Advertisements
Chapters
1: Solid State
▶ 2: Solutions
3: Electrochemistry
4: Chemical Kinetics
5: Surface Chemistry
Inorganic Chemistry
6: General Principles and Processes of Isolation of Elements
7: p-Block Elements
8: d-and ƒ-Block Elements
9: Coordination Compounds
Organic Chemistry
10: Haloalkanes and Haloarenes
Chapter 11: Alcohols, Phenols and Ethers
Chapter 12: Aldehydes, Ketones and Carboxylic Acids
Chapter 13: Organic Compounds containing Nitrogen
Chapter 14: Biomolecules
15: Polymers
Chapter 16: Chemistry in Everyday Life
![Nootan solutions for केमिस्ट्रि [इंग्रजी] इयत्ता १२ आयएससी chapter 2 - Solutions Nootan solutions for केमिस्ट्रि [इंग्रजी] इयत्ता १२ आयएससी chapter 2 - Solutions - Shaalaa.com](/images/chemistry-english-class-12-isc_6:a6407550bf9a46188b122e0d354548f8.png)
Advertisements
Solutions for Chapter 2: Solutions
Below listed, you can find solutions for Chapter 2 of CISCE Nootan for केमिस्ट्रि [इंग्रजी] इयत्ता १२ आयएससी.
Nootan solutions for केमिस्ट्रि [इंग्रजी] इयत्ता १२ आयएससी 2 Solutions REVIEW EXERCISES [Pages 66 - 105]
Define Molarity (M).
Define molality.
Define Normality.
Which of the following units do not vary with change in temperature?
molarity
molality
normality
mole fraction
Calculate the normality and molarity of H2SO4 solution containing 4.9 g of H2SO4 per litre of the solution.
6 g of NaOH are dissolved in 200 cm3 of water. What is the relation between molarity and normality of the solution thus obtained?
Calculate the mass of the solute in the following solution:
100 cm3 of `N/10` KOH
Calculate the mass of the solute in the following solution:
250 cm3 of N H2SO4
Calculate the mass of the solute in the following solution:
250 cm3 of 2M HNO3
Calculate the mass of the solute in the following solution:
150 cm3 of `M/2` HCl
What is the molality of a semimolar NaCl solution if the density of the solution is 1.16 g cm−3?
What is the molality of ammonia in a solution containing 0.85 g of NH3 in 100 cm3 of a liquid of density 0.85 g cm−3?
Calculate the molality of 1 litre solution of 93% H2SO4 (weight/volume). The density of the solution is 1.84 g mL−1.
5.85 g of NaCl are dissolved in 500 cm3 of water. Calculate the formality of the solution.
Calculate the mole fraction of water in a mixture of 12 g of water, 108 g of acetic acid and 92 g of ethyl alcohol.
2.46 g of sodium hydroxide (molar mass = 40) are dissolved in water and the solution is made to 100 cm3 in a volumetric flask. Calculate the molarity of the solution.
Concentrated nitric acid used as a laboratory reagent is usually 69% by mass of nitric acid. Calculate the volume of the solution which contained 23 g of HNO3. Density of conc. HNO3 solution is 1.41 g cm−3.
100 cm3 of a centimolar solution of an acid contain 0.098 g of the acid. Find the molecular mass of the acid.
Calculate the mole fraction of ethyl alcohol and water in a solution in which 46 g of ethyl alcohol and 180 g of water are mixed together.
The density of 3M aqueous solution of sodium thiosulphate is 1.25 g/ml. Calculate
- mole fraction of sodium thiosulphate
- molalities of Na+ and \[\ce{S2O^2-_3}\] ions.
8.0575 × 10−2 kg of Glauber’s salt are dissolved in water to obtain 1 dm3 of a solution of density 1077.2 kg m−3. Calculate the molarity, molality, and mole fraction of Na2SO4 in the solution.
The mole fraction of benzene in a solution in toluene is 0.40. Calculate the weight percent of benzene in the solution.
A solution contains 410.3 g of H2SO4 per litre of the solution at 20°C. If its density is 1.243 g cm−3, what will be its molality and molarity?
The solubility of Ba(OH)2.8H2O in water at 288 K is 5.6 g per 100 g of water. What is the molality of the hydroxide ions in the saturated solution of barium hydroxide at 288 K? (Atomic masses: Ba = 137, O = 16, H = 1)
Calculate molality of 1 litre solution of 90% H2SO4 (weight/volume). The density of solution is 1.80 g mL−1.
Calculate the number of molecules of oxalic acid (H2C2O4·2H2O) in 100 ml of 0.2N oxalic acid solution.
150 cm3 of a decimolar NaOH solution are diluted to 750 cm3. Find the molarity of the diluted solution.
One tonne of air contains 2 × 10−3 g of carbon as smoke. Calculate the concentration of carbon in ppm in air.
A sample of hard water is found to contain 50 mg of CaSO4 in 12 kg of the sample. Calculate the hardness of the sample in terms of ppm of CaSO4.
A 6.90 M solution of KOH in water contains 30% by weight of KOH. What is the density of the solution?
Define vapour pressure of a liquid.
What is the effect on vapour pressure when a non-volatile solute is added to it?
Why is the vapour pressure of an aqueous solution of glucose lower than that of water?
What does an ideal solution mean at molecular level?
State Raoult’s law.
Distinguish between ideal and non-ideal solutions.
Mention any three characteristics of an ideal solution.
What does cause deviation from ideal behaviour?
State Raoult’s law for the solutions of non-volatile solutes in volatile solvents.
How does Raoult’s law depend upon temperature?
Explain why some of the non-ideal solutions show positive deviations, while some others show negative deviations from ideal behaviour.
Benzene and toluene form nearly ideal solution. At 313 K, the vapour pressure of pure benzene is 150 mm Hg and of pure toluene is 50 mm Hg. Calculate the vapour pressure of a mixture of these two containing their equal masses at 313 K.
Benzene (C6H6) and toluene (C6H5CH3) form an ideal solution. At 313 K, the vapour pressures of pure benzene and toluene are 160 mm Hg and 60 mm Hg respectively. Calculate the partial pressures of benzene and toluene and the total pressure of the solution when equal masses of benzene and toluene are mixed at 313 K.
Benzene (C6H6) and toluene (C6H5CH3) form an ideal solution. At 313 K, the vapour pressures of pure benzene and toluene are 160 mm Hg and 60 mm Hg respectively. Calculate the partial pressures of benzene and toluene, and the total pressure of the solution when equal moles of benzene and toluene are mixed at 313 K.
Benzene (C6H6) and toluene (C6H5CH3) form an ideal solution. At 313 K, the vapour pressures of pure benzene and toluene are 160 mm Hg and 60 mm Hg respectively. Calculate the partial pressures of benzene and toluene and the total pressure of the solution when 1 mole of benzene and 4 moles of toluene are mixed at 313 K.
Methanol and ethanol form a nearly ideal solution at 300 K. A solution is made by mixing 32 g of methanol and 23 g of ethanol at 300 K. Calculate the partial pressures of its constituents and the total pressure of the solution (At 300 K, \[\ce{p^\circ_{CH_3OH}}\] = 90 mm Hg, \[\ce{p^\circ_{C_2H_5OH}}\] = 51 mm Hg)
The vapour pressures of benzene and toluene at 20°C are 75 mm and 22 mm Hg respectively. 23.4 g of benzene and 64.4 g of toluene are mixed. If the two form an ideal solution, calculate the mole fraction of benzene in vapour phase when vapours are in equilibrium with the liquid phase.
At 25°C, the vapour pressure of water is 23.75 mm Hg. Calculate the vapour pressure at the same temperature of 5% aqueous solution of urea (NH2CONH2).
The vapour pressure of water at 20°C is 17 mm Hg. Calculate the vapour pressure of a solution containing 6 g of glucose in 50 g of water.
Solutions of two volatile liquids A and B obey Raoult’s law. At a certain temperature, it is found that when the total pressure above a given solution is 400 mm Hg, the mole fraction of A in the vapour is 0.45 and in the liquid it is 0.65. What are the vapour pressures of the two liquids in the pure state at the given temperature?
State Raoult’s law for the solutions of non-volatile solutes in volatile solvents.
Derive a mathematical expression for Raoult’s law for solutions of non-volatile solutes in volatile solvents.
What are non-ideal solutions?
Explain as to why non-ideal solutions deviate from Raoult’s law.
Carbon tetrachloride and water are immiscible, whereas ethanol and water are miscible in all proportions. Correlate this behaviour with molecular structures of these compounds.
State Henry’s law.
Two liquids X and Y boil at 110°C and 130°C respectively. Which one of them has higher vapour pressure at 50°C?
A and B liquids on mixing produce a warm solution. Which type of deviation from Raoult’s law is there?
State the formula relating the pressure of a gas with its mole fraction in a liquid in contact with it.
What are colligative properties?
Explain two colligative properties.
Why is the vapour pressure of an aqueous solution of glucose lower than that of water?
How is the molar mass of a non-volatile substance determined from the lowering of vapour pressure?
Why does the boiling point of a solvent increase by the presence of a solute in it?
What is the molal elevation constant, Kb?
How is the molal elevation constant, Kb is related to the molality of a solution?
The outer shells of two eggs are removed. One of the eggs is placed in pure water and the other is placed in saturated solution of sodium chloride. What will be observed and why?
Why does sodium chloride solution freeze at lower temperature than water, but boils at higher temperature than water?
Define osmotic pressure.
How does osmotic pressure depend on temperature and atmospheric pressure?
Show that the relative lowering of vapour pressure is a colligative property.
Why is the osmotic pressure considered to be a colligative property?
The vapour pressure of a 5% aqueous solution of a non-volatile organic substance at 373 K is 745 mm Hg. Calculate the molar mass of the substance.
A solution containing 34.2 g of sugar dissolved in 1 litre of water has an osmotic pressure of 2.522 atmospheres at 20°C. Calculate the value of gas constant, R.
The vapour pressure of a dilute aqueous solution of a non-volatile solute (molar mass 180 g mol−1) at 373 K is 750 mm Hg. Calculate
- molality
- mole fraction of solute.
