- Thermal decomposition involves heating a compound to break it into simpler substances, e.g.,
2Pb(NO₃)₂ ⟶ 2PbO + 4NO₂ + O₂ (brown fumes of NO₂ observed). - Electrolytic decomposition uses electricity to decompose compounds, e.g.,
2H₂O ⟶ 2H₂ + O₂ (Electrolysis of water gives hydrogen and oxygen gases). - Photodecomposition occurs when light energy breaks down a compound, e.g.,
2AgCl ⟶ 2Ag + Cl₂ (white silver chloride turns grey in sunlight). - Thermal decomposition of calcium carbonate produces lime (CaO) and CO₂ gas, which turns lime water milky.
- Decomposition reactions are usually endothermic and require heat, light, or electricity to occur (e.g., the decomposition of ferrous sulphate, calcium carbonate, or sugar).
Topics
Chemical Reactions and Equations
- Chemical Reactions in Daily Life
- Chemical Equations
- Balancing Chemical Equation
- Types of Chemical Reactions > Combination Reaction
- Types of Chemical Reactions > Decomposition Reaction
- Types of Chemical Reactions > Single Displacement Reaction
- Types of Chemical Reactions > Double Displacement Reaction
- Chemical Properties of Carbon Compounds > Oxidation
- The Effects of Oxidation Reactions in Everyday Life
Acids, Bases and Salts
- Acids and Bases in Daily Life
- Acids and Bases in the Laboratory
- Acids and Bases React with Metals
- Reaction of Metal Carbonates with Acids
- Acids and Bases Reaction with each other
- Reaction of Metallic Oxides with Acids
- Reaction of a Non-metallic Oxide with Base
- Common Properties of Acids and Bases
- The pH Scale
- Importance of pH in Everyday Life
- Salts > Family of Salts
- Salts > pH of Salts
- Salts > Chemicals from Common Salt
- Salts > Water in Salt Crystals
Metals and Non-metals
Carbon and its Compounds
- Importance of Carbon
- The Covalent Bond
- Allotropes of Carbon > Diamond
- Allotropes of Carbon > Graphite
- Allotropes of Carbon > Fullerene
- Carbon: A Versatile Element
- Organic Compounds
- Classification of Hydrocarbons
- Carbon Compounds: Chains, Branches, Rings
- Homologous Series
- Nomenclature
- Chemical Properties of Carbon Compounds > Combustion
- Ethanol
- Ethanoic Acid
- Soaps and Detergents
Life Processes
- Life Processes in Living Organisms
- Nutrition
- Autotrophic Nutrition
- Heterotrophic Nutrition
- Nutrition in Human Beings
- Dental Caries
- Production of ATP
- Blood Circulatory System
- Blood Vessels Entering and Leaving The Heart
- Valves of the Heart
- Tissue Fluid (Or Intercellular Fluid)
- Transportation in Plants
- Transportation of Water
- Excretion
- Excretion in Human Beings
- Kidney and Its Internal Structure
- Structure of a Kidney Tubule (Nephrons)
- Dialysis and Artificial Kidney
- Excretion in Plants
- Organ and Body Donation
Control and Co-ordination
How do Organisms Reproduce?
Heredity
Light – Reflection and Refraction
- Light and Its Straight-Line Propagation
- Reflection of Light
- Spherical Mirrors
- Image Formation by Spherical Mirrors
- Representation of Images Formed by Spherical Mirrors
- Image Formation by Concave Mirror
- Image Formation by a Convex Mirror
- Sign Convention for Reflection by Spherical Mirrors
- Ray Optics - Mirror Formula
- Refraction of Light
- Refraction through a Rectangular Glass Slab
- The Refractive Index
- Refraction by Spherical Lenses
- Image Formation by Lenses
- Image Formation in Lenses Using Ray Diagrams
- Sign Convention for Spherical Lenses
- Lens Formula
- Power of a Lens
The Human Eye and the Colourful World
Electricity
Magnetic Effects of Electric Current
- Magnetic Effect of Electric Current
- Applications of Biot-Savart's Law > Magnetic Field due to a Finite Straight Current-Carrying Wire
- Magnetic Field Due to a Current-Carrying Conductor
- Right-hand Thumb Rule
- Applications of Biot-Savart's Law > Magnetic Field at the Centre of a Circular Loop
- Applications of Ampere’s Circuital Law > Magnetic Field of a Long Straight Solenoid
- Force on a Current Carrying Conductor in a Magnetic Field
- Fleming’s Left Hand Rule
- Magnetism in Medicine
- Domestic Electric Circuits
Our Environment
- Definition: Decomposition Reaction
- Definition: Thermal Decomposition
- Key Points: Decomposition Reaction
CBSE: Class 10
Maharashtra State Board: Class 10
Maharashtra State Board: Class 10
Definition: Decomposition Reaction
A single reactant breaks down to give simpler products. This is a decomposition reaction.
OR
The chemical reaction in which two or more products are formed from a single reactant is a decomposition reaction.
CBSE: Class 10
Definition: Thermal Decomposition
When a decomposition reaction is carried out by heating, it is called thermal decomposition.
CBSE: Class 10
Maharashtra State Board: Class 10
Maharashtra State Board: Class 10
Key Points: Decomposition Reaction
Maharashtra State Board: Class 10
Experiment
1. Aim: To observe and understand decomposition reactions, where a single reactant breaks down into two or more simpler substances.
2. Requirements
- Apparatus: evaporating dish, test tubes, bent tube, rubber cork, Bunsen burner.
- Chemicals: sugar, calcium carbonate (CaCO₃), sulphuric acid, freshly prepared lime water.
4. Procedure
Experiment 1: Decomposition of Sugar
- Take some sugar in an evaporating dish.
- Heat it using a Bunsen burner and observe the changes.
- A black residue (carbon) forms, and water (H₂O) vapour is released.
\[\mathrm{C}_{12}\mathrm{H}_{22}\mathrm{O}_{11}\xrightarrow{\mathrm{Heat}}12C+11H_2O\]
Experiment 2: Decomposition of Calcium Carbonate
- Take some calcium carbonate (CaCO₃) in a test tube.
- Fit a bent tube into the test tube using a rubber cork.
- Insert the other end of the bent tube into another test tube containing freshly prepared lime water.
- Heat the calcium carbonate strongly. The lime water turns milky, indicating the presence of carbon dioxide (CO₂) gas.
\[\mathrm{CaCO}_3(s)\xrightarrow{\Delta}\mathrm{CaO}(s)+\mathrm{CO}_2\uparrow\]
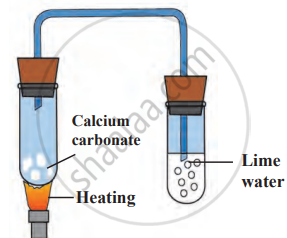
Decomposition of calcium carbonate
Experiment 3: Electrolysis of Water
- Pass electric current through acidulated water.
- Observe the formation of hydrogen (H₂) and oxygen (O₂) gases.
\[2H_2O(l)\xrightarrow{\text{Electrical Energy}}2H_2\uparrow+O_2\uparrow\]
5. Conclusion: A decomposition reaction occurs when a single reactant breaks down into two or more simpler substances. These reactions can be thermal (heat-driven), electrolytic (electricity-driven), or biological (natural degradation by microorganisms). Decomposition reactions are crucial in nature, industry, and energy production, such as in the formation of biogas from organic waste.
Video Tutorials
Shaalaa.com | Types of reaction - Decomposition reaction
to track your progress

