Topics
Gravitation
- Concept of Gravitation
- Force and Motion
- Centripetal Force
- Kepler’s Laws
- Law of Orbit or Kepler's First Law
- Law of Areas or Kepler's Second Law
- Law of Periods or Kepler's Third Law
- Newton's Universal Law of Gravitation
- Uniform Circular Motion (UCM)
- Earth’s Gravitational Force
- Earth’s Gravitational Acceleration
- Mass and Weight
- Gravitational Waves
- Free Fall
- Gravitational Potential Energy
- Escape Velocity
- Weightlessness in Space
Periodic Classification of Elements
- Classification of Elements
- Dobereiner’s Triads
- Newland's Law of Octaves
- Mendeleev’s Periodic Table
- Insights into Mendeleev’s Periodic Table
- Modern Periodic Law
- The Modern Periodic Table
- Structure of the Modern Periodic Table
- Modern Periodic Table and Electronic Configuration of Elements
- Groups and Electronic Configuration
- Periods and Electronic Configuration
- Periodic Trends in the Modern Periodic Table
- Atomic Size
- Metallic and Non-metallic Characters
- Gradation in Halogen Family
Chemical Reactions and Equations
- Chemical Reaction
- Chemical Equations
- Balancing Chemical Equation
- Types of Chemical Reactions > Combination Reaction
- Types of Chemical Reactions > Decomposition Reaction
- Types of Chemical Reactions > Single Displacement Reaction
- Types of Chemical Reactions > Double Displacement Reaction
- Endothermic and Exothermic Processes
- Rate of Chemical Reaction
- Factors Affecting the Rate of a Chemical Reaction
- Chemical Properties of Carbon Compounds > Oxidation
- Chemical Properties of Carbon Compounds > Reduction
- Corrosion of Metals
- Rancidity
Effects of Electric Current
- Electric Circuit
- Heating Effect of Electric Current
- Magnetic Effect of Electric Current
- Right-hand Thumb Rule
- Applications of Biot-Savart's Law > Magnetic Field at the Centre of a Circular Loop
- Applications of Ampere’s Circuital Law > Magnetic Field of a Long Straight Solenoid
- Force on a Current Carrying Conductor in a Magnetic Field
- Fleming’s Left Hand Rule
- Electric Motor
- Electromagnetic Induction
- Galvanometer
- Faraday's Laws of Electromagnetic Induction
- Fleming’s Right Hand Rule
- Alternating current (AC) and Direct Current (DC)
- Electric Generator
Heat
Refraction of Light
Lenses
- Concept of Lenses
- Images Formed by Convex Lenses
- Images Formed by Concave Lenses
- Sign Convention
- Lens Formula
- Magnification
- Power of a Lens
- Combination of Lenses
- Defects of Vision and Their Corrections > Myopia
- Defects of Vision and Their Corrections > Hypermetropia
- Defects of Vision and Their Corrections > Presbyopia
- Apparent Size of an Object
- Use of Concave Lenses
- Use of Convex Lenses
- Persistence of Vision
Metallurgy
- Physical Properties of Metals
- Physical Properties of Non-metal
- Chemical Properties of Metal
- Reactions of Metals
- Reactivity Series of Metals
- Chemical Properties of Non-metal
- Ionic Compounds
- Metallurgy
- Basic Principles of Metallurgy > Concentration of Ores
- Basic Principles of Metallurgy > Extraction of Metals
- Basic Principles of Metallurgy > Refining of Metals
- Corrosion of Metals
- Prevention of Corrosion
Carbon Compounds
- Bonds in Carbon Compounds
- Carbon: A Versatile Element
- Hydrocarbons
- Straight chains, Branched chains, and Rings of Carbon atoms
- Functional Groups in Carbon Compounds
- Homologous Series
- Nomenclature
- Chemical Properties of Carbon Compounds > Combustion
- Chemical Properties of Carbon Compounds > Oxidation
- Chemical Properties of Carbon Compounds > Addition Reaction
- Chemical Properties of Carbon Compounds > Substitution Reaction
- Ethanol
- Ethanoic Acid
- Macromolecules and Polymers
Space Missions
School of Elements
The Magic of Chemical Reactions
- Chemical Equations
- Types of Chemical Reactions > Combination Reaction
- Types of Chemical Reactions > Decomposition Reaction
- Types of Chemical Reactions > Single Displacement Reaction
- Types of Chemical Reactions > Double Displacement Reaction
- Chemical Properties of Carbon Compounds > Oxidation