The vapour pressure of pure benzene at a certain temperature is 640 mm Hg. A non-volatile solute weighing 2.175 g is added to 39.0 g of benzene. The vapour pressure of the solution is 600 mm Hg. What is the molar mass of the solute?
What weight of glucose (mol. wt. = 180) should be added to 1700 g of water at 20°C to lower its vapour pressure by 0.001 mm? The vapour pressure of water at 20°C is 17 mm Hg.
A current of dry air was passed through a solution of 2.5 g of a non-volatile solute in 100 g of water and through water alone. The loss in the weight of solution was 1.25 g and that of water was 0.005 g. Calculate the molecular mass of the solute.
The molal elevation constant for water is 0.56°C per kg of water. Calculate the boiling point of solution made by dissolving 6 g of urea in 200 g of water.
0.72 g of camphor in 32 g of acetone produces an elevation of 0.25°C in the boiling point of acetone. Calculate the molecular mass of camphor. (Kb for acetone = 1.72 K kg mol−1)
A solution of 12.5 g urea in 170 g of water gave a boiling point elevation of 0.63 K. Calculate the molar mass of urea taking Kb = 0.52 K/m.
An aqueous solution of glucose boils at 100.02°C. What is the number of glucose molecules in the solution containing 100 g of water? What will be the osmotic pressure of this glucose solution at 27°C?
(Given: Kb for water = 0.5 K kg mol−1, R = 0.082 L atm mol−1 K−1, Avogadro’s constant, NA = 6.02 × 1023 mol−1)
An aqueous solution freezes at 272.4 K, while pure water freezes at 273.0 K. Determine
- the molality of the solution
- boiling point of the solution
- lowering of vapour pressure of water at 298 K.
(Given: Kf = 1.86 K kg mol−1, Kb = 0.512 K kg mol−1 and vapour pressure of water at 298 K = 23.756 mm Hg)
A solution of sucrose has been prepared by dissolving 68.4 g of sucrose in one kg of water. Calculate the following:
- The vapour pressure of the solution at 298 K.
- Osmotic pressure of the solution at 298 K.
- Freezing point of the solution.
(Given: Vapour pressure of water at 298 K = 0.024 atm, Kf for water = 1.86 K kg mol−1, R = 0.0821 L atm mol−1 K−1)
A solution containing 0.513 g of naphthalene (molar mass = 128) in 50 g of CCl4 gives a boiling point elevation of 0.402°C, while a solution of 0.625 g of an unknown solute gives a boiling point elevation of 0.650°C. Find the molecular mass of the unknown solute.
A solution of 2.95 g of sulphur in 100 g of cyclohexane had a freezing point of 4.18°C. The freezing point of pure cyclohexane is 6.50°C. What is the molecular formula of sulphur in the solution?
A solution containing 18 g of a non-volatile solute in 200 g of water freezes at 272.07 K. Calculate the molecular mass of the solute.
(Given: Kf = 1.86 K/m)
Find the molality of a water solution which freezes at 263.15 K.
(Given: Kf of water = 1.86 K kg mol−1)
In a cold climate, water gets frozen causing damage to the car radiators. Ethylene glycol is used as an antifreeze. Calculate the amount of ethylene glycol to be added to 4 kg of water to prevent it from freezing at −6°C. (Kf for water = 1.85 K kg mol−1)
The molal freezing point constant of benzene is 4.90 and its melting point is 5.51°C. A solution of 0.816 g of a compound A when dissolved in 7.5 g of benzene freezes at 1.59°C. Determine the molecular weight of A.
The latent heat of fusion of benzene is 30.25 cal/g. 0.0802 g of a substance when dissolved in 13.03 g of benzene lower the freezing point by 0.490°C. If pure benzene freezes at 5.4°C, calculate the molecular mass of the substance.
Calculate the osmotic pressure of 0.01 M solution of cane sugar at 27°C. (R = 0.0821 L atm K−1 mol−1)
Calculate the concentration of a solution of glucose which is isotonic at the same temperature with a solution of urea containing 6.2 g/L.
Osmotic pressure of a solution containing 7.0 g of a protein in 100 ml of solution is 20 mm Hg at 37°C. Calculate the molecular mass of the protein.
(R = 0.0821 L atm K−1 mol−1)
Calculate the freezing point of an aqueous solution of a non-electrolyte having osmotic pressure of 2.0 atm at 300 K.
(Kf = 1.86 K kg mol−1, R = 0.0821 L atm K−1 mol−1)
A solution of sucrose (mol. wt. 342) is prepared by dissolving 68.4 g of it per litre of solution. What is the osmotic pressure at 300 K?
(R = 8.314 k Pa dm3 K−1 mol−1)
Osmotic pressure of a solution containing 2 g of a protein (soluble in water) per 300 cm3 of the solution is 20 mm of Hg at 27°C. Calculate the molecular mass of protein.
(R = 0.0821 L atm K−1 mol−1)
The osmotic pressure of blood at 37°C is 8.21 atm. How much glucose in grams should be used per litre of aqueous solution for an intravenous injection so that it is isotonic with blood? (Molecular wt of glucose = 180 g/mol)
The vapour pressure of a solution of urea is 736.2 mm at 100°C. Calculate the osmotic pressure of this solution at 15°C.
The boiling point of a glucose solution containing 12 g of glucose in 100 g of water is 100.34°C. Calculate the molal elevation constant of water. Boiling point of water is 100°C.
A mixture of 1 g naphthalene (Mol. mass 128) and 10 g camphor melts at 147°C whereas pure camphor melts at 177.5°C. What is cryoscopic constant (Kf) of camphor?
0.90 g of a non-electrolyte was dissolved in 87.90 g of benzene. This raised the boiling point of benzene by 0.25°C. If the molecular mass of the non-electrolyte is 102.0 g mol−1, calculate the molal elevation constant for benzene.
How is it that the boiling point of the following solutions in water are different:
- 0.1 M NaCl solution
- 0.1 M sugar solution?
Two elements A and B form compounds having formula AB2 and AB4. When dissolved in 20 g of benzene (C6H6), 1 g of AB2 lowers the freezing point by 2.3 K whereas 1.0 g of AB4 lowers it by 1.3 K. The molar depression constant for benzene is 5.1 K kg mol−1. Calculate the atomic masses of A and B.
A 0.1539 molal aqueous solution of cane sugar (mol mass = 342 g mol−1) has a freezing point of 271 K while the freezing point of pure water is 273.15 K. What will be the freezing point of an aqueous solution containing 5 g of glucose (mol. mass = 180 g mol−1) per 100 g of solution?
What is meant by abnormal molecular mass? Illustrate it with an example.
What is the cause of obtaining abnormal molecular masses?
What is van’t Hoff factor?
What type of values can van’t Hoff factor have if in solution the solute molecules undergo association?
What type of values can van’t Hoff factor have if in solution the solute molecules undergo dissociation?
What is van’t Hoff factor?
How does van’t Hoff factor modify the equation for the elevation of boiling point?
Assuming complete dissociation, calculate the expected freezing point of a solution prepared by dissolving 6.00 g of Glauber’s salt, Na2SO4 . 10H2O in 0.100 kg of water. (Given: Kf for water = 1.86 K kg mol−1, atomic masses: Na = 23, S = 32, O = 16, H = 1 amu)
The freezing point depression of 0.1 molal solution of acetic acid in benzene is 0.25 K. Kf for benzene is 5.12 K kg mol−1. What conclusion can you draw about the molecular state of acetic acid in benzene?
When 2 g of benzoic acid is dissolved in 25 g of benzene, it shows a depression in freezing point equal to 1.62 K. The molal depression constant (Kf) of benzene is 4.9 K kg mol−1, and the molecular weight of benzoic acid is 122 g/mol. What will be the percentage association of the benzoic acid?
(Benzoic acid forms dimers when dissolved in benzene.)
Calculate the van’t Hoff factor of CdSO4 (molecular mass = 208.4) if the dissociation of 5.21 g of CdSO4 in half litre water gives a depression in freezing point of 0.168°C. (Kf of water is 1.86 K kg mol−1)
A 0.1 M solution of NaCl is found to be isotonic with a 1% urea solution. Calculate:
- van’t Hoff factor, and
- degree of dissociation of NaCl.
Calculate the boiling point of a solution containing 0.61 g of benzoic acid in 50 g of carbon-disulphide assuming 84% dimerisation of the acid. The boiling point and Kb of CS2 are 46.2°C and 2.3 K kg mol−1 respectively.
A decimolar solution of potassium ferrocyanide is 50% dissociated at 300 K. Calculate the osmotic pressure of the solution.
(Given: R = 8.314 J K−1 mol−1)
Why do we get abnormal molecular masses of the substances using colligative properties of solutions only sometimes? What are the nature of these abnormalities?
A solution contains 0.8960 g K2SO4 in 500 mL solution. Its osmotic pressure is found to be 0.690 atm at 27°C. Calculate the value of van’t Hoff factor.
The freezing point of a solution containing 0.3 gms of acetic acid in 30 gms of benzene is lowered by 0.45 K. Calculate the Van’t Hoff factor. (at. wt. of C = 12, H = 1, O = 16, Kf for benzene = 5.12 K kg mole−1).
An aqueous solution containing 1.248 g of barium chloride (molar mass = 208.34 g mol−1) in 100 g of water boils at 100.0832°C. Calculate the degree of dissociation of barium chloride. (Kb for water = 0.52 K kg mol−1)
What is van’t Hoff factor?
The osmotic pressure of a 0.0103 molar solution of an electrolyte is found to be 0.70 atm at 27°C. Calculate the van't Hoff factor. (R = 0.082 L atm mol−1 K−1). What conclusion do you draw about the molecular state of the solute in the solution?
Give an example of a solution containing a solid solute in a solid solvent.
When 2 g of benzoic acid is dissolved in 25 g of benzene, it shows a depression in freezing point equal to 1.62 K. The molal depression constant (Kf) of benzene is 4.9 K kg mol−1, and the molecular weight of benzoic acid is 122 g/mol. What will be the percentage association of the benzoic acid?
(Benzoic acid forms dimers when dissolved in benzene.)
How is it that the boiling point of the following solutions in water are different:
- 0.1 M NaCl solution
- 0.1 M sugar solution?
