Advertisements
Advertisements
प्रश्न
Use Einstein’s photoelectric equation to show how from this graph,
(i) Threshold frequency, and (ii) Planck’s constant can be determined.
Advertisements
उत्तर
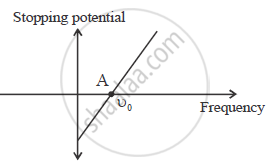
eV0 =hυ - hυ0
`V_0 = h/e(υ - v_0)`
Point A on the graph shows threshold frequency.
eV0 = hv - Φ0
`V_0 = h/e υ - (phi _0)/e`
Slope of the given graphs gives `h/e`
Slope `= h/e`
h = e × slope of the graph
APPEARS IN
संबंधित प्रश्न
In an experiment on the photoelectric effect, the slope of the cut-off voltage versus the frequency of incident light is found to be 4.12 × 10−15 Vs. Calculate the value of Planck’s constant.
The work function for a certain metal is 4.2 eV. Will this metal give photoelectric emission for incident radiation of wavelength 330 nm?
Light of wavelength 488 nm is produced by an argon laser which is used in the photoelectric effect. When light from this spectral line is incident on the emitter, the stopping (cut-off) potential of photoelectrons is 0.38 V. Find the work function of the material from which the emitter is made.
point out any two characteristic properties of photons on which Einstein’s photoelectric equation is based ?
Is p − E/c valid for electrons?
A monochromatic light source of intensity 5 mW emits 8 × 1015 photons per second. This light ejects photoelectrons from a metal surface. The stopping potential for this setup is 2.0 V. Calculate the work function of the metal.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
How does one explain the emission of electrons from a photosensitive surface with the help of Einstein’s photoelectric equation?
According to Einstein's photoelectric equation, the plot of the kinetic energy of the emitted photoelectrons from a metal versus the frequency of the incident radiation gives a straight line, whose slope ______.
Each photon has the same speed but different ______.
Radiation of frequency 1015 Hz is incident on three photosensitive surfaces A, B and C. Following observations are recorded:
Surface A: no photoemission occurs
Surface B: photoemission occurs but the photoelectrons have zero kinetic energy.
Surface C: photo emission occurs and photoelectrons have some kinetic energy.
Using Einstein’s photo-electric equation, explain the three observations.
