Advertisements
Advertisements
Question
Define the terms "stopping potential' and 'threshold frequency' in relation to the photoelectric effect. How does one determine these physical quantities using Einstein's equation?
Advertisements
Solution
Stopping potential:
For a particular frequency of incident radiation, the minimum negative (retarding) potential V0 given to the anode plate for which the photocurrent stops or becomes zero is called the cut-off or stopping potential.
Threshold frequency:
There exists a certain minimum cut-off frequency ν0, for which the stopping potential is zero and below ν0 the electron emission is not possible.
This cut-off frequency is known as threshold frequency ν0, which is different for different metal. In the photoelectric effect, an electron absorbs a quantum of energy (hν ) of radiation. If this quantum of energy absorbed by electron exceeds the minimum energy required to come out of the metal surface by electron, the kinetic energy of the emitted electron is
`"K" = "hv" - phi` ...(1)
Where `phi` is the minimum energy for electron to come out of the metal, and is different for different electrons in the metal. The maximum kinetic energy of photoelectrons is given by
`"K""max" = "hv" - phi0` ...(2)
Where, `phi0 - ` work function or least value of φ equation (2) is known as Einstein's photoelectric equation.
Explanation of photoelectric effect with the help of Einstein's photoelectric equation
(i) According to equation (2), Kmax depends linearly on ν, and is independent of the intensity of radiation. This happens because, here, the photoelectric effect arises from the absorption of a single quantum of radiation by a single electron. The intensity of the radiation (that is proportional to the number of energy quanta per unit area per unit time) is irrelevant to this basic process.
(ii) Since Kmax must be non-negative, equation (2) implies that photoelectric emission is possible only if h ν > `phi0`.
or v > v0, where v0 = `"V"_0 = phi_0/"h"`
Thus, there exists a threshold frequency v0 `"V"_0 = phi_0/"h"` exists, below which photoelectric emission is not possible, and is independent of intensity.
(iii) As the intensity of radiation is proportional to the number of energy quanta per unit area per unit time. The greater the number of energy quanta available, the greater is the number of electrons absorbing the energy quanta, and therefore, the number of electrons coming out of the metal (for ν > ν0) is more and so is photoelectric current.
APPEARS IN
RELATED QUESTIONS
The following graph shows the variation of photocurrent for a photosensitive metal :
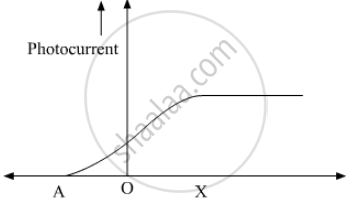
(a) Identify the variable X on the horizontal axis.
(b) What does the point A on the horizontal axis represent?
(c) Draw this graph for three different values of frequencies of incident radiation v1, v2 and v3 (v1 > v2 > v3) for same intensity.
(d) Draw this graph for three different values of intensities of incident radiation I1, I2 and I3 (I1 > I2 > I3) having same frequency.
Can we find the mass of a photon by the definition p = mv?
Let nr and nb be the number of photons emitted by a red bulb and a blue bulb, respectively, of equal power in a given time.
When stopping potential is applied in an experiment on photoelectric effect, no photoelectric is observed. This means that
A point source causes photoelectric effect from a small metal plate. Which of the following curves may represent the saturation photocurrent as a function of the distance between the source and the metal?
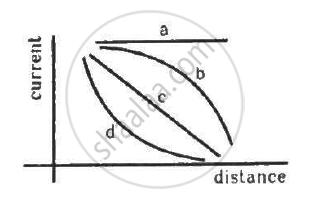
A photon of energy hv is absorbed by a free electron of a metal with work-function hv − φ.
A 100 W light bulb is placed at the centre of a spherical chamber of radius 20 cm. Assume that 60% of the energy supplied to the bulb is converted into light and that the surface of the chamber is perfectly absorbing. Find the pressure exerted by the light on the surface of the chamber.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
The electric field associated with a light wave is given by `E = E_0 sin [(1.57 xx 10^7 "m"^-1)(x - ct)]`. Find the stopping potential when this light is used in an experiment on photoelectric effect with the emitter having work function 1.9 eV.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
Answer the following question.
Plot a graph of photocurrent versus anode potential for radiation of frequency ν and intensities I1 and I2 (I1 < I2).
A metallic plate exposed to white light emits electrons. For which of the following colours of light, the stopping potential will be maximum?