What would be the value of van’t Hoff factor for a dilute solution of K2SO4 in water?
Nootan solutions for केमिस्ट्रि [इंग्रजी] इयत्ता १२ आयएससी 2 Solutions VERY SHORT ANSWER TYPE QUESTIONS [Pages 109 - 110]
What is meant by a solution?
What is meant by the binary solution?
What are the criteria for two liquids to be completely miscible?
What are the criteria for two liquids to be partially miscible?
On which factors does the solubility of a gas in a liquid depend?
Define the term solute.
Define Solvent.
Why is NaCl soluble in water but not in benzene?
Define the term mole fraction giving one example.
What is the difference between molality and molarity?
Explain the term mass fraction.
Explain the term mole fraction.
Why does the molality of a solution remain unchanged with temperature?
Why does the molarity of a solution depend upon temperature?
Why does normality depend upon temperature?
What do you understand by a decimolar solution?
What is formality?
What are the units of formality?
What do you understand by the normality of a solution?
Among (M/10) H2SO4 and (N/10) H2SO4, which one is more concentrated?
What is the relationship between molarity and normality of a solution?
Does the mole fraction of a substance in a solution vary with temperature?
Define vapour pressure of a liquid.
What happens to the vapour pressure of a liquid when a non volatile solute is dissolved in it?
State Raoult’s law for solutions containing volatile solutes.
State Raoult’s law for the solutions of non-volatile solutes in volatile solvents.
Define the term:
Ideal solution
Draw the vapour pressure versus composition diagram for an ideal solution.
Define a non-ideal solution.
For a non-ideal solution, explain why `V_"mixing" ne 0`.
For a non-ideal solution, explain why `Delta H_"mixing" ne 0`.
Which type of behaviour is expected on adding water to sulphuric acid?
What are colligative properties?
Show that the relative lowering of vapour pressure is a colligative property.
Define boiling point of a liquid.
What do you understand by elevation of boiling point?
Why is common salt added to water used for boiling eggs?
Define the following term:
Molal elevation constant (Kb)
Mention units of molal elevation constant.
Write the expression that correlates molal elevation constant to molar enthalpy of vaporisation.
What is the relationship between molal elevation constant and the elevation of boiling point of a solution?
Does the molal elevation constant depend upon the nature of solute?
Why does the boiling point of a solvent increase by the presence of a solute in it?
Define Freezing point.
What do you understand by depression of freezing point?
Define Cryoscopic constant.
Write the SI unit of the cryoscopic constant.
Write the expression which correlates ΔTf with Kf.
How is the latent heat of fusion per gram of the solvent related to molal depression constant?
Name the solid that separates first when a salt solution is slowly frozen.
What is a semipermeable membrane?
What happens when two solutions of different concentrations are separated by a semipermeable membrane?
Why is the osmotic pressure considered to be a colligative property?
Define Osmotic pressure.
What is reverse osmosis?
State Boyle-van’t Hoff law.
What are isotonic solutions?
What do you understand by plasmolysis?
State whether a saturated salt solution is hypertonic or hypotonic to a dilute salt solution.
What happens when red blood corpuscles (RBCs) are placed in distilled water?
What is meant by abnormal molecular mass?
When do we get a higher molecular mass of a substance as compared to the normal value for the substance?
Define van’t Hoff factor.
What is the value of van’t Hoff factor for KCl in dilute aqueous solutions?
Which has the lowest freezing point and why?
1 molar urea, 1 molar NaCl or 1 molar sodium sulphate.
What is the value of van’t Hoff factor when a substance neither dissociates nor associates in the solution?
How is ΔTb related to Kb for the substance whose molecular state changes in the solution?
Nootan solutions for केमिस्ट्रि [इंग्रजी] इयत्ता १२ आयएससी 2 Solutions SHORT ANSWER TYPE QUESTIONS [Pages 110 - 111]
What do you understand by the term solvent with reference to a solution?
What do you understand by the term solute with reference to a solution?
What is ion-dipole interaction?
How is an ion-dipole interaction responsible for the solubility of an ionic solid in water?
Explain why benzene is not soluble in water.
State Henry’s law.
Define Molarity (M).
Define molality.
Define formality.
Define Mole fraction.
Why is the unit molality preferred over molarity?
What do you understand by semi-molar solutions?
What do you understand by semi-normal solutions?
Among 1 M and 1 m solutions of glucose, which one is more concentrated and why?
Which of the following concentration units are dimensionless?
Weight percentage, volume percentage, molarity, molality and mole fraction.
Which of the following units vary with temperature and why?
Normality, Molarity, Molality, Formality and Mole fraction.
Define vapour pressure of a liquid.
What happens to the vapour pressure of a liquid when a volatile solute dissolves in it?
What happens to the vapour pressure of a liquid when a non volatile solute is dissolved in it?
How can you justify the observation that the vapour pressure of a solution of a non-volatile solute in a given solvent is less than that of the pure solvent? Also state the law related to this observation.
State Raoult’s law for the solutions of non-volatile solutes in volatile solvents.
Derive a mathematical expression for Raoult’s law for solutions of non-volatile solutes in volatile solvents.
Show that the relative lowering of vapour pressure for a solution is equal to the mole fraction of solute when solvent alone is volatile.
On which factors does the vapour pressure of a liquid depend?
What is an ideal solution?
Mention any three characteristics of an ideal solution.
When 1 mole of A is mixed with 1 mole of B, the temperature of the solution increases. Is it an ideal solution? If not, why?
Why do certain solutions show positive deviation from Raoult’s law?
Which type of deviation (positive or negative) from ideal behaviour will be shown by the solution of cyclohexane and ethanol? Give suitable reasons.
State Raoult’s law.
Distinguish between ideal and non-ideal solutions.
Why do certain solutions show negative deviation from Raoult’s law?
Distinguish between ideal and non-ideal solutions.
What is an ideal solution?
What are non-ideal solutions?
Give reasons for the formation of ideal solutions.
Give reasons for the formation of non-ideal solutions.
Give one example of an ideal solution.
Give one example of non-ideal solutions.
What are colligative properties?
Explain two colligative properties.
What do you understand by lowering of vapour pressure and relative lowering of vapour pressure? Show that these are colligative properties.
Define the following term:
Boiling point
Define elevation of boiling point.
Why does the boiling point of a liquid get elevated when a non-volatile solute is dissolved in it?
If ΔTb is the elevation of boiling point and m is the molality of solution, show that ΔTb = Kbm.
Define the following term:
Molal elevation constant (Kb)
How is the molal elevation constant related to the latent heat of vaporisation of the solvent?
Does the molal elevation constant depend upon the nature of solute?
Derive the expression for molar mass of solute in terms of boiling point elevation of solvent.
Show that elevation of boiling point is a colligative property.
Comment on the statement:
Equimolal solutions of different non-volatile and non-electrolytic substances dissolved in the same solvent exhibit the same elevation of boiling point.
What do you understand by freezing point of a liquid?
What happens to the freezing point of a liquid when a non-volatile solute is added to it?
Explain why the freezing point of a solvent gets lowered when a non-volatile solute is dissolved in it.
Explain how the measurement of depression of freezing point is useful in the determination of molecular mass of a solute.
Explain the phenomenon of osmosis.
Write down the differences between diffusion and osmosis.
Answer the following in one or two sentences.
What is osmotic pressure?
How is osmotic pressure dependent upon the number of moles of a solute?
Answer the following in one or two sentences.
What is osmotic pressure?
How is osmotic pressure related to the concentration of a solute in a solution?
Derive van’t Hoff general solution equation.
Why is the osmotic pressure considered to be a colligative property?
Answer the following in one or two sentences.
What is osmotic pressure?
How will you determine the molecular mass of a substance by osmotic pressure method?
Define isotonic solutions.
Define the following:
Hypertonic solution
Define the following term:
Hypotonic solution
What is the molal elevation constant, Kb?
How is the molal elevation constant, Kb is related to the molality of a solution?
What is the importance of osmosis for plants and animals?
What is an antifreeze?
If glycerine (C3H8O3) and methyl alcohol (CH3OH) are sold at the same price per kg, which would be cheaper for preparing an antifreeze for the radiator of a car?
What is meant by abnormal molecular mass? Illustrate it with an example.
Explain why molecular mass of ethanoic acid when determined by colligative properties of its solutions is abnormal. Suggest the value of van’t Hoff factor in this case.
What do you understand by the association of solute molecules in solution?
What do you understand by dissociation of solute molecules in solution?
How do association of solute molecules affect a colligative property?
How do dissociation of solute molecules affect a colligative property?
What is van’t Hoff factor?
What type of values can van’t Hoff factor have if in solution the solute molecules undergo association?
What type of values can van’t Hoff factor have if in solution the solute molecules undergo dissociation?
Why do we sometimes get abnormal molecular masses of substances?
Give the modified equations for the colligative properties for substances exhibiting abnormal molecular masses.
Define van’t Hoff factor.
Answer the following in one or two sentences.
How is van’t Hoff factor related to degree of ionization?
Define the degree of dissociation.
Derive the relationship between the degree of dissociation of an electrolyte and van’t Hoff factor.
The mole fraction of a solute in a solution is 0.1. At 298 K, molarity of this solution is the same as its molality. Density of this solution at 298 K is 2.0 g cm−3. Find the ratio of the molecular weights of the solute and solvent, `((MW_"solute")/(MW_"solvent"))`.
Nootan solutions for केमिस्ट्रि [इंग्रजी] इयत्ता १२ आयएससी 2 Solutions LONG ANSWER TYPE QUESTIONS [Pages 112 - 113]
Explain the term:
Solute
Explain the term of solvent.
Explain the term of solution.
Describe various types of solutions and mention the cause of solubility in each case.
Mention briefly the various units used to represent the composition of a solution. Also mention the effect of temperature on each of them.
Define vapour pressure of a liquid.
On which factors does the vapour pressure of a liquid depend?
Why does the boiling point of a liquid get elevated when a non-volatile solute is dissolved in it?
State and explain Raoult’s law. Show graphically the variation of total vapour pressure over a mixture of two volatile liquids with the composition of the mixture.
State Raoult’s law for solutions containing volatile solutes.
