh)/(4πm_e) (vecL=(nh)/(2π))`
`μ⃗ =−e/(2m_e)vecL`
The negative sign has been included for the reason that electron has negative charge.
उत्तर २
Electrons revolve around the nucleus. A revolving electron is like a loop of current. which has a definite dipole moment.When electron revolves in anticlockwise direction, the equivalent current is clockwise. Therefore, upper face of the electron loop acts as south pole and lower face acts as north pole. Hence, an atom behaves as a magnetic dipole.

If e is the charge on an electron revolving in an orbit of radius r with a uniform angular velocity ω, then equivalent current `i="charge"/"time"=e/T`
T=the period of revolution of electron
`i=e/((2π)/ω)=(ωe)/(2π)`
`A=πr^2`
magnetic moment of the atom is given by
`μ=iA=(ωe)/(2π)πr^2`
`μ=1/2eω^2`
According to Bohr's theory
`mvr=(nh)/(2π)` where n=1,2,3.... denotes the number of the orbit.
`v=rω`
`m(rω)r=(nh)/(2π)`
`ωr^2=(nh)/(2πm_e)`
`vecμ =1/2 e nh/(2πme)`
`vecmu=n(eh)/(4πm_e) (vecL=(nh)/(2π))`
`μ⃗ =−e/(2m_e)vecL`
The negative sign has been included for the reason that electron has negative charge.
APPEARS IN
संबंधित प्रश्न
A proton and an α-particle have the same de-Broglie wavelength Determine the ratio of their speeds.
Calculate the de Broglie wavelength of the electrons accelerated through a potential difference of 56 V.
What is the de Broglie wavelength of a ball of mass 0.060 kg moving at a speed of 1.0 m/s?
An electron and a photon each have a wavelength of 1.00 nm. Find
(a) their momenta,
(b) the energy of the photon, and
(c) the kinetic energy of electron.
State any one phenomenon in which moving particles exhibit wave nature.
What are matter waves?
Show with the help of a labelled graph how their wavelength (λ) varies with their linear momentum (p).
Two particles A1 sand A2 of masses m1, m2 (m1 > m2) have the same de Broglie wavelength. Then ______.
- their momenta are the same.
- their energies are the same.
- energy of A1 is less than the energy of A2.
- energy of A1 is more than the energy of A2.
An alpha particle is accelerated through a potential difference of 100 V. Calculate:
- The speed acquired by the alpha particle, and
- The de-Broglie wavelength is associated with it.
(Take mass of alpha particle = 6.4 × 10−27 kg)
The ratio of wavelengths of proton and deuteron accelerated by potential Vp and Vd is 1 : `sqrt2`. Then, the ratio of Vp to Vd will be ______.
