Topics
Gravitation
- Concept of Gravitation
- Force and Motion
- Centripetal Force
- Kepler’s Laws
- Law of Orbit or Kepler's First Law
- Law of Areas or Kepler's Second Law
- Law of Periods or Kepler's Third Law
- Newton's Universal Law of Gravitation
- Uniform Circular Motion (UCM)
- Earth’s Gravitational Force
- Earth’s Gravitational Acceleration
- Mass and Weight
- Gravitational Waves
- Free Fall
- Gravitational Potential Energy
- Escape Velocity
- Weightlessness in Space
Periodic Classification of Elements
- Classification of Elements
- Dobereiner’s Triads
- Newland's Law of Octaves
- Mendeleev’s Periodic Table
- Insights into Mendeleev’s Periodic Table
- Modern Periodic Law
- The Modern Periodic Table
- Structure of the Modern Periodic Table
- Modern Periodic Table and Electronic Configuration of Elements
- Groups and Electronic Configuration
- Periods and Electronic Configuration
- Periodic Trends in the Modern Periodic Table
- Atomic Size
- Metallic and Non-metallic Characters
- Gradation in Halogen Family
Chemical Reactions and Equations
- Chemical Reaction
- Chemical Equations
- Balancing Chemical Equation
- Types of Chemical Reactions > Combination Reaction
- Types of Chemical Reactions > Decomposition Reaction
- Types of Chemical Reactions > Single Displacement Reaction
- Types of Chemical Reactions > Double Displacement Reaction
- Endothermic and Exothermic Processes
- Rate of Chemical Reaction
- Factors Affecting the Rate of a Chemical Reaction
- Chemical Properties of Carbon Compounds > Oxidation
- Chemical Properties of Carbon Compounds > Reduction
- Corrosion of Metals
- Rancidity
Effects of Electric Current
- Electric Circuit
- Heating Effect of Electric Current
- Magnetic Effect of Electric Current
- Right-hand Thumb Rule
- Applications of Biot-Savart's Law > Magnetic Field at the Centre of a Circular Loop
- Applications of Ampere’s Circuital Law > Magnetic Field of a Long Straight Solenoid
- Force on a Current Carrying Conductor in a Magnetic Field
- Fleming’s Left Hand Rule
- Electric Motor
- Electromagnetic Induction
- Galvanometer
- Faraday's Laws of Electromagnetic Induction
- Fleming’s Right Hand Rule
- Alternating current (AC) and Direct Current (DC)
- Electric Generator
Heat
Refraction of Light
Lenses
- Concept of Lenses
- Images Formed by Convex Lenses
- Images Formed by Concave Lenses
- Sign Convention
- Lens Formula
- Magnification
- Power of a Lens
- Combination of Lenses
- Defects of Vision and Their Corrections > Myopia
- Defects of Vision and Their Corrections > Hypermetropia
- Defects of Vision and Their Corrections > Presbyopia
- Apparent Size of an Object
- Use of Concave Lenses
- Use of Convex Lenses
- Persistence of Vision
Metallurgy
- Physical Properties of Metals
- Physical Properties of Non-metal
- Chemical Properties of Metal
- Reactions of Metals
- Reactivity Series of Metals
- Chemical Properties of Non-metal
- Ionic Compounds
- Metallurgy
- Basic Principles of Metallurgy > Concentration of Ores
- Basic Principles of Metallurgy > Extraction of Metals
- Basic Principles of Metallurgy > Refining of Metals
- Corrosion of Metals
- Prevention of Corrosion
Carbon Compounds
- Bonds in Carbon Compounds
- Carbon: A Versatile Element
- Hydrocarbons
- Straight chains, Branched chains, and Rings of Carbon atoms
- Functional Groups in Carbon Compounds
- Homologous Series
- Nomenclature
- Chemical Properties of Carbon Compounds > Combustion
- Chemical Properties of Carbon Compounds > Oxidation
- Chemical Properties of Carbon Compounds > Addition Reaction
- Chemical Properties of Carbon Compounds > Substitution Reaction
- Ethanol
- Ethanoic Acid
- Macromolecules and Polymers
Space Missions
School of Elements
The Magic of Chemical Reactions
- Chemical Equations
- Types of Chemical Reactions > Combination Reaction
- Types of Chemical Reactions > Decomposition Reaction