State Raoult’s law for the solutions of non-volatile solutes in volatile solvents.
Derive an expression for relative lowering of vapour pressure on the basis of Raoult’s law.
What do you understand by an ideal solution?
Which type of liquids do form an ideal solution?
Mention any three characteristics of an ideal solution.
Distinguish between ideal and non-ideal solutions.
What is a relative lowering of vapour pressure?
How is relative lowering of vapour pressure measured?
Derive an expression to show that the relative lowering of vapour pressure is a colligative property. On the basis of this expression, how would you determine the molecular mass of a solute?
Show that for a dilute solution `Delta p = (wM)/(WM') xx p^circ`
where, w = mass of solute, W = mass of solvent, M' = molecular mass of solute. M = molecular mass of solvent and p° = vapour pressure of pure solvent.
With the help of vapour pressure-temperature curves for solution and solvent, explain why boiling point of solvent is elevated when a nonvolatile solute is dissolved into it.
Derive the expression for molar mass of solute in terms of boiling point elevation of solvent.
Show that elevation of boiling point is a colligative property.
Derive the expression for molar mass of solute in terms of boiling point elevation of solvent.
Define the following term:
Molal elevation constant (Kb)
Mention units of molal elevation constant.
How is the molal elevation constant related to the latent heat of vaporisation of the solvent?
Define Freezing point.
Explain why the freezing point of a solvent gets lowered when a non-volatile solute is dissolved in it.
Derive an expression for the determination of molecular mass of a non-volatile solute on the basis of depression of freezing point.
Show that the depression of freezing point is a colligative property.
Define Cryoscopic constant.
Write the SI unit of the cryoscopic constant.
How is the latent heat of fusion per gram of the solvent related to molal depression constant?
Explain the following:
An aqueous solution of urea boils above 100°C but freezes below 0°C.
Explain the following:
NaCl is soluble in water but insoluble in benzene.
Explain the following:
In cold countries, ethylene glycol is added to water used in car radiators.
Why is common salt added to water used for boiling eggs?
What is osmosis?
Answer the following in one or two sentences.
What is osmotic pressure?
How is the osmotic pressure of a solution measured?
What is reverse osmosis?
Describe various laws of osmotic pressure.
Derive van’t Hoff general solution equation.
Why is the osmotic pressure considered to be a colligative property?
Derive an expression for the determination of molecular mass of a solute on the basis of osmotic pressure.
What is meant by abnormal molecular mass?
How are abnormal molecular masses related to the dissociation of a solute in solution?
How are abnormal molecular masses related to the association of a solute in solution?
Define van’t Hoff factor.
How are abnormal molecular masses related to the dissociation of a solute in solution?
How are abnormal molecular masses related to the association of a solute in solution?
Arrange the following solutions as directed:
0.1 M urea, 0.001 M sucrose, 1 M glucose (in the order of increasing osmotic pressure)
Explain the reason for your answers also.
Arrange the following solutions as directed:
0.1 M urea, 0.1 M NaCl, 0.1 M BaCl2, 0.1 M K4Fe(CN)6 (in the order of increasing osmotic pressure)
Explain the reason for your answers also.
Arrange the following solutions as directed:
1 M KCl, 1 M BaCl2, 1 M glucose (in the order of increasing boiling points)
Explain the reason for your answers also.
Arrange the following solutions as directed:
0.01 M KCl, 0.01 M sucrose, 0.01 M Na2SO4 (in the order of increasing freezing points)
Explain the reason for your answers also.
Explain why i = 1, when the molecular state of solute remains unchanged in solution.
Explain why i > 1, when the solute molecules dissociate in solution.
Explain why i < 1, when the solute molecules associate in solution.
Water boils at 100°C and ethyl alcohol at 78°C. The vapour pressure composition curve of this binary liquid system shows a maximum value. Sketch its boiling point composition curve.
Explain what happens when a solution containing 90% water and 10% alcohol is distilled. Can pure alcohol be obtained from this mixture by distillation?
Why is the osmotic pressure considered to be a colligative property?
Show that elevation of boiling point is a colligative property.
Define Osmotic pressure.
Arrange the following in increasing order of osmotic pressure and give reasons in support of your answer.
- 34.2 g per litre of sucrose (MW = 342)
- 90.0 g per litre of glucose (MW = 180)
- 5.85 g per litre of sodium chloride (MW = 58.5)
The molecular weights of sodium chloride and glucose are determined by the depression of freezing point method. Compared to their theoretical molecular weights, what will be their observed molecular weights when determined by the above method? Justify your answer.
An aqueous solution containing 1.70 g of cane sugar in 100 mL water begins to freeze at −0.093°C. The cryoscopic constant (molal depression constant) of water is 1.86 K kg mo1−1. Calculate the molecular weight of cane sugar.
A decinormal solution of sodium chloride exerts an osmotic pressure of 4.82 atmosphere at 27°C. Calculate the degree of dissociation of sodium chloride.
Nootan solutions for केमिस्ट्रि [इंग्रजी] इयत्ता १२ आयएससी 2 Solutions INTEGER TYPE QUESTIONS [Page 113]
2 molal solution of a weak acid HA has a freezing point of 3.885°C. The degree of dissociation of this acid is ______ × 10−3. (Round off to the nearest integer).
(Given: Molal depression constant of water = 1.85 K kg mol−1, Freezing point of pure water = 0°C)
A solute dimerises in water. The boiling point of a 2 molal solution of A is 100.52°C. The percentage association of A is ______. (Round off to the nearest integer).
[Use: Kb for water = 0.52 K kg mol−1, Boiling point of water = 100°C]
The boiling point of water in a 0.1 molal silver nitrate solution (solution A) is x°C. To this solution A, an equal volume of 0.1 molal aqueous barium chloride solution is added to make a new solution B. The difference in the boiling points of water in the two solutions A and B is y × 10−2 °C.
Assume: Densities of the solutions A and B are the same as that of water and the soluble salts dissociate completely.
Use: Molal elevation constant (kb = 0.5 K kg mol−1, Boiling point of pure water as 100°C)
- The value of ‘x’ is ______.
- The value of ‘y’ is ______.
A solution containing 2.5 × 10−3 kg of a solute dissolved in 75 × 10−3 kg of water, boils at 373.535 K. The molar mass of solute is ______ g mol−1. (Nearest integer)
(Given: kb (H2O) = 0.52 K molal−1, boiling point of water = 373.15 K)
The vapour pressures of two volatile liquids A and B at 25°C are 50 Torr and 100 Torr, respectively. If the liquid mixture contains 0.3 molar fraction of A, then the mole fraction of liquid B in the vapour phase is `x/17`. The value of x is ______.
An aqueous solution is prepared by dissolving 0.1 mol of an ionic salt in 1.8 kg of water at 35°C. The salt remains 90% dissociated in the solution. The vapour pressure of the solution is 59.724 mm of Hg. Vapour pressure of water at 35°C is 60 mm Hg. The number of ions present per formula unit of the ionic salt is ______.
Nootan solutions for केमिस्ट्रि [इंग्रजी] इयत्ता १२ आयएससी 2 Solutions OBJECTIVE (MULTIPLE CHOICE) TYPE QUESTIONS [Pages 114 - 118]
Choose the correct option in the following questions.
An electrolyte dissolves in water when ______.
hydration energy is less than lattice energy.
hydration energy is equal to lattice energy.
hydration energy is greater than lattice energy.
dissolution is exothermic.
An azeotropic mixture of two liquids boils at a lower temperature than either of them when ______.
it is saturated.
it does not deviate from Raoult’s law.
it shows negative deviation from Raoult’s law.
it shows positive deviation from Raoult’s law.
Which of the following solution pairs can be separated by fractional distillation?
Water-HNO3
Water-HCl
Benzene-toluene
C2H5OH-water
If 18 g of glucose are present in 1000 g of solvent, the solution is said to be ______.
1 molar
0.1 molar
0.5 molal
0.1 molal
Mole fraction of C3H5(OH)3 in a solution of 36 g of water and 46 g of glycerine is ______.
0.46
0.36
0.20
0.40
1000 g aqueous solution of CaCO3 contains 10 g of calcium carbonate. Concentration of the solution is ______.
10 ppm
100 ppm
1000 ppm
10000 ppm
Which of the following is not a colligative property?
ΔTf
ΔTb
Kb
Osmotic pressure
The molal freezing point constant for water is 1.86 K kg mol−1. Therefore, the freezing point of 0.1 molal NaCl solution in water is expected to be ______.
−1.86°C
−0.186°C
−0.372°C
+0.372°C
The molal elevation of an unknown solution is equal to the molal elevation of 0.1 M solution of urea. The concentration of unknown solution is ______.
1 M
0.01 M
0.1 M
none of these
A solution of 1.25 g of a non-electrolyte in 20 g of water freezes at 271.94 K. If Kf is 1.86 K kg mol−1, then the molecular mass of solute will be ______.
207.8
179.79
209.6
109.5
Which of the following 0.1 M aqueous solutions will have the lowest freezing point?
K2SO4
NaCl
Urea
Glucose
Which one of the following salts would have the same value of the van't Hoff factor as that of K3Fe(CN)6?
Al2(SO4)3
NaCl
Na2SO4
Al(NO3)3
When mercuric chloride is added to the aqueous solution of potassium iodide, then ______.
the boiling point does not change
the freezing point is raised
the freezing point is lowered
the freezing point does not change
The molecular weight of benzoic acid in benzene as determined by depression of freezing point method corresponds to ______.
ionisation of benzoic acid
dimerisation of benzoic acid
trimerisation of benzoic acid
salvation of benzoic acid.
The molal freezing point constant for water is 1.86. If 342 g of cane sugar is dissolved in 1000 g of water, the solution will freeze at ______.
1.86°C
−1.86°C
−3.92°C
3.92°C
When acetone and chloroform are mixed, hydrogen bonding takes place between them. Such a liquid pair ______.
causes positive deviation from Raoult’s law
causes negative deviation from Raoult’s law
causes no deviation from Raoult’s law
forms an ideal solution
Dry air was passed successively through a solution of 5 g of a solute in 80 g of water and then through pure water. The loss in mass of solution was 2.5 g and that of pure solvent 0.04 g. What is the molecular mass of solute?
