5. Conclusion: The experiments show that chemical changes result in the formation of new substances with distinct properties, whereas physical changes are reversible and do not alter the composition of the substance.
Topics
Gravitation
- Concept of Gravitation
- Force and Motion
- Centripetal Force
- Kepler’s Laws
- Law of Orbit or Kepler's First Law
- Law of Areas or Kepler's Second Law
- Law of Periods or Kepler's Third Law
- Newton's Universal Law of Gravitation
- Uniform Circular Motion (UCM)
- Earth’s Gravitational Force
- Earth’s Gravitational Acceleration
- Mass and Weight
- Gravitational Waves
- Free Fall
- Gravitational Potential Energy
- Escape Velocity
- Weightlessness in Space
Periodic Classification of Elements
- Classification of Elements
- Dobereiner’s Triads
- Newland's Law of Octaves
- Mendeleev’s Periodic Table
- Insights into Mendeleev’s Periodic Table
- Modern Periodic Law
- The Modern Periodic Table
- Structure of the Modern Periodic Table
- Modern Periodic Table and Electronic Configuration of Elements
- Groups and Electronic Configuration
- Periods and Electronic Configuration
- Periodic Trends in the Modern Periodic Table
- Atomic Size
- Metallic and Non-metallic Characters
- Gradation in Halogen Family
Chemical Reactions and Equations
- Chemical Reaction
- Chemical Equations
- Balancing Chemical Equation
- Types of Chemical Reactions > Combination Reaction
- Types of Chemical Reactions > Decomposition Reaction
- Types of Chemical Reactions > Single Displacement Reaction
- Types of Chemical Reactions > Double Displacement Reaction
- Endothermic and Exothermic Processes
- Rate of Chemical Reaction
- Factors Affecting the Rate of a Chemical Reaction
- Chemical Properties of Carbon Compounds > Oxidation
- Chemical Properties of Carbon Compounds > Reduction
- Corrosion of Metals
- Rancidity
Effects of Electric Current
- Electric Circuit
- Heating Effect of Electric Current
- Magnetic Effect of Electric Current
- Right-hand Thumb Rule
- Applications of Biot-Savart's Law > Magnetic Field at the Centre of a Circular Loop
- Applications of Ampere’s Circuital Law > Magnetic Field of a Long Straight Solenoid
- Force on a Current Carrying Conductor in a Magnetic Field
- Fleming’s Left Hand Rule
- Electric Motor
- Electromagnetic Induction
- Galvanometer
- Faraday's Laws of Electromagnetic Induction
- Fleming’s Right Hand Rule
- Alternating current (AC) and Direct Current (DC)
- Electric Generator
Heat
Refraction of Light
Lenses
- Concept of Lenses
- Images Formed by Convex Lenses
- Images Formed by Concave Lenses
- Sign Convention
- Lens Formula
- Magnification
- Power of a Lens
- Combination of Lenses
- Defects of Vision and Their Corrections > Myopia
- Defects of Vision and Their Corrections > Hypermetropia
- Defects of Vision and Their Corrections > Presbyopia
- Apparent Size of an Object
- Use of Concave Lenses
- Use of Convex Lenses
- Persistence of Vision
Metallurgy
- Physical Properties of Metals
- Physical Properties of Non-metal
- Chemical Properties of Metal
- Reactions of Metals
- Reactivity Series of Metals
- Chemical Properties of Non-metal
- Ionic Compounds
- Metallurgy
- Basic Principles of Metallurgy > Concentration of Ores
- Basic Principles of Metallurgy > Extraction of Metals
- Basic Principles of Metallurgy > Refining of Metals
- Corrosion of Metals
- Prevention of Corrosion
Carbon Compounds
- Bonds in Carbon Compounds
- Carbon: A Versatile Element
- Hydrocarbons
- Straight chains, Branched chains, and Rings of Carbon atoms
- Functional Groups in Carbon Compounds
- Homologous Series
- Nomenclature
- Chemical Properties of Carbon Compounds > Combustion
- Chemical Properties of Carbon Compounds > Oxidation
- Chemical Properties of Carbon Compounds > Addition Reaction
- Chemical Properties of Carbon Compounds > Substitution Reaction
- Ethanol
- Ethanoic Acid
