Topics
Gravitation
- Concept of Gravitation
- Force and Motion
- Centripetal Force
- Kepler’s Laws
- Law of Orbit or Kepler's First Law
- Law of Areas or Kepler's Second Law
- Law of Periods or Kepler's Third Law
- Newton's Universal Law of Gravitation
- Uniform Circular Motion (UCM)
- Earth’s Gravitational Force
- Earth’s Gravitational Acceleration
- Mass and Weight
- Gravitational Waves
- Free Fall
- Gravitational Potential Energy
- Escape Velocity
- Weightlessness in Space
Periodic Classification of Elements
- Classification of Elements
- Dobereiner’s Triads
- Newland's Law of Octaves
- Mendeleev’s Periodic Table
- Insights into Mendeleev’s Periodic Table
- Modern Periodic Law
- The Modern Periodic Table
- Structure of the Modern Periodic Table
- Modern Periodic Table and Electronic Configuration of Elements
- Groups and Electronic Configuration
- Periods and Electronic Configuration
- Periodic Trends in the Modern Periodic Table
- Atomic Size
- Metallic and Non-metallic Characters
- Gradation in Halogen Family
Chemical Reactions and Equations
- Chemical Reaction
- Chemical Equations
- Balancing Chemical Equation
- Types of Chemical Reactions > Combination Reaction
- Types of Chemical Reactions > Decomposition Reaction
- Types of Chemical Reactions > Single Displacement Reaction
- Types of Chemical Reactions > Double Displacement Reaction
- Endothermic and Exothermic Processes
- Rate of Chemical Reaction
- Factors Affecting the Rate of a Chemical Reaction
- Chemical Properties of Carbon Compounds > Oxidation
- Chemical Properties of Carbon Compounds > Reduction
- Corrosion of Metals
- Rancidity
Effects of Electric Current
- Electric Circuit
- Heating Effect of Electric Current
- Magnetic Effect of Electric Current
- Right-hand Thumb Rule
- Applications of Biot-Savart's Law > Magnetic Field at the Centre of a Circular Loop
- Applications of Ampere’s Circuital Law > Magnetic Field of a Long Straight Solenoid
- Force on a Current Carrying Conductor in a Magnetic Field
- Fleming’s Left Hand Rule
- Electric Motor
- Electromagnetic Induction
- Galvanometer
- Faraday's Laws of Electromagnetic Induction
- Fleming’s Right Hand Rule
- Alternating current (AC) and Direct Current (DC)
- Electric Generator
Heat
Refraction of Light
Lenses
- Concept of Lenses
- Images Formed by Convex Lenses
- Images Formed by Concave Lenses
- Sign Convention
- Lens Formula
- Magnification
- Power of a Lens
- Combination of Lenses
- Defects of Vision and Their Corrections > Myopia
- Defects of Vision and Their Corrections > Hypermetropia
- Defects of Vision and Their Corrections > Presbyopia
- Apparent Size of an Object
- Use of Concave Lenses
- Use of Convex Lenses
- Persistence of Vision
Metallurgy
- Physical Properties of Metals
- Physical Properties of Non-metal
- Chemical Properties of Metal
- Reactions of Metals
- Reactivity Series of Metals
- Chemical Properties of Non-metal
- Ionic Compounds
- Metallurgy
- Basic Principles of Metallurgy > Concentration of Ores
- Basic Principles of Metallurgy > Extraction of Metals
- Basic Principles of Metallurgy > Refining of Metals
- Corrosion of Metals
- Prevention of Corrosion
Carbon Compounds
- Bonds in Carbon Compounds
- Carbon: A Versatile Element
- Hydrocarbons
- Straight chains, Branched chains, and Rings of Carbon atoms
- Functional Groups in Carbon Compounds
- Homologous Series
- Nomenclature
- Chemical Properties of Carbon Compounds > Combustion
- Chemical Properties of Carbon Compounds > Oxidation
- Chemical Properties of Carbon Compounds > Addition Reaction
- Chemical Properties of Carbon Compounds > Substitution Reaction
- Ethanol
- Ethanoic Acid
- Macromolecules and Polymers
Space Missions
School of Elements
The Magic of Chemical Reactions
- Chemical Equations
- Types of Chemical Reactions > Combination Reaction
- Types of Chemical Reactions > Decomposition Reaction
- Types of Chemical Reactions > Single Displacement Reaction
- Types of Chemical Reactions > Double Displacement Reaction
- Chemical Properties of Carbon Compounds > Oxidation
- Types of Double Displacement: Neutralization Reaction
The Acid Base Chemistry
- Properties of Acids > Physical Properties
- The pH Scale
- Acids, Bases and Their Reactivity
- Acid or a Base in a Water Solution
- Preparation and Uses of Baking Soda
- Preparation and Uses of Bleaching Powder
- Preparation and Uses of Washing Soda
- Preparation and Uses of Plaster of Paris
- Chemicals from Common Salt - Soap as a Salt
The Electric Spark
All about Electromagnetism
- Magnetic force
- Bar Magnet and Solenoid Analogy
- Right-hand Thumb Rule
- Applications of Biot-Savart's Law > Magnetic Field at the Centre of a Circular Loop
- Applications of Ampere’s Circuital Law > Magnetic Field of a Long Straight Solenoid
- Force on a Current Carrying Conductor in a Magnetic Field
- Electric Motor
- Electromagnetic Induction
- A.C. Generator
- Simple D.C. Motor
- Household Electrical Circuits
Wonders of Light 1
Wonders of Light 2
Striving for better Environment 1
- Abatement of Pollution
- Sustainable Use of Resources
- Key Points: Orbits of Artificial Satellites
Orbit and Critical Velocity of Artificial Satellites
Artificial satellites are not placed in the same type of orbit. The height, shape, and tilt of the orbit are decided based on the purpose of the satellite. These satellites are launched using rockets (launch vehicles) and positioned at a specific height (h) above the Earth's surface. Then, they are given a specific tangential velocity, called critical velocity (Vc), which allows them to remain in orbit due to the balance between gravitational pull and their circular motion.