71.43
7.143
714.3
80
An example of colligative property is ______.
freezing point
boiling point
vapour pressure
osmotic pressure
What is the molality of the solution of a certain solute in a solvent, if there is a freezing point depression of 0.184°C and if the freezing point constant is 18.4°C m−1?
0.01
1.0
0.001
100
Which inorganic precipitate acts as semipermeable membrane?
Calcium sulphate
Barium oxalate
Nickel phosphate
Copper ferrocyanide
The vapour pressure of a solution of 5 g of a non-electrolyte in 100 g of water at a particular temperature is 2985 Nm−2. The vapour pressure of pure water at that temperature is 3000 Nm−2. The molecular weight of solute is ______.
180
90
270
200
van’t Hoff factor for 0.1 M ideal solution is ______.
0.1
1
0.01
none of these
A solution of glycol containing 1.82 g/litre has an osmotic pressure of 51.8 cm of mercury at 10°C. What is the molecular weight of glycol?
62.04
70.16
80.54
100.21
Azeotropic mixture of HCl and H2O has ______.
48% HCl
22.2% HCl
36% HCl
20.2% HCl
Which of the following 0.10 m aqueous solutions will have the lowest freezing point?
Al2(SO4)3
C5H10O5
KI
C12H22O11
A solution which has lower osmotic pressure as compared to other solution is known as ______.
hypotonic
hypertonic
isotonic
isosmotic
A mixture which boils off like a single pure component is called ______.
eutectic
azeotropic
ideal solution
non-ideal solution
The osmotic pressure of equimolal solutions of glucose, sodium chloride and barium chloride will be in the order ______.
BaCl2 > NaCl > Glucose
BaCl2 > Glucose > NaCl
Glucose > BaCl2 > NaCl
NaCl > BaCl2 > Glucose
A 500 g toothpaste sample has 0.2 g fluoride concentration. What is the concentration of fluorine in terms of ppm level?
250
200
400
1000
Which is not affected by temperature?
Normality
Formality
Molarity
Molality
The molecular weight of sodium chloride determined by measuring the osmotic pressure of its aqueous solution is ______.
double the theoretical value
same as the theoretical value
half the theoretical value
three times the theoretical value
Consider the following figure and choose the correct option:
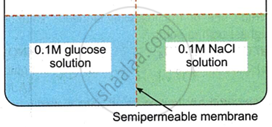
There will be no movement of any solution across the membrane.
The osmotic pressure of glucose solution is equal to that of NaCl solution.
The osmotic pressure of NaCl solution is lower than that of glucose solution.
The osmotic pressure of NaCl solution is higher than that of glucose solution.
PtCl4 . 6H2O can exist as a hydrated complex. One molal aqueous solution of the salt recorded a depression in freezing point of 3.72°C. If the salt is assumed to be 100% ionised in solution and Kf for H2O is 1.86 K kg mo1−1, the molecular formula of the complex is likely to be ______.
[Pt(H2O)6)]Cl4
[Pt(H2O)4Cl2]Cl2 . 2H2O
[Pt(H2O)3Cl3]Cl . 3H2O
[Pt(H2O)2Cl4]Cl . 4H2O
Freezing point of water is 0°C. At which temperature will one molal solution of NaCl freeze, if NaCl is taken to be completely dissociated (molal depression constant of water = 18.6)?
−3.72°C
−1.86°C
0°C
3.72°C
For an aqueous solution, freezing point is −0.186°C. Elevation of the boiling point of the same solution (Kf = 1.86° mol−1 kg and Kb = 0.512° mol−1 kg) is ______.
0.186°
0.0512°
1.86°
5.12°
In a mixture of A and B, components show negative deviation when ______.
A-B interaction is stronger than A-A and B-B interaction.
A-B interaction is weaker than A-A and B-B interaction.
`Delta V_"mix" > 0`, `Delta S_"mix" > 0`
`Delta V_"mix" = 0`, `Delta S_"mix" > 0`
In a 0.2 molal aqueous solution of a weak acid HX, the degree of ionisation is 0.3. Taking Kf for water as 1.85, the freezing point of the solution will be nearest to ______.
−0.480°C
−0.360°C
−0.260°C
+0.480°C
25 ml of a solution of barium hydroxide on titration with a 0.1 molar solution of hydrochloric acid gave a titre value of 35 ml. The molarity of barium hydroxide solution was ______.
0.07
0.14
0.28
0.35
If liquids A and B form an ideal solution, the ______.
enthalpy of mixing is zero.
entropy of mixing is zero.
free energy of mixing is zero.
free energy as well as the entropy of mixing are each zero.
Which one of the following aqueous solutions will exhibit highest boiling point?
0.01 M Na2SO4
0.01 M KNO3
0.015 M urea
0.015 M glucose
Which of the following liquid pairs shows a positive deviation from Raoult’s law?
Water + hydrochloric acid
Benzene + methanol
Water + nitric acid
Acetone + chloroform
Which one of the following statements is false?
Raoult’s law states that the vapour pressure of a component over a solution is proportional to its mole fraction.
The osmotic pressure (π) of a solution is given by the equation π = MRT, where M is the molarity of the solution.
The correct order of osmotic pressure for 0.01 M aqueous solution of each compound is BaCl2 > KCl > CH3COOH > sucrose.
Two sucrose solutions of same molality prepared in different solvents will have the same freezing point depression.
If α is the degree of dissociation of Na2SO4, the van’t Hoff factor (i) used for calculating the molecular mass is ______.
1 − 2α
1 + 2α
1 − α
1 + α
Equimolal solutions in the same solvent have ______.
different boiling and different freezing points.
same boiling and same freezing points.
same freezing point but different boiling points.
same boiling point but different freezing points.
Among the following mixtures, dipole-dipole as the major interaction, is present in
benzene and ethanol
acetonitrile and acetone
KCl and water
benzene and carbon tetrachloride
18 g of glucose (C6H12O6) is added to 178.2 g of water. The vapour pressure of water for this aqueous solution at l00°C is ______.
759.00 torr
7.60 torr
76.00 torr
752.40 torr
A 5.25% solution of a substance is isotonic with a 1.5% solution of urea (molar mass = 60 g mol−1) in the same solvent. If the densities of both the solutions are assumed to be equal to 1.0 g cm−3, molar mass of the substance will be ______.
90.0 g mol−1
115.0 g mol−1
105.0 g mol−1
210.0 g mol−1
A mixture of ethyl alcohol and propyl alcohol has a vapour pressure of 290 mm at 300 K. The vapour pressure of propyl alcohol is 200 mm. If the mole fraction of ethyl alcohol is 0.6, its vapour pressure (in mm) at the same temperature will be ______.
350
300
700
360
At 80°C, the vapour pressure of pure liquid ‘A’ is 520 mm Hg and that of pure liquid ‘B’ is 1000 mm Hg. If a mixture solution of ‘A’ and ‘B’ boils at 80°C and 1 atm pressure, the amount of ‘A’ in the mixture is ______. (1 atm = 760 mm Hg)
52 mol per cent
34 mol per cent
48 mol per cent
50 mol per cent
The vapour pressure of water at 20°C is 17.5 mm Hg. If 18 g of glucose (C6H12O6) is added to 178.2 g of water at 20°C, the vapour pressure of the resulting solution will be ______.
17.675 mm Hg
15.750 mm Hg
16.500 mm Hg
17.325 mm Hg
An aqueous solution is 1.00 molal in Kl. Which change will cause the vapour pressure of the solution to increase?
Addition of NaCl
Addition of Na2SO4
Addition of 1.00 molal KI
Addition of water
A solution of sucrose (molar mass = 342 g mol−1) has been prepared by dissolving 68.5 g of sucrose in 1000 g of water. The freezing point of the solution obtained will be ______. (kf for water = 1.86 K kg mol−1)
−0.372°C
−0.520°C
+0.372°C
−0.570°C
The freezing point depression constant for water is −l.86°C m−1. If 5.00 g Na2SO4 is dissolved in 45.0 g H2O, the freezing point is changed by −3.82°C. Calculate the van't Hoff factor for Na2SO4.
2.63
3.11
0.381
2.05
pA and pB are the vapour pressure of pure liquid components, A and B, respectively of an ideal binary solution. If χA represents the mole fraction of component A, the total pressure of the solution will be ______.
pA + χA(pB − pA)
pA + χA(pA − pB)
pB + χA(pB − pA)
pB + χA(pA − pB)
Which of the following 0.10 m aqueous solutions will have maximum ΔTf value?
C12H22O11
KI
Al2(SO4)3
NH2-CO-NH2
C6H12O6
KCl
K2SO4
Consider the separate solution of 0.500 M C2H5OH (aq.), 0.100 M Mg3(PO4)2 (aq.), 0.250 M KBr (aq.) and 0.125 M Na3PO4 (aq.) at 25°C. Which statement is true about these solutions, assuming all salts to be strong electrolytes?
They all have same osmotic pressure.
0.100 M Mg3(PO4)2 (aq.) has the highest osmotic pressure.
0.125 M Na3PO4 (aq.) has the highest osmotic pressure.
0.500 M C2H5OH (aq.) has the highest osmotic pressure.
The vapour pressure of acetone at 20°C is 185 torr. When 1.2 g of a non-volatile substance was dissolved in 100 g of acetone at 20°C, its vapour pressure was 183 torr. The molar mass (g mol−1) of the substance is ______.
32
64
128
488
18 g glucose (C6H12O6) is added to 178.2 g water. The vapour pressure of water (in torr) for this aqueous solution is ______.
7.6
76.0
752.4
759.0
Consider the following liquid-vapour equilibrium:
\[\ce{Liquid <=> Vapour}\]
Which of the following relation is correct?
`(dlnG)/(dT^2) = (Delta H_v)/(RT^2)`
`(dlnP)/(dT) = (-Delta H_v)/(RT)`
`(dlnP)/(dT^2) = (-Delta H_v)/(T^2)`
`(dlnP)/(dT) = (Delta H_v)/(RT^2)`
Which of the following statements about the composition of the vapour over an ideal 1 : 1 molar mixture of benzene and toluene is correct? Assume that the temperature is constant at 25°C. (Given, vapour pressure data at 25°C, benzene = 12.8 kPa, toluene = 3.85 kPa)
The vapour will contain a higher percentage of benzene.