- Macromolecules and Polymers
Space Missions
School of Elements
The Magic of Chemical Reactions
- Chemical Equations
- Types of Chemical Reactions > Combination Reaction
- Types of Chemical Reactions > Decomposition Reaction
- Types of Chemical Reactions > Single Displacement Reaction
- Types of Chemical Reactions > Double Displacement Reaction
- Chemical Properties of Carbon Compounds > Oxidation
- Types of Double Displacement: Neutralization Reaction
The Acid Base Chemistry
- Properties of Acids > Physical Properties
- The pH Scale
- Acids, Bases and Their Reactivity
- Acid or a Base in a Water Solution
- Preparation and Uses of Baking Soda
- Preparation and Uses of Bleaching Powder
- Preparation and Uses of Washing Soda
- Preparation and Uses of Plaster of Paris
- Chemicals from Common Salt - Soap as a Salt
The Electric Spark
All about Electromagnetism
- Magnetic force
- Bar Magnet and Solenoid Analogy
- Right-hand Thumb Rule
- Applications of Biot-Savart's Law > Magnetic Field at the Centre of a Circular Loop
- Applications of Ampere’s Circuital Law > Magnetic Field of a Long Straight Solenoid
- Force on a Current Carrying Conductor in a Magnetic Field
- Electric Motor
- Electromagnetic Induction
- A.C. Generator
- Simple D.C. Motor
- Household Electrical Circuits
Wonders of Light 1
Wonders of Light 2
Striving for better Environment 1
- Abatement of Pollution
- Sustainable Use of Resources
- Definition: Chemical Change
- Definition: Reactants
- Definition: Products
Maharashtra State Board: Class 10
Definition: Chemical Change
A chemical change is a process in which the composition of matter changes, resulting in the formation of one or more new substances.
Maharashtra State Board: Class 10
Definition: Reactants
The substances taking part in chemical reaction are called reactants.
Maharashtra State Board: Class 10
Definition: Products
The substances formed as a result of a chemical reaction by formation of new bonds are called products.
Maharashtra State Board: Class 8
Types of Chemical Reaction:
| Type of reaction | Definition | Equation |
| Combination | Two or more elements or compounds combine to make a more complex substance. | A + B = AB |
| Decomposition | Compounds break down into simpler substances. | AB = A + B |
| Replacement | Occurs when one element replaces another in a compound | AB + C = AC + B |
| Double replacement | Occurs when different atoms in two different compounds trade places | AB + CD = AC + BD |
Maharashtra State Board: Class 10
Experiment
1. Aim: To observe and differentiate between chemical and physical changes through various experiments.
2. Requirements
- Apparatus: thermometer, evaporating dish, tripod stand, funnel, Bunsen burner.
- Chemicals: limestone powder (CaCO₃), copper sulphate (CuSO₄), zinc dust (Zn), potassium chromate (K₂CrO₄), barium sulphate (BaSO₄), sodium carbonate (Na₂CO₃), calcium chloride (CaCl₂), and phthalic anhydride.
3. Procedure
- Heat limestone powder strongly in an evaporating dish over a high blue flame.
- Add zinc dust into the copper sulphate solution and observe any changes.
- Mix potassium chromate solution with barium sulphate solution and note the reaction.
- Add sodium carbonate solution to calcium chloride solution and observe any precipitate formation.
- Heat phthalic anhydride in an evaporating dish with an inverted funnel placed over it and observe changes.
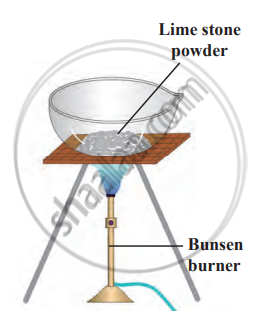
To heat limestone powder
4. Observation
| Activity | Colour Change (if present) | Gas Released (Yes/No) | Temperature Change (if present) | Nature of Change (Chemical/Physical) |
|---|---|---|---|---|
| 1 | No | Yes | Yes | Chemical |
| 2 | Yes | No | Yes | Chemical |
| 3 | Yes | No | Yes | Chemical |
| 4 | Yes | No | Yes | Chemical |
| 5 | No | No | No | Physical |
Shaalaa.com | Types of Chemical Reactions
to track your progress