Critical Velocity Formula:
The satellite experiences a centripetal force which keeps it in orbit. This force is provided by the Earth's gravitational attraction.

Orbit of an artificial satellite
Centripetal Force = Gravitational Force
\[\frac{\mathrm{mv_{c}}^{2}}{\mathrm{R+h}}=\frac{\mathrm{GMm}}{(\mathrm{R+h})^{2}}\]
\[\mathrm{v_c}^2=\frac{\mathrm{GM}}{\mathrm{R+h}}\]
\[\mathbf{v}_{\mathrm{c}}=\sqrt{\frac{\mathrm{GM}}{\mathrm{R}+\mathrm{h}}}\]
Where:
- G = 6.67 × 10⁻¹¹ Nm²/kg² (Gravitational constant)
- M = 6 × 10²⁴ kg (Mass of Earth)
- R = 6.4 × 10⁶ m (Earth’s radius)
- h = Height of satellite above Earth
This formula shows that critical velocity does not depend on the mass of the satellite. As height increases, the required critical velocity decreases.
Types of Satellite Orbits
| Type of Orbit | Height Range | Time to Orbit Earth | Characteristics | Common Uses | Examples |
|---|---|---|---|---|---|
| High Earth Orbit (HEO) | Above 35,780 km | ~24 hours | Appears stationary if aligned with equator (Geosynchronous) | Weather monitoring, satellite TV, telephone signals | INSAT, GSAT (Geostationary satellites) |
| Medium Earth Orbit (MEO) | 2,000 km to 35,780 km | 2 to 24 hours | Includes elliptical and polar orbits; suitable for wider Earth coverage | GPS and navigation systems, Earth monitoring | IRNSS (NavIC), GPS satellites |
| Low Earth Orbit (LEO) | 180 km to 2,000 km | ~90 minutes | Fast orbit, close to Earth; ideal for detailed imaging and experiments | Earth observation, research, space stations | ISS, Hubble Space Telescope, Cartosat |
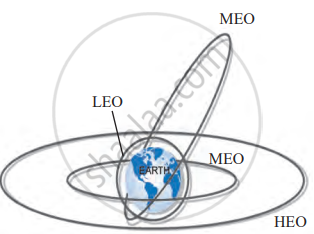
Orbits of satellites
Key Points: Orbits of Artificial Satellites
- Satellites are placed into orbit using launchers and require a specific tangential speed (the critical velocity).
- Low Earth Orbits (180–2000 km) are used for space stations and weather studies.
- Medium Earth Orbits (2000–35780 km) are used for GPS and polar satellites.
- High Earth Orbits (≥35780 km) include geostationary satellites used for TV, phone, and weather.
- The higher the orbit, the lower the speed needed; satellite speed doesn’t depend on its mass.
Related QuestionsVIEW ALL [33]
Note the relationship between the entries in all the three columns in the table and rewrite the table.
| Column-1 (Location) |
Column-2 Height from the earth’s surface (km) |
Column-3 g (m/s2) |
| Earth’s surface (average) | 8.8 | 0.225 |
| Mount Everest | 36.6 | 9.81 |
| Maximum height ever reached by manmade balloon | 400 | 9.8 |
| Orbit of a typical weather satellite | 35700 | 9.77 |
| Orbit of communication satellite | 0 | 8.7 |
