Topics
Heredity and Evolution
Life Processes in Living Organisms Part -1
- Life Processes in Living Organisms
- Living Organisms and Energy Production
- Glycolysis
- Tricarboxylic Acid Cycle (Citric Acid Cycle or Kreb’s Cycle)
- Phases of Respiration: Electron Transport Chain (Electron Transfer System)
- Production of ATP
- Cellular Respiration
- Energy From Different Food Components
- Cell Division: an Essential Life Process
- Mitosis > Karyokinesis (Division of Nucleus)
- Mitosis > Cytokinesis (Division of Cytoplasm)
- Significance of Mitosis
- Meiosis: Reduction Division
- Stages of Meiosis: Meiosis I
- Stages of Meiosis: Meiosis II
- Significance of Meiosis
- Mitosis Vs Meiosis
Life Processes in Living Organisms Part - 2
Environmental Management
Towards Green Energy
- Energy and Use of Energy
- Generation of Electrical Energy
- Power Plants Based on Nuclear Energy
- Power Plant Based on Thermal Energy
- Power Plants Based on Natural Gas
- Electric Energy Generation and Environment
- Electricity Generation using Hydroelectric Energy
- Electricity Generation using Wind Energy
- Electricity Generation using Solar Energy
- Solar Photovoltaic Cell
- Solar Thermal Power Plant
Animal Classification
- Classification of Living Organisms
- History of Animal Classification
- Traditional Method of Animal Classification
- Conventional System of Animal Classification
- Criteria for New System of Classification > Grades of Organization
- Criteria for New System of Classification > Body Symmetry
- Criteria for New System of Classification > Germ Layers
- Criteria for New System of Classification > Body cavity (Coelom)
- Criteria for New System of Classification > Body Segmentation
- Phylum: Porifera
- Phylum: Cnidaria/Coelenterata
- Phylum: Platyhelminthes
- Phylum: Aschelminthes
- Phylum: Annelida
- Phylum: Arthropoda
- Phylum: Mollusca
- Phylum: Echinodermata
- Phylum: Hemichordata
- Phylum: Chordata
- Subphylum: Urochordata
- Subphylum: Cephalochordata
- Subphylum: Vertebrata/Craniata
- Subphylum: Vertebrata/Craniata > Class: Cyclostomata
- Subphylum: Vertebrata/Craniata > Class: Pisces
- Subphylum: Vertebrata/Craniata > Class: Amphibia
- Subphylum: Vertebrata/Craniata > Class: Reptilia
- Subphylum: Vertebrata/Craniata > Class: Aves
- Subphylum: Vertebrata/Craniata > Class: Mammalia
Introduction to Microbiology
Cell Biology and Biotechnology
- Cell Biology (Cytology)
- Stem Cells
- Stem Cell Research
- Organ Transplantation
- Organ and Body Donation
- Concept of Biotechnology
- Crop Biotechnology > Hybrid Seeds
- Crop Biotechnology > Genetically Modified Organisms (GMOs)
- Crop Biotechnology > Biofertilizers
- Animal Husbandry (Livestock)
- Human Health
- Vaccination and Immunization
- Edible Vaccines
- Treatment
- Interferons
- Gene Therapy
- Cloning
- Industrial Products/White Biotechnology
- Environment and Biotechnology
- Food Biotechnology
- DNA Fingerprinting
- Green Revolution
- White Revolution
- Blue Revolution
- Fertilizers
- Insecticides
- Types of Farming in India > Organic Farming
- Animal Husbandry (Livestock) > Apiculture (Bee Farming)
- Cultivation of Medicinal Plants
- Processing and Preservation of Fruits
Social Health
Disaster Management
Life's Internal Secrets
- Autotrophic Plants
- Heterotrophic Plants
- Human Digestive System
- Nutrition in Plants
- Transport
The Regulators of Life
The Life Cycle
Mapping Our Genes
Understanding Metals and Non-Metals
Striving for Better Environment 2
- Use of Efficient and Eco-friendly Technology
- Sustainable Use of Resources
- Enforcement of Acts, Laws and Policies
Amazing World of Carbon Compounds
- Definition: Aromatic Compounds
- Key Points: Straight Chains, Branched Chains, and Rings of Carbon Atoms
Various Types of Hydrocarbons
| Category | Saturated Hydrocarbons | Unsaturated Hydrocarbons |
| Straight-chain hydrocarbons |
\[\begin{array}{cc} Propane (C₃H₈)
|
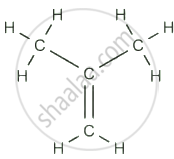
\[\begin{array}{cc} Propene (C₃H₆) \[\begin{array}{cc}\ce{\phantom{.......}H}\\ \phantom{.......}|\\ \ce{H-C≡C-C-H}\\ \phantom{.......}|\\ \ce{\phantom{.......}H}\\ \end{array}\] Propyne (C₃H₄) |
| Branched-chain hydrocarbons |
\[\begin{array}{cc} Isobutane (C₄H₁₀) |
Isobutylene (C₄H₈) |
| Cyclic hydrocarbons |
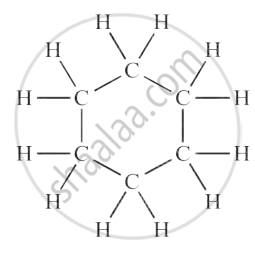
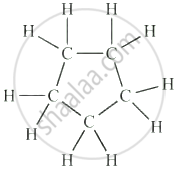
Cyclohexane (C₆H₁₂)
|
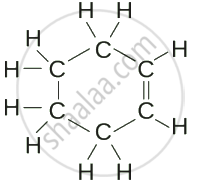
Cyclohexene (C₆H₁₀) 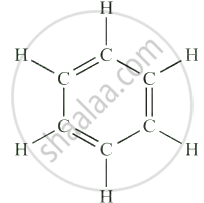 |
Physical Properties of Carbon:
- Carbon can form charcoal when it is heated in the absence of air.