- Types of Double Displacement: Neutralization Reaction
The Acid Base Chemistry
- Properties of Acids > Physical Properties
- The pH Scale
- Acids, Bases and Their Reactivity
- Acid or a Base in a Water Solution
- Preparation and Uses of Baking Soda
- Preparation and Uses of Bleaching Powder
- Preparation and Uses of Washing Soda
- Preparation and Uses of Plaster of Paris
- Chemicals from Common Salt - Soap as a Salt
The Electric Spark
All about Electromagnetism
- Magnetic force
- The Bar Magnet
- Right-hand Thumb Rule
- Applications of Biot-Savart's Law > Magnetic Field at the Centre of a Circular Loop
- Applications of Ampere’s Circuital Law > Magnetic Field of a Long Straight Solenoid
- Force on a Current Carrying Conductor in a Magnetic Field
- Electric Motor
- Electromagnetic Induction
- A.C. Generator
- Simple D.C. Motor
- Household Electrical Circuits
Wonders of Light 1
Wonders of Light 2
Striving for better Environment 1
- Abatement of Pollution
- Sustainable Use of Resources
- Definition: Catalyst
- Key Points: Factors Affecting the Rate of a Chemical Reaction
Definition: Catalyst
“The substance in whose presence the rate of a chemical reaction increases, without causing any chemical change to it, is called a catalyst.”
Key Points: Factors Affecting the Rate of a Chemical Reaction
- Nature of Reactants: More reactive substances (e.g., Al over Zn) react faster, affecting the reaction rate.
- Particle Size: Smaller particles have more surface area, leading to a faster reaction (e.g., powder reacts faster than pieces).
- Concentration: A higher concentration of reactants increases the rate of reaction (e.g., concentrated HCl reacts faster than dilute HCl).
- Temperature: Higher temperature increases particle energy, leading to a faster reaction rate.
- Catalyst: A catalyst speeds up a reaction without undergoing any chemical change itself (e.g., MnO₂ in decomposition reactions).
Nature of the Reactants
The nature of reactants plays a crucial role in determining the speed of a chemical reaction. Different substances react at different rates based on their reactivity and bonding type.
1. Reactivity of Metals
Let’s compare the reaction of two metals, aluminium (Al) and zinc (Zn), with dilute hydrochloric acid (HCl):
- Both Al and Zn react with HCl, producing hydrogen gas (H₂) and forming water-soluble salts.
- However, aluminium reacts faster than zinc because Al is more reactive than Zn.
- Since aluminium has higher reactivity, it reacts more vigorously, increasing the rate of reaction. Thus, more reactive metals react faster with acids, while less reactive metals react slower.
2. Effect of Bonding Type on Reaction Rate
The type of bonding in reactants also affects the rate of reaction:
- Ionic Compounds: React faster because they involve the direct transfer of ions.
- Covalent Compounds: React slower as their bonds need to be broken before the reaction can occur.
Since ionic bonds involve ion transfer, reactions occur quickly. On the other hand, covalent bonds require bond cleavage, which slows down the reaction. Thus, the reactivity of a substance and its bonding type significantly impact how fast a reaction takes place.
Size of the Particles of Reactants
The size of the reactant particles is another important factor that affects the rate of a chemical reaction.
- When a solid reacts with a liquid or gas, the reaction takes place at the surface of the solid.
- If the solid is broken into smaller pieces or powdered, its surface area increases.
- A larger surface area allows more particles to come in contact with the reactant, increasing the rate of reaction.
Example:
- Powdered zinc (Zn) reacts faster with hydrochloric acid (HCl) than a solid zinc strip because the powder has a greater surface area.
- Fine wood dust catches fire more easily than a solid piece of wood due to increased exposure to oxygen. Thus, smaller particle size leads to a faster reaction rate, while larger particles result in a slower reaction due to reduced surface area.