The vapour will contain a higher percentage of toluene.
The vapour will contain equal amounts of benzene and toluene.
Not enough information is given to make a prediction.
At 100°C the vapour pressure of a solution containing 6.5 g a solute in 100 g water is 732 mm. If Kb = 0.52, the boiling point of this solution will be ______.
102°C
100°C
101°C
100.52°C
If molality of dilute solution is doubled, the value of molal depression constant (Kf) will be ______.
doubled
halved
tripled
unchanged
Which of the following is dependent on temperature?
Molality
Molarity
Mole fraction
Weight percentage
The freezing point of benzene decreases by 0.45°C when 0.2 g of acetic acid is added to 20 g of benzene. If acetic acid associates to form a dimer in benzene, percentage association of acetic acid in benzene will be ______.
(Kf for benzene = 5.12 K kg mol−1)
74.6%
94.6%
64.6%
80.4%
For 1 molal aqueous solution of the following compounds, which one will show the highest freezing point?
[CO(H2O)6]Cl3
[CO(H2O)5Cl]Cl2 . H2O
[CO(H2O)4Cl2]Cl . 2H2O
[CO(H2O)3Cl3] . 3H2O
The recommended concentration of fluoride ion in drinking water is up to 1 ppm as fluoride ion is required to make teeth enamel harder by converting [3Ca3(PO4)2 . Ca(OH2)] to ______.
[CaF2]
[3(CaF2) . Ca(OH)2]
[3Ca3(PO4)2 . CaF2]
[3{Ca(OH)2} . CaF2]
At room temperature, a dilute solution of urea is prepared by dissolving 0.60 g of urea in 360 g of water. If the vapour pressure of pure water at this temperature is 35 mm Hg, lowering of vapour pressure will be ______. (molar mass of urea = 60 g mol−1)
0.031 mm Hg
0.017 mm Hg
0.028 mm Hg
0.027 mm Hg
1 g of a non-volatile non-electrolyte solute is dissolved in 100 g of two different solvents A and B, whose ebullioscopic constants are in the ratio of 1 : 5. The ratio of the elevation in their boiling points, `(Delta T_b (A))/(Delta T_b (B))`, is ______.
5 : 1
1 : 0.2
10 : 1
1 : 5
The mole fraction of a solvent in aqueous solution of a solute is 0.8. The molality (in mol kg−1) of the aqueous solution is ______.
13.88 × 10−1
13.88 × 10−3
13.88
13.88 × 10−2
The density of 2 M aqueous solution of NaOH is 1.28 g/cm3. The molality of the solution is ______. [Given that molecular mass of NaOH = 40 g mol−1]
1.32 m
1.20 m
1.56 m
1.67 m
Which of the following statements is correct regarding a solution of two components A and B exhibiting positive deviation from ideal behaviour?
Intermolecular attractive forces between A-A and B-B are equal to those between A-B.
Intermolecular attractive forces between A-A and B-B are stronger than those between A-B.
Δmix H = 0 at constant T and P.
Δmix V = 0 at constant T and P.
In water saturated air, the mole fraction of water vapour is 0.02. If the total pressure of the saturated air is 1.2 atm, the partial pressure of dry air is ______.
0.98 atm
1.18 atm
1.76 atm
1.176 atm
The mixture that forms maximum boiling azeotrope is ______.
ethanol + water
acetone + carbon disulphide
heptane + octane
water + nitric acid
For an ideal solution, the correct option is:
Δmix S = 0 at constant T and P.
Δmix V ≠ 0 at constant T and P.
Δmix H = 0 at constant T and P.
Δmix G = 0 at constant T and P.
The mixture which shows positive deviation from Raoult’s law is ______.
Ethanol + Acetone
Benzene + Toluene
Acetone + Chloroform
Chloroethane + Bromoethane
The freezing point depression constant (Kf) of benzene is 5.12 K kg mol−1. The freezing point depression for the solution of molality 0.078 m containing a non-electrolyte solute in benzene is ______. (rounded off upto two decimal places)
0.20 K
0.80 K
0.40 K
0.60 K
An open beaker of water in equilibrium with water vapour is in sealed container. When a few grams of glucose are added to the beaker of water, the rate at which water molecules ______.
Leaves the vapour decreases
Leaves the solution decreases
Leaves the vapour increases
Leaves the solution increases
The following solutions were prepared by dissolving 10 g of glucose (C6H12O6) in 250 ml of water (P1), 10 g of urea (CH4N2O) in 250 ml of water (P2) and 10 g of sucrose (C12H22O11) in 250 ml of water (P3). The right option for the decreasing order of osmotic pressure of these solutions is
P3 > P1 > P2
P2 > P1 > P3
P1 > P2 > P3
P2 > P3 > P1
The correct option for the value of vapour pressure of a solution at 45°C with benzene to octane in a molar ratio of 3 : 2 is
[At 45°C vapour pressure of benzene is 280 mm Hg and that of octane is 420 mm Hg. Assume Ideal gas]
350 mm of Hg
160 mm of Hg
168 mm of Hg
336 mm of Hg
In one molal solution that contains 0.5 mole of a solute, there is ______.
500 g of solvent
100 ml of solvent
1000 g of solvent
500 ml of solvent
Solute A associates in water. When 0.7 g of solute A is dissolved in 42.0 g of water, it depresses the freezing point by 0.2°C. The percentage association of solute A in water is ______.
(Given: Molar mass of A = 93 g mol−1, molal depression constant of water is 1.86 K kg rnol−1)
50%
60%
70%
80%
Nootan solutions for केमिस्ट्रि [इंग्रजी] इयत्ता १२ आयएससी 2 Solutions FILL IN THE BLANKS TYPE QUESTIONS [Pages 118 - 119]
Two liquids are partially miscible when the intermolecular attraction of one liquid is ______ from that of the other.
The ppm of a solute is defined as the mass of solute (in grams) present in ______ grams of the solution.
Since the molarity involves ______ which is ______ on temperature, the molarity of a solution ______ with temperature.
The vapour pressure of a liquid ______ with increase in temperature.
For a solution of volatile liquids, the partial vapour pressure of a component is equal to the product of its ______ in ______ and its ______ in the solution.
Two liquids A and B can form an ideal solution only when the A-B molecular interaction forces are ______ to the A-A or B-B interaction forces.
For a non-ideal solution showing positive deviation from Raoult’s law, `Delta H_"mixing"` is ______.
For a non-ideal solution showing negative deviation from Raoult’s law, A-B molecular interaction forces are ______ than A-A and B-B molecular interaction forces.
The solution of two completely miscible liquids which boils at a constant temperature like a pure liquid and distils over ______ any change in composition, is called an ______ mixture.
The properties of solutions which depend upon the number of solute particles present in a definite amount of solvent and not on the chemical nature of solute are called ______ properties.
The addition of a solute to a pure solvent ______ its vapour pressure but ______ its boiling point.
The molal elevation constant is the ______ of boiling point produced on dissolving ______ mole of a solute in ______ g of the solvent.
ΔTf = Kf × ______
______ solutions of different non-volatile and non-electrolytic substances dissolved in the same solvent exhibit the ______ depression of freezing point.
A membrane which allows the passage of only ______ molecules and not of ______ is called a ______.
In osmosis, the net flow of solvent molecules is from ______ concentration to ______ concentration.
According to Charles’ van’t Hoff law, π ∝ ______ at constant ______.
Osmotic pressure of a solution depends upon the number of moles of ______ dissolved in a definite volume of solution and not upon the ______ of ______.
When RBCs are placed in ______% solution of pure sodium chloride, the corpuscles neither ______ nor ______ because ______ water is ______ to red blood corpuscles.
Association of solute particles always leads to a ______ value of observed molecular mass of the solute.
Dissociation of solute particles leads to an ______ in the magnitude of a colligative property and to a ______ in the value of the observed molecular mass.
The ratio of the observed value of a colligative property to the normal value of the same property is termed as ______ and is represented by ______.
Normal molecular mass = ______ × observed molecular mass.
When solute undergoes dissociation in solution, the value of i is ______ than 1.
The degree of association is defined as the ______ of total number of ______ of solute which undergoes ______ in solution.
Nootan solutions for केमिस्ट्रि [इंग्रजी] इयत्ता १२ आयएससी 2 Solutions ASSERTION-REASON TYPE QUESTIONS [Page 119]
The questions given below consist of an Assertion and a Reason. You have to choose the correct answer (a), (b), (c) or (d) according to the following clue:
Assertion: The mixing of two completely miscible liquids A and B showing positive deviation from Raoult’s law is followed by an absorption of heat.
Reason: The A-A molecular interaction forces are stronger than the A-A or B-B molecular interaction forces.
If both Assertion and Reason are CORRECT and Reason is the CORRECT explanation of the Assertion.
If both Assertion and Reason are CORRECT but Reason is not the CORRECT explanation of the Assertion.
If Assertion is CORRECT but Reason is INCORRECT.
If Assertion is INCORRECT but Reason is CORRECT.
Assertion: Equimolal solutions of different non-volatile and non-electrolytic substances dissolved in the same solvent exhibit the same elevation of boiling point.
Reason: All solvents boil when their vapour pressures become equal to the atmospheric pressure.
If both Assertion and Reason are CORRECT and Reason is the CORRECT explanation of the Assertion.
If both Assertion and Reason are CORRECT but Reason is not the CORRECT explanation of the Assertion.
If Assertion is CORRECT but Reason is INCORRECT.
If Assertion is INCORRECT but Reason is CORRECT.
Assertion: Depression of freezing point is a colligative property.
Reason: The depression of freezing point for a solution is directly proportional to the molality of the solution.
If both Assertion and Reason are CORRECT and Reason is the CORRECT explanation of the Assertion.
If both Assertion and Reason are CORRECT but Reason is not the CORRECT explanation of the Assertion.
If Assertion is CORRECT but Reason is INCORRECT.
If Assertion is INCORRECT but Reason is CORRECT.
Assertion: Osmotic pressure of a dilute solution is inversely proportional to its concentration provided temperature remains constant.