- It is a unique element that exists in various forms, such as coal and ash, which are pure carbon.
- The density of carbon varies based on its form and origin.
- Some forms of carbon are pure, while others, like coal, are mixtures of carbon and hydrogen.
- Carbon exists in multiple allotropic forms, each having distinct physical and chemical properties.
- Allotropes are variations of the same element with different characteristics.
- The colour of carbon is typically soft and appears as a dull grey or black.
- These properties contribute to carbon's versatility and widespread occurrence in nature.
Straight-Chain Hydrocarbons
A straight-chain hydrocarbon consists of carbon atoms joined in a continuous chain without branching. These belong to the alkane family, which contains only single bonds (C–C).
| Straight Chain of Carbon Atoms | Structural Formula | Molecular Formula | Name |
|---|---|---|---|
| C | CH₄ | CH₄ | Methane |
| C–C | H₃C—CH₃ | C₂H₆ | Ethane |
| C–C–C | H₃C—CH₂—CH₃ | C₃H₈ | Propane |
| C–C–C–C | H₃C—(CH₂)₂—CH₃ | C₄H₁₀ | Butane |
| C–C–C–C–C | H₃C—(CH₂)₃—CH₃ | C₅H₁₂ | Pentane |
| C–C–C–C–C–C | H₃C—(CH₂)₄—CH₃ | C₆H₁₄ | Hexane |
| C–C–C–C–C–C–C | H₃C—(CH₂)₅—CH₃ | C₇H₁₆ | Heptane |
| C–C–C–C–C–C–C–C | H₃C—(CH₂)₆—CH₃ | C₈H₁₈ | Octane |
| C–C–C–C–C–C–C–C–C | H₃C—(CH₂)₇—CH₃ | C₉H₂₀ | Nonane |
| C–C–C–C–C–C–C–C–C–C | H₃C—(CH₂)₈—CH₃ | C₁₀H₂₂ | Decane |
These hydrocarbons are saturated (alkanes) because they contain only single bonds between carbon atoms.
Chemical Properties of Carbon:
- Carbon has an atomic number of 6 and an atomic mass of 12.011 g/mol⁻¹.
- Its electronegativity is 2.5, and its density at 20°C is 2.2 g.cm⁻³.
- The melting and boiling points are 3652°C and 4827°C, respectively.
- The Van der Waals radius is 0.091 nm, and the ionic radius is 0.015 nm (+4) or 0.26 nm (-4).
- Its electronic configuration is He 2s² 2p².
- The ionisation energies are 1086.1 kJ.mol⁻¹ (first), 2351.9 kJ.mol⁻¹ (second), and 4618.8 kJ.mol⁻¹ (third).
- Carbon participates in allotropic transformations, such as the conversion between graphite and diamond under high pressure and temperature.
- Carbon is non-metallic and generally unreactive at room temperature but reacts with oxygen at high temperatures to form carbon dioxide (CO₂) or carbon monoxide (CO).
Branched-Chain Hydrocarbons
When a carbon chain has side branches, it forms a branched hydrocarbon. These branches are called alkyl groups, and their presence leads to structural isomerism (same molecular formula, different structure).
Example: Butane (C₄H₁₀) – Two Isomers
| Isomer Type | Structural Formula | Name |
|---|---|---|
| Straight Chain | H₃C—CH₂—CH₂—CH₃ | n-Butane |
| Branched Chain | H₃C—CH—CH₃ | Iso-butane |
This phenomenon is called structural isomerism because the same molecular formula represents different compounds with distinct physical and chemical properties.
