Topics
Chemical Reactions and Equations
- Chemical Reactions in Daily Life
- Chemical Equations
- Balancing Chemical Equation
- Types of Chemical Reactions > Combination Reaction
- Types of Chemical Reactions > Decomposition Reaction
- Types of Chemical Reactions > Single Displacement Reaction
- Types of Chemical Reactions > Double Displacement Reaction
- Chemical Properties of Carbon Compounds > Oxidation
- The Effects of Oxidation Reactions in Everyday Life
Acids, Bases and Salts
- Acids and Bases in Daily Life
- Acids and Bases in the Laboratory
- Acids and Bases React with Metals
- Reaction of Metal Carbonates with Acids
- Acids and Bases Reaction with each other
- Reaction of Metallic Oxides with Acids
- Reaction of a Non-metallic Oxide with Base
- Common Properties of Acids and Bases
- The pH Scale
- Importance of pH in Everyday Life
- Salts > Family of Salts
- Salts > pH of Salts
- Salts > Chemicals from Common Salt
- Salts > Water in Salt Crystals
Metals and Non-metals
Carbon and its Compounds
- Importance of Carbon
- The Covalent Bond
- Allotropes of Carbon > Diamond
- Allotropes of Carbon > Graphite
- Allotropes of Carbon > Fullerene
- Carbon: A Versatile Element
- Organic Compounds
- Classification of Hydrocarbons
- Carbon Compounds: Chains, Branches, Rings
- Homologous Series
- Nomenclature
- Chemical Properties of Carbon Compounds > Combustion
- Ethanol
- Ethanoic Acid
- Soaps and Detergents
Life Processes
- Life Processes in Living Organisms
- Nutrition
- Autotrophic Nutrition
- Heterotrophic Nutrition
- Nutrition in Human Beings
- Dental Caries
- Production of ATP
- Blood Circulatory System
- Blood Vessels Entering and Leaving The Heart
- Valves of the Heart
- Tissue Fluid (Or Intercellular Fluid)
- Transportation in Plants
- Transportation of Water
- Excretion
- Excretion in Human Beings
- Kidney and Its Internal Structure
- Structure of a Kidney Tubule (Nephrons)
- Dialysis and Artificial Kidney
- Excretion in Plants
- Organ and Body Donation
Control and Co-ordination
How do Organisms Reproduce?
Heredity
Light – Reflection and Refraction
- Light and Its Straight-Line Propagation
- Reflection of Light
- Spherical Mirrors
- Image Formation by Spherical Mirrors
- Representation of Images Formed by Spherical Mirrors
- Image Formation by Concave Mirror
- Image Formation by a Convex Mirror
- Sign Convention for Reflection by Spherical Mirrors
- Ray Optics - Mirror Formula
- Refraction of Light
- Refraction through a Rectangular Glass Slab
- The Refractive Index
- Refraction by Spherical Lenses
- Image Formation by Lenses
- Image Formation in Lenses Using Ray Diagrams
- Sign Convention for Spherical Lenses
- Lens Formula
- Power of a Lens
The Human Eye and the Colourful World
Electricity
Magnetic Effects of Electric Current
- Magnetic Effect of Electric Current
- Applications of Biot-Savart's Law > Magnetic Field due to a Finite Straight Current-Carrying Wire
- Magnetic Field Due to a Current-Carrying Conductor
- Right-hand Thumb Rule
- Applications of Biot-Savart's Law > Magnetic Field at the Centre of a Circular Loop
- Applications of Ampere’s Circuital Law > Magnetic Field of a Long Straight Solenoid
- Force on a Current Carrying Conductor in a Magnetic Field
- Fleming’s Left Hand Rule
- Magnetism in Medicine
- Domestic Electric Circuits
Our Environment
- Graphite
- Experiment
- Uses of Graphite
Graphite:
Graphite is a naturally occurring form of carbon found in countries like Russia, New Zealand, America, and India. It is widely used in pencils, where it is mixed with clay, a process discovered by Nicholas Jacques Conte in 1795. Graphite has a unique structure where carbon atoms are bonded in hexagonal layers, allowing the layers to slide over one another, making it versatile and useful in various applications.

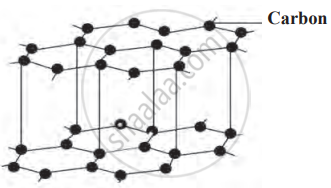
Graphite and structure of carbon atoms in graphite
- Graphite is black, soft, brittle, and has a slippery texture, making it distinct from other forms of carbon.
- It is a good conductor of electricity because free electrons move within each layer of its structure.
- The layered structure enables its use in writing, as layers easily slip off onto paper.
- The density of graphite ranges from 1.9 to 2.3 g/cm³, depending on its form.
- Graphite is highly resistant to most solvents, maintaining its stability in various environments.
- Its structure consists of multiple layers of carbon atoms; one individual layer is referred to as graphene.
- The ability of layers to slide over each other makes graphite an excellent lubricant in industrial applications.
Experiment
1. Aim: To study the physical and electrical properties of pencil lead (graphite) and its behaviour in different liquids.
2. Requirements: pencil lead, electrical wires, battery/cell, small bulb, water, kerosene, test tubes, and lead dust.
3. Procedure
- Remove the lead from a pencil, feel its texture and colour, and check its brittleness by attempting to break it.
- Assemble the electrical circuit using the pencil lead as shown in the diagram and switch on the current. Observe whether the bulb glows.
- Add lead dust to water in one test tube and kerosene in another. Observe its behaviour in both liquids.
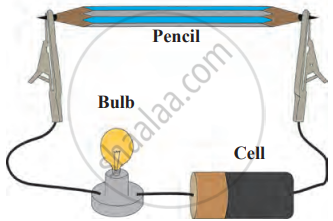
Flow of electric current through graphite
4. Conclusion: Pencil lead (graphite) is brittle, black, and soft. Graphite conducts electricity, as indicated by the glowing bulb in the circuit. Lead dust does not dissolve in water or kerosene, showing its insolubility in common solvents.
Uses of Graphite:
- Graphite is used to make lubricants due to its slippery and layered structure.
- It is used in the production of carbon electrodes because it conducts electricity.
- Graphite is commonly used in pencils for writing, as it easily leaves marks on paper.
- It is an essential ingredient in paints and polishes for adding smoothness and durability.
- Graphite is utilised in arc lamps, which produce very bright light, making them useful in film and photography.
- It is used as a moderator in nuclear reactors due to its ability to slow down neutrons.
- Graphite is applied in industrial moulds and foundries because it can withstand high temperatures.
- It is used in batteries as an electrode material, especially in lithium-ion batteries.
- Graphite is used in brake linings and gaskets for its durability and thermal resistance.