Reason: Osmotic pressure is a colligative property and depends upon the number of moles of solute dissolved in a definite volume of the solution.
If both Assertion and Reason are CORRECT and Reason is the CORRECT explanation of the Assertion.
If both Assertion and Reason are CORRECT but Reason is not the CORRECT explanation of the Assertion.
If Assertion is CORRECT but Reason is INCORRECT.
If Assertion is INCORRECT but Reason is CORRECT.
Assertion: 1 M solution of NaCl has a higher osmotic pressure than 1 M solution of glucose at the same temperature.
Reason: The number of particles present in 1 M NaCl solution are more than those present in 1 M glucose solution and osmotic pressure depends upon number of particles present in the solution.
If both Assertion and Reason are CORRECT and Reason is the CORRECT explanation of the Assertion.
If both Assertion and Reason are CORRECT but Reason is not the CORRECT explanation of the Assertion.
If Assertion is CORRECT but Reason is INCORRECT.
If Assertion is INCORRECT but Reason is CORRECT.
Assertion: At 10°C, the density of 5 M solution of KCl [atomic masses of K and Cl are 39 and 35.5 g mol−1]. The solution is cooled to −21°C. The molality of solution will remain unchanged.
Reason: The molality of a solution does not change with temperature as mass remains unaffected with temperature.
If both Assertion and Reason are CORRECT and Reason is the CORRECT explanation of the Assertion.
If both Assertion and Reason are CORRECT but Reason is not the CORRECT explanation of the Assertion.
If Assertion is CORRECT but Reason is INCORRECT.
If Assertion is INCORRECT but Reason is CORRECT.
Nootan solutions for केमिस्ट्रि [इंग्रजी] इयत्ता १२ आयएससी 2 Solutions NUMERICAL PROBLEMS [Page 120]
An aqueous solution of a non-volatile and non-electrolytic substance boils at 100.5°C. Calculate the osmotic pressure of this solution at 27°C. Kb for water per 1000 g = 0.50.
At 27°C, 36 g of glucose per litre has an osmotic pressure of 4.92 atmosphere. If the osmotic pressure of another solution of glucose is 1.5 atmosphere at the same temperature, what would be its concentration?
A very small amount of a non-volatile solute (that does not dissociate) is dissolved in 56.8 cm3 of benzene (density = 0.889 g cm−3). At room temperature, vapour pressure of this solution is 98.88 mm Hg while that of benzene is 100 mm Hg. Find the molality of this solution. If the freezing point of this solution is 0.73 degree lower than that of benzene, what is the value of molal freezing point depression constant of benzene?
A solution prepared by dissolving 0.300 g of an unknown compound in 30 g of CCl4 has a boiling point that is 0.392°C higher than that of pure CCl4. What is the molecular weight of solute? (Molal boiling point elevation constant of CCl4 is 5.02°C/m)
0.85% aqueous solution of NaNO3 is apparently 90% dissociated at 27°C. Calculate its osmotic pressure. (R = 0.082 L atm K−1 mol−1)
A 1.2% solution of NaCl is isotonic with 7.2% solution of glucose. Calculate the van’t Hoff factor of NaCl.
2 g of benzoic acid dissolved in 25 g of C6H6 show a depression in freezing point equal to 1.62 K. Molal depression constant of C6H6 is 4.9 K mo1−1 kg. What is the percentage association of acid if it forms double molecules in solution?
x g of a non-electrolytic compound (molar mass = 200) are dissolved in 1.0 L of 0.05 M NaCl aqueous solution. The osmotic pressure of this solution is found to be 4.92 atm at 27°C. Calculate the value of x. Assume complete dissociation of NaCl and ideal behaviour of the solution (R = 0.082 L atm K−1 mol−1).
The degree of dissociation of Ca(NO3)2 in a dilute aqueous solution containing 7.0 g of the salt per 100 g of water at 100°C is 70 per cent. If the vapour pressure of water at 100°C is 760 mm, calculate the vapour pressure of the solution.
Calculate the boiling point of a solution containing 0.61 g of benzoic acid in 50 g of carbon-disulphide assuming 84% dimerisation of the acid. The boiling point and Kb of CS2 are 46.2°C and 2.3 K kg mol−1 respectively.
How many grams of KCl should be added to 1.00 kilogram of water to lower its freezing point to −8.0°C? Kf(H2O) = l .88°C kg mo1−1 (At. masses: K = 39, Cl = 35.5).
Calculate the freezing point of an aqueous solution of a non-electrolyte having osmotic pressure of 2.0 atm at 300 K.
(Kf = 1.86 K kg mol−1, R = 0.0821 L atm K−1 mol−1)
A solution of a non-volatile solute in water freezes at −0.30°C. The vapour pressure of pure water at 298 K is 23.51 mm Hg and Kf for water is 1.86 degree/molal. Calculate the vapour pressure of this solution at 298 K.
Nootan solutions for केमिस्ट्रि [इंग्रजी] इयत्ता १२ आयएससी 2 Solutions 'NCERT TEXT-BOOK' Exercises [Pages 123 - 129]
Define the term solution.
Describe various types of solutions and mention the cause of solubility in each case.
Suppose a solid solution is formed between two substances, one whose particles are very large and the other whose particles are very small. What kind of solid solution is this likely to be?
Define Mole fraction.
Define molality.
Define Molarity (M).
Define Mass percentage.
Concentrated nitric acid used in laboratory work is 68% nitric acid by mass in aqueous solution. What should be the molarity of such a sample of the acid if the density of the solution is 1.504 g mL−1?
A solution of glucose in water is labelled as 10% w/w, what would be the molality and mole fraction of each component in the solution? If the density of solution is 1.2 g mL−1, then what shall be the molarity of the solution?
How many mL of 0.1 M HCl are required to react completely with 1 g mixture of Na2CO3 and NaHCO3 containing equimolar amounts of both?
A solution is obtained by mixing 300 g of 25% solution and 400 g of 40% solution by mass. Calculate the mass percentage of the resulting solution.
An antifreeze solution is prepared from 222.6 g of ethylene glycol (C2H6O2) and 200 g of water. Calculate the molality of the solution. If the density of the solution is 1.072 g mL−1, then what shall be the molarity of the solution?
A sample of drinking water was found to be severely contaminated with chloroform, CHCl3, supposed to be carcinogenic in nature. The level of contamination was 15 ppm (by mass).
- Express this in percent by mass.
- Determine the molality of chloroform in the water sample.
What role does the molecular interaction play in a solution of alcohol and water?
Why do gases always tend to be less soluble in liquids as the temperature is raised?
State Henry’s law.
Write the two applications of Henry’s law.
The partial pressure of ethane over a solution containing 6.56 × 10−3 g of ethane is 1 bar. If the solution contains 5.00 × 10−2 g of ethane, then what shall be the partial pressure of the gas?
What is meant by positive deviations from Raoult’s law?
How is the sign of ΔmixH related to positive deviations from Raoult’s law?
What is meant by negative deviations from Raoult’s law?
How is the sign of ΔmixH related to negative deviations from Raoult’s law?
An aqueous solution of 2% non-volatile solute exerts a pressure of 1.004 bar at the normal boiling point of the solvent. What is the molar mass of the solute?
Heptane and octane form an ideal solution. At 373 K, the vapour pressures of the two liquid components are 105.2 kPa and 46.8 kPa respectively. What will be the vapour pressure of a mixture of 26.0 g of heptane and 35 g of octane?
The vapour pressure of water is 12.3 kPa at 300 K. Calculate vapour pressure of 1 molal solution of a non-volatile solute in it.
Calculate the mass of a non-volatile solute (molar mass 40 g mol−1) which should be dissolved in 114 g octane to reduce its vapour pressure to 80%.
A solution containing 30 g of non-volatile solute exactly in 90 g of water has a vapour pressure of 2.8 kPa at 298 K. Further, 18 g of water is then added to the solution and the new vapour pressure becomes 2.9 kPa at 298 K. Calculate:
- molar mass of the solute.
- vapour pressure of water at 298 K.
A 5% solution (by mass) of cane sugar in water has freezing point of 271 K. Calculate the freezing point of 5% glucose in water if freezing point of pure water is 273.15 K.
Two elements A and B form compounds having formula AB2 and AB4. When dissolved in 20 g of benzene (C6H6), 1 g of AB2 lowers the freezing point by 2.3 K whereas 1.0 g of AB4 lowers it by 1.3 K. The molar depression constant for benzene is 5.1 K kg mol−1. Calculate the atomic masses of A and B.
At 300 K, 36 g of glucose present in a litre of its solution has an osmotic pressure of 4.98 bar. If the osmotic pressure of the solution is 1.52 bars at the same temperature, what would be its concentration?
Suggest the most important type of intermolecular attractive interaction in the following pair.
n-hexane and n-octane
Suggest the most important type of intermolecular attractive interaction in the following pair:
I2 and CCl4
Suggest the most important type of intermolecular attractive interaction in the following pair.
NaClO4 and water
Suggest the most important type of intermolecular attractive interaction in the following pair.
Methanol and acetone
Suggest the most important type of intermolecular attractive interaction in the following pair.
acetonitrile (CH3CN) and acetone (C3H6O)
Based on solute-solvent interactions, arrange the following in order of increasing solubility in n-octane and explain.
Cyclohexane, KCl, CH3OH, CH3CN
Amongst the following compounds, identify which are insoluble, partially soluble and highly soluble in water?
- Phenol
- Toluene
- Formic acid
- Ethylene glycol
- Chloroform
- Pentanol
If the density of some lake water is 1.25 g mL−1 and contains 92 g of Na+ ions per kg of water, calculate the molality of Na+ ions in the lake.
If the solubility product of CuS is 6 × 10−16, calculate the maximum molarity of CuS in aqueous solution.
Calculate the mass percentage of aspirin (C9H8O4) in acetonitrile (CH3CN) when 6.5 g of C9H8O4 is dissolved in 450 g of CH3CN.
Nalorphene (C19H21NO3), similar to morphine, is used to combat withdrawal symptoms in narcotic users. Dose of nalorphene generally given is 1.5 mg. Calculate the mass of 1.5 × 10−3 m aqueous solution required for the above dose.