Cyclic Hydrocarbons (Rings of Carbon Atoms)
Cyclic hydrocarbons have carbon atoms arranged in a closed-ring structure. They can be:
- Saturated (Cycloalkanes): Only single bonds between carbon atoms.
- Unsaturated (Cycloalkenes & Cycloalkynes): contain double or triple bonds within the ring.
| Cyclic Hydrocarbon | Molecular Formula | Structural Formula |
| Cyclopropane | C₃H₆ | 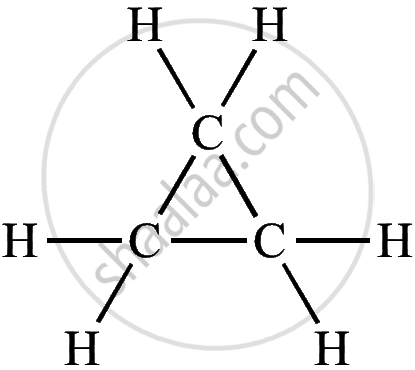 |
| Cyclobutane | C₄H₈ | 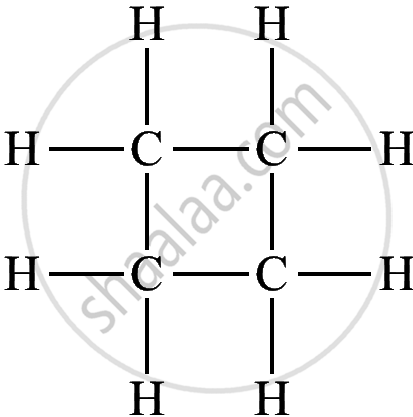 |
| Cyclopentane | C₅H₁₀ | 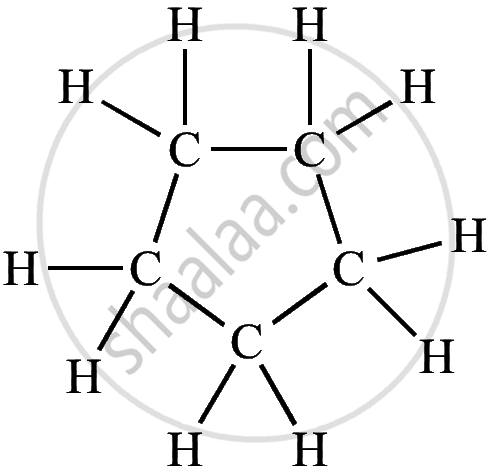 |
| Cyclohexane | C₆H₁₂ | 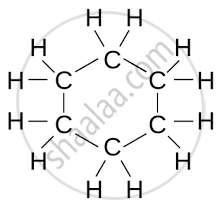 |
| Cyclohexene | C₆H₁₀ | 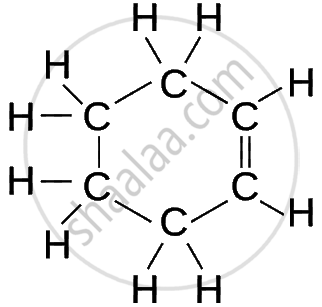 |
| Benzene (Aromatic Hydrocarbon) | C₆H₆ | 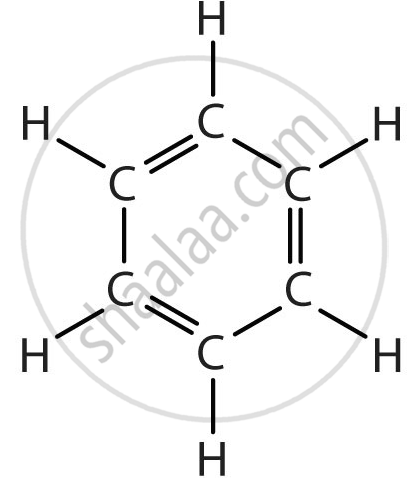 |
Benzene and similar compounds are called aromatic hydrocarbons due to their stability and resonance structure.
Definition: Aromatic Compounds
The compounds having this characteristic unit in their structure are called aromatic compounds.
Key Points: Straight chains, Branched chains, and Rings of Carbon atoms
- Carbon compounds can have straight, branched, or ring chains of carbon atoms.
- Isomers have the same formula but different structures (e.g., butane).
- Crude oil is a mix of hydrocarbons; fuels like LPG and petrol are separated by fractional distillation.
- Saturated hydrocarbons have single bonds, while unsaturated ones have double or triple bonds.
- Aromatic compounds, such as benzene, have rings with alternating double bonds.