- Types of Chemical Reactions > Single Displacement Reaction
- Types of Chemical Reactions > Double Displacement Reaction
- Chemical Properties of Carbon Compounds > Oxidation
- Types of Double Displacement: Neutralization Reaction
The Acid Base Chemistry
- Properties of Acids > Physical Properties
- The pH Scale
- Acids, Bases and Their Reactivity
- Acid or a Base in a Water Solution
- Preparation and Uses of Baking Soda
- Preparation and Uses of Bleaching Powder
- Preparation and Uses of Washing Soda
- Preparation and Uses of Plaster of Paris
- Chemicals from Common Salt - Soap as a Salt
The Electric Spark
All about Electromagnetism
- Magnetic force
- Bar Magnet and Solenoid Analogy
- Right-hand Thumb Rule
- Applications of Biot-Savart's Law > Magnetic Field at the Centre of a Circular Loop
- Applications of Ampere’s Circuital Law > Magnetic Field of a Long Straight Solenoid
- Force on a Current Carrying Conductor in a Magnetic Field
- Electric Motor
- Electromagnetic Induction
- A.C. Generator
- Simple D.C. Motor
- Household Electrical Circuits
Wonders of Light 1
Wonders of Light 2
Striving for better Environment 1
- Abatement of Pollution
- Sustainable Use of Resources
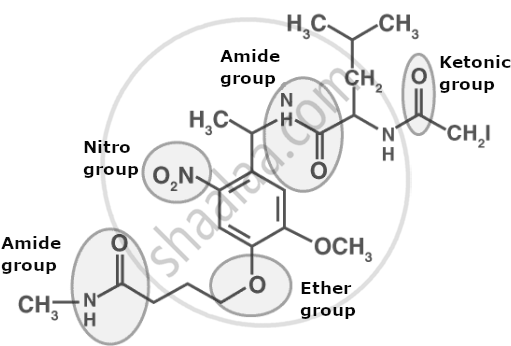
- Definition: Functional Groups
Functional Groups
Carbon compounds are formed by bonding carbon with other elements like hydrogen, halogens, oxygen, nitrogen, and sulphur. When one or more hydrogen atoms in a hydrocarbon are replaced by these elements or groups containing them, the resulting compound exhibits specific chemical properties. These specific atoms or groups responsible for the characteristic behaviour of a compound are called Functional Groups.
'A functional group is an atom or group of atoms that determines the chemical properties of a molecule, regardless of its carbon chain length.'
Key Characteristics of Functional Groups:
- Defines Chemical Reactions: Determines how the molecule reacts.
- Independent Behaviour: Reacts in a similar way across different molecules.
- Predictable Properties: Helps in systematic synthesis and identification of compounds.
- Affects Solubility: A functional group influences polarity and solubility (e.g., alcohols dissolve in water due to the -OH group).
- Can Carry Charges: Some functional groups exist in ionic forms (e.g., -COO⁻ in carboxylates).
- Affect Molecular Structure & Reactivity: Influence how molecules interact with other substances.
Types of Functional Groups
1. Hydrocarbon Functional Groups
- Contain only carbon (C) and hydrogen (H).
- Classified based on bond types between carbon atoms.
| Functional Group | Structural Formula | Prefix | Suffix | Example |
|---|---|---|---|---|
| Alkane | R-H | None | -ane | Methane (CH₄) |
| Alkene | R=R | None | -ene | Ethene (C₂H₄) |
| Alkyne | R≡R | None | -yne | Ethyne (C₂H₂) |
| Aromatic (Benzene) | C₆H₆ | None | -benzene | Toluene (C₆H₅CH₃) |
2. Halogen-Containing Functional Groups
A halogen (Cl, Br, I, F) replaces hydrogen in hydrocarbons.
| Functional Group | Structural Formula | Prefix | Suffix | Example |
|---|---|---|---|---|
| Haloalkane (Alkyl Halide) | R-X (X = Cl, Br, I, F) | Halo- | -halide | Chloroethane (C₂H₅Cl) |
3. Oxygen-Containing Functional Groups
These contain oxygen in different bonding patterns.
| Functional Group | Structural Formula | Prefix | Suffix | Example |
|---|---|---|---|---|
| Alcohol | R-OH | Hydroxy- | -ol | Ethanol (C₂H₅OH) |
| Aldehyde | R-CHO | Formyl- | -al | Ethanal (CH₃CHO) |
| Ketone | R-CO-R' | Oxo- | -one | Propanone (Acetone) (CH₃COCH₃) |
| Carboxylic Acid | R-COOH | Carboxy- | -oic acid | Ethanoic Acid (CH₃COOH) |
| Ester | R-COO-R' | Alkoxycarbonyl- | -oate | Methyl Ethanoate (CH₃COOCH₃) |
| Ether | R-O-R' | Alkoxy- | -ether | Dimethyl Ether (CH₃OCH₃) |
4. Nitrogen-Containing Functional Groups
These groups contain nitrogen in different bonding structures.
