Advertisements
Advertisements
Question
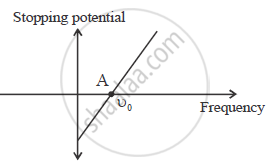
Use Einstein’s photoelectric equation to show how from this graph,
(i) Threshold frequency, and (ii) Planck’s constant can be determined.
Advertisements
Solution
eV0 =hυ - hυ0
`V_0 = h/e(υ - v_0)`
Point A on the graph shows threshold frequency.
eV0 = hv - Φ0
`V_0 = h/e υ - (phi _0)/e`
Slope of the given graphs gives `h/e`
Slope `= h/e`
h = e × slope of the graph
APPEARS IN
RELATED QUESTIONS
Briefly explain the three observed features which can be explained by Einstein’s photoelectric equation.
Define the terms (i) ‘cut-off voltage’ and (ii) ‘threshold frequency’ in relation to the phenomenon of photoelectric effect.
Using Einstein’s photoelectric equation shows how the cut-off voltage and threshold frequency for a given photosensitive material can be determined with the help of a suitable plot/graph.
Is p − E/c valid for electrons?
A monochromatic light source of intensity 5 mW emits 8 × 1015 photons per second. This light ejects photoelectrons from a metal surface. The stopping potential for this setup is 2.0 V. Calculate the work function of the metal.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
A small metal plate (work function φ) is kept at a distance d from a singly-ionised, fixed ion. A monochromatic light beam is incident on the metal plate and photoelectrons are emitted. Find the maximum wavelength of the light beam, so that some of the photoelectrons may go round the ion along a circle.
Consider the situation of the previous problem. Consider the faster electron emitted parallel to the large metal plate. Find the displacement of this electron parallel to its initial velocity before it strikes the large metal plate.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
How does one explain the emission of electrons from a photosensitive surface with the help of Einstein’s photoelectric equation?
Choose the correct answer from given options
Photons of frequency v are incident on the surface of two metals A and B of threshold frequency 3/4 v and 2/3 v, respectively. The ratio of maximum kinetic energy of electrons emitted from A to that from B is
The wavelength of a photon needed to remove a proton from a nucleus which is bound to the nucleus with 1 MeV energy is nearly ______.
There are materials which absorb photons of shorter wavelength and emit photons of longer wavelength. Can there be stable substances which absorb photons of larger wavelength and emit light of shorter wavelength.