Experiment
1. Aim: To study how the size of reactant particles affects the rate of a chemical reaction.
2. Requirements: two test tubes, a balance, a measuring cylinder, pieces of Shahabad tile, powdered Shahabad tile, and dilute hydrochloric acid (HCl).
3. Procedure
- Take equal weights of Shahabad tile pieces in one test tube and powdered Shahabad tile in another.
- Add 10 ml of dilute HCl to each test tube.
- Observe the formation of effervescence due to the release of carbon dioxide (CO₂).
4. Observation
- The reaction is slower with Shahabad tile pieces.
- The reaction is faster with the powdered Shahabad tile.
5. Conclusion: Smaller reactant particles increase the surface area, leading to a faster reaction rate.
Concentration of the Reactants
The concentration of reactants plays a crucial role in determining the speed of a chemical reaction. A higher concentration leads to a faster reaction, while a lower concentration slows it down.
For example, when calcium carbonate (CaCO₃) reacts with hydrochloric acid (HCl):
- Dilute HCl reacts slowly with CaCO₃, causing it to disappear gradually and releasing carbon dioxide (CO₂) at a slower rate.
- Concentrated HCl reacts much faster, making CaCO₃ dissolve quickly and releasing CO₂ more rapidly.
This shows that the reaction rate is directly proportional to the concentration of reactants.
- According to collision theory, increasing the concentration of reactants results in more frequent molecular collisions, which speeds up the reaction.
- The law of mass action states that the rate of a chemical reaction is directly proportional to the concentration of the reactants.
- As the reaction progresses, the concentration of reactants decreases, leading to a slower reaction over time.
- A higher concentration of reactants increases the reaction rate by enhancing molecular collisions. Time also plays a significant role, as changes in reactant concentration affect how fast the reaction occurs.
Temperature of the Reaction
Temperature plays a crucial role in determining the speed of a chemical reaction. Generally, increasing the temperature increases the reaction rate, while lowering the temperature slows it down.
For example, in the decomposition of limestone (CaCO₃), lime water does not turn milky before heating because the reaction rate is zero at room temperature. However, once heated, the reaction begins, and carbon dioxide (CO₂) is released, turning the lime water milky.
- When temperature increases, the kinetic energy of reactant molecules also increases.
- This causes more frequent and energetic collisions between molecules.
- A higher proportion of molecules gain the activation energy, leading to more successful reactions.
Increasing temperature speeds up a reaction by providing molecules with more energy for effective collisions. Thus, temperature is a key factor affecting the rate of chemical reactions.
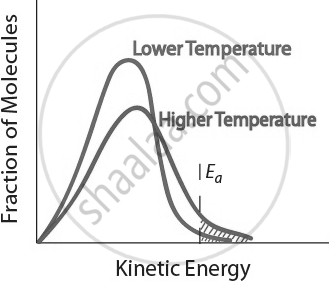
Temperature and Reaction Rate. Effect of temperature on the kinetic energy distribution of molecules in a sample
Catalyst:
A catalyst is a substance that increases the rate of a chemical reaction without undergoing any permanent change itself. It provides an alternative reaction pathway with lower activation energy, allowing reactants to convert into products more easily. Catalysts are essential in many chemical and biological reactions. They speed up reactions by lowering activation energy without being consumed in the process.
Examples,
1. Decomposition of Potassium Chlorate (KClO₃):
- When heated alone, KClO₃ decomposes slowly.
- In the presence of manganese dioxide (MnO₂), the reaction occurs rapidly, releasing oxygen (O₂).
- MnO₂ remains unchanged after the reaction.
2. Decomposition of Hydrogen Peroxide (H₂O₂):
- H₂O₂ decomposes slowly at room temperature.
- Adding MnO₂ speeds up the reaction, breaking H₂O₂ into water and oxygen faster.
Types of Catalysts:
- Positive Catalysts: Increase the reaction rate (e.g., MnO₂ in KClO₃ decomposition).
- Negative Catalysts: Slow down the reaction by increasing activation energy.