| Functional Group | Structural Formula | Prefix | Suffix | Example |
|---|---|---|---|---|
| Amine (Primary) | R-NH₂ | Amino- | -amine | Methylamine (CH₃NH₂) |
| Amine (Secondary) | R₂NH | - | -amine | Dimethylamine (CH₃NHCH₃) |
| Amine (Tertiary) | R₃N | - | -amine | Trimethylamine (N(CH₃)₃) |
| Amide | R-CO-NH₂ | Carbamoyl- | -amide | Ethanamide (CH₃CONH₂) |
| Nitrile | R-CN | Cyano- | -nitrile | Ethanenitrile (CH₃CN) |
5. Sulfur-Containing Functional Groups
These contain sulphur, affecting properties like smell and reactivity.
| Functional Group | Structural Formula | Prefix | Suffix | Example |
|---|---|---|---|---|
| Thiol (Mercaptan) | R-SH | Mercapto- | -thiol | Methanethiol (CH₃SH) |
| Sulfide (Thioether) | R-S-R' | - | -sulphide | Dimethyl Sulfide (CH₃SCH₃) |
Some functional groups in carbon compounds:
| Hetero Atom | Functional Group | Structural Formula | Condensed Structural Formula |
| Halogen (chlorine, bromine, iodine) | Halo (chloro/bromo/iodo) | -X (-Cl, -Br, -I) | -X (-Cl, -Br, -I) |
| Oxygen | 1. Alcohol | -O-H | -OH |
| 2. Aldehyde | \[\begin{array}{cc} \ce{O}\phantom{..}\\ ||\phantom{..}\\ \phantom{}\ce{- C - H} \end{array}\] |
-CHO | |
| 3. Ketone | \[\begin{array}{cc} \ce{O}\phantom{}\\ ||\phantom{}\\ \phantom{}\ce{- C -} \end{array}\] |
-CO- | |
| 4. Carboxylic Acid | \[\begin{array}{cc} \ce{O}\phantom{......}\\ ||\phantom{......}\\ \phantom{}\ce{- C - O - H} \end{array}\] |
-COOH | |
| 5. Ether | -O- | -O- | |
| 6. Ester | \[\begin{array}{cc} \ce{O}\phantom{.....}\\ ||\phantom{.....}\\ \phantom{}\ce{- C - O -} \end{array}\] |
-COO- | |
| Nitrogen | Amines | \[\begin{array}{cc} \phantom{}\ce{- N - H}\\ |\phantom{..}\\ \ce{H}\phantom{..}\\ \end{array}\] |
NH₂ |
Role of Functional Groups in Organic Chemistry
1. Chemical Reactivity
Functional groups determine the reactivity of molecules. They can modify each other’s behaviour and undergo interconversion (e.g., alcohol to aldehyde or carboxylic acid).
2. Bonding and Structural Influence
Covalent bonds link functional groups to molecules. In polymers, they attach to the carbon backbone, altering chemical properties. Some functional groups exist in ionic forms (e.g., -COO⁻ in carboxylates).
3. Coordination Complexes
Functional groups act as ligands, binding to metal centres, affecting stability, colour, and reactivity.
4. Solubility & Polarity
Functional groups influence solubility based on polarity. Similar functional groups in solute and solvent increase solubility (e.g., sugar dissolves in water due to -OH). Electronegative groups can induce polarity in nonpolar molecules, enhancing water solubility.
5. Molecular Interactions
Functional groups contribute to hydrogen bonding, dipole interactions, and ionic forces, affecting boiling/melting points and biological activity.

Definition: Functional Groups
"The hetero atoms or the groups of atoms containing hetero atoms are called functional groups."
Related QuestionsVIEW ALL [31]
Complete the given chart with writing the correct functional group of carbon compounds.
(Ester, Aldehyde, Ketone, Carboxylic acid, Alcohol, Ether)
| -O-H | |
| \[\begin{array}{cc} \ce{O}\phantom{..}\\ ||\phantom{..}\\ \ce{-C-H} \end{array}\] |
|
| \[\begin{array}{cc} \ce{O}\\ ||\\ \ce{-C-} \end{array}\] |
|
| \[\begin{array}{cc} \ce{O}\phantom{.....}\\ ||\phantom{.....}\\ \ce{-C-O-H} \end{array}\] |
|
| -O- | |
| \[\begin{array}{cc} \ce{O}\phantom{..}\\ ||\phantom{..}\\ \ce{-C-O} \end{array}\] |
|
|
\[\begin{array}{cc} |
Match the columns.
| Group A | Group B |
| 1. Straight chain hydrocarbon | a) Benzene |
| 2. Branched chain hydrocarbon | b) Propyne |
| 3. Cyclic hydrocarbon | c) Isobutylene |