Calculate the amount of benzoic acid (C6H5COOH) required for preparing 250 mL of 0.15 M solution in methanol.
The depression in freezing point of water observed for the same amount of acetic acid, trichloroacetic acid and trifluoroacetic acid increases in the order given above. Explain briefly.
Calculate the depression in the freezing point of water when 10 g of CH3CH2CHClCOOH is added to 250 g of water. Ka = 1.4 × 10−3, Kf = 1.86 K kg mol−1.
19.5 g of CH2FCOOH is dissolved in 500 g of water. The depression in the freezing point of water observed is 1.0°C. Calculate the van’t Hoff factor and dissociation constant of fluoroacetic acid.
Vapour pressure of water at 293 K is 17.535 mm Hg. Calculate the vapour pressure of water at 293 K when 25 g of glucose is dissolved in 450 g of water.
Henry’s law constant for the molality of methane in benzene at 298 K is 4.27 × 105 mm Hg. Calculate the solubility of methane in benzene at 298 K under 760 mm Hg.
100 g of liquid A (molar mass 140 g mol−1) was dissolved in 1000 g of liquid B (molar mass 180 g mol−1). The vapour pressure of pure liquid B was found to be 500 torr. Calculate the vapour pressure of pure liquid A and its vapour pressure in the solution if the total vapour pressure of the solution is 475 Torr.
Vapour pressure of pure acetone and chloroform at 328 K are 741.8 mm Hg and 632.8 mm Hg respectively. Assuming that they form ideal solution over the entire range of composition, plot `p_"total"`, `p_"chloroform"` and `p_"acetone"` as a function of `chi_"acetone"`. The experimental data observed for different compositions of mixtures is:
| `bb(100 xx chi_"acetone")` | `bb(P_"acetone"//"mm Hg")` | `bb(p_"chloroform"//"mm Hg")` |
| 0 | 0 | 632.8 |
| 11.8 | 54.9 | 548.1 |
| 23.4 | 110.1 | 469.4 |
| 36 | 202.4 | 359.7 |
| 50.8 | 322.7 | 257.7 |
| 58.2 | 405.9 | 193.6 |
| 64.5 | 454.1 | 161.2 |
| 72.1 | 521.1 | 120.7 |
Plot this data also on the same graph paper. Indicate whether it has a positive or negative deviation from the ideal solution.
Benzene and toluene form ideal solution over the entire range of composition. The vapour pressure of pure benzene and toluene at 300 K are 50.71 mm Hg and 32.06 mm Hg respectively. Calculate the mole fraction of benzene in vapour phase if 80 g of benzene is mixed with 100 g of toluene.
The air is a mixture of a number of gases. The major components are oxygen and nitrogen with approximate proportion of 20% is to 79% by volume at 298 K. The water is in equilibrium with air at a pressure of 10 atm. At 298 K if the Henry’s law constants for oxygen and nitrogen are 3.30 × 107 mm and 6.51 × 107 mm respectively, calculate the composition of these gases in water.
Determine the amount of CaCl2 (i = 2.47) dissolved in 2.5 litre of water such that its osmotic pressure is 0.75 atm at 27°C.
Determine the osmotic pressure of a solution prepared by dissolving 25 mg of K2SO4 in 2 liter of water at 25°C, assuming that it is completely dissociated.
Nootan solutions for केमिस्ट्रि [इंग्रजी] इयत्ता १२ आयएससी 2 Solutions QUESTIONS FROM ISC EXAMINATION PAPERS [Pages 130 - 132]
The freezing point of nitrobenzene is 278.8 K. A 0.25 molal solution of a substance (mol. wt. = 120) in nitrobenzene has a freezing point of 276.8 K. Calculate the molal depression constant of nitrobenzene.
Calculate the osmotic pressure of a solution containing 3.42 g of sucrose in 1 litre of water at 400 K.
What are colligative properties?
The osmotic pressure of blood is 7.65 atm at 27°C. How much glucose should be used per litre to prepare an intravenous injection that has the same osmotic pressure as blood?
Molarity of a solution is independent of temperature. Comment.
Calculate the mole fraction of water in a sodium hydroxide solution which has 80 g of NaOH and 54 g of H2O. (Relative atomic masses: Na = 23, O = 16, H = 1)
The depression in the freezing point of a sugar solution was found to be 0.402°C. Calculate the osmotic pressure of the sugar solution at 27°C. (Kf = 1.85 K kg mol−1)
A solution is prepared by dissolving 2.0 g of sucrose and 2.0 g of urea in 100 g of water at 298 K. Calculate the vapour pressure of the solution, if the vapour pressure of pure water at 298 K is 23.756 torr. (Mol. wt. of urea = 60 and sucrose = 342)
If the molality of an aqueous solution of cane sugar is 0.4445, what is the mole fraction of cane sugar?
Albumins are the most abundant proteins in blood. At 25°C, 3.5g of albumin in 100 ml of water produces an osmotic pressure of 0.014 atm. What is the molecular weight of albumin?
Schematically represent the temperature-composition curve for the vapour in equilibrium with an ideal mixture of two completely miscible liquids of different boiling points. Explain with the aid of this diagram how fractional distillation is done.
The boiling point of pure water is 373 K. Calculate the boiling point of an aqueous solution containing 18 g of glucose (MW = 180) in 100 g of water. Molal elevation constant of water is 0.52 K kg mol−1.
Equal weights of two substances X and Y are dissolved in equal volumes of water. The osmotic pressure of the solution containing Y is five times the osmotic pressure of the solution containing X. What is the molecular weight of X if that of Y is 60?
Which of the following solutions will have a lower vapour pressure and why?
- A 5% solution of cane sugar (C12H22O11)
- A 5% solution of urea (NH2CONH2)
(Relative atomic masses: H = 1, C = 12, O = 16, N = 14)
Out of the following solutions, the one having the highest boiling point will be:
0.1 M NaCl
0.1 M BaCl2
0.1 M KNO3
0.1 M K4Fe(CN)6
Answer the following question:
0.1 M urea solution shows less depression in freezing point than 0.1 M MgCl2 solution. Explain.
46 g of ethyl alcohol is dissolved in 18 g of water. Calculate the mole fraction of ethyl alcohol. (at. wt. of C = 12, O = 16, H = 1).
The osmotic pressure of 0.01 molar solution of an electrolyte is found to be 0.65 atm at 27°C. Calculate the van’t Hoff factor. What conclusion can you draw about the molecular state of the solute in the solution?
Fill in the blanks:
Solutions which strictly obey ______ law are called ______ solutions.
A solution X is prepared by dissolving three moles of glucose in one litre of water and a solution Y is prepared by dissolving 1.5 moles of sodium chloride in one litre of water. Will the osmotic pressure of X be higher, lower or equal to that of Y? Give a reason for your answer.
A solution of urea in water has a boiling point of 100.18°C. Calculate the freezing point of the solution. (Kf for water is 1.86 K kg mol−1 and Kb for water is 0.512 K kg mol−1).
A solution of lactose containing 8.45 g of lactose in 100 g of water has a vapour pressure of 4.559 mm of Hg at 0°C. If the vapour pressure of pure water is 4.579 mm of Hg, calculate the molecular weight of lactose.
Fill in the blanks:
An aqueous solution of sugar boils ______ 100°C and freezes ______°C.
Among equimolal solutions of MgCl2, NaCl, FeCl3 and C12H22O11, which will show minimum osmotic pressure? Why?
A certain aqueous solution boils at 100.303°C. What is its freezing point? Kb for water = 0.5 K kg mol−1 and Kf = 1.87 K kg mol−1?
A solution containing 1 g of sodium chloride in 100 g of water freezes at 0.604°C. Calculate the degree of dissociation of sodium chloride. (Na = 23, Cl = 35.5, Kf for water = 1.87 K mol−1)
Complete the following statements by selecting the correct alternative from the choices given
Of the following terms used for denoting the concentration of a solution, the one which does not get affected by temperature is:
Molarity
Molality
Normality
Formality
Define Raoult’s law for the elevation of the boiling point of a solution.
What will be the vapour pressure of a solution containing 5 moles of sucrose (C12H22O11) in 1 kg of water, if the vapour pressure of pure water is 4.57 mm of Hg? [C = 12, H = 1, O = 16]
A 2 molal solution of sodium chloride in water causes an elevation in the boiling point of water by 1.88 K. What is the value of Van’t Hoff factor? What does it signify? [Kb = 0.52 K kg mol−1]
Two liquids A and B form type II non-ideal solution which shows a minimum in its temperature-mole fraction plot (T-χ diagram). Can the two liquids be completely separated by fractional distillation?
A solution containing 0.5 g of KCl dissolves in 100 g of water and freezes at −0·24°C. Calculate the degree of dissociation of the salt. (Kf for water = 1.86°C) Atomic weights, K = 39, Cl = 35.5.
If 1.71 g of sugar (molar mass = 342) are dissolved in 500 ml of an aqueous solution at 300 K, what will be its osmotic pressure?
0.70 g of an organic compound, when dissolved in 32 g of acetone, produces an elevation of 0.25°C in the boiling point. Calculate the molecular mass of the organic compound (Kb for acetone = 1.72 K kg mol−1).
Fill in the blanks:
Ideal solutions obey ______ law and they ______ form azeotropic mixtures.
A 10% aqueous solution of cane sugar (mol. wt. 342) is isotonic with 1.754% aqueous solution of urea. Find the molecular mass of urea.
The molecular weight of an organic compound is 58 g mol−1. What will be the boiling point of a solution containing 48 grams of the solute in 1200 grams of water?
[Kb for water = 0.513°C kg mol−1, Boiling point of water = 100°C]
What will be the value of Van’t Hoff factor (i) of benzoic acid if it dimerises in aqueous solution? How will the experimental molecular weight vary as compared to the normal molecular weight?
Why the freezing point depression (ΔTf) of 0.4 M NaCl solution is nearly twice than that of 0.4 M glucose solution?
Determine the freezing point of a solution containing 0.625 g of glucose (C6H12O6) dissolved in 102.8 g of water.
(Freezing point of water = 273 K, Kf for water = 1.87 K kg mol−1, at. wt. C = 12, H = 1, O = 16)
A 0.15 M aqueous solution of KCl exerts an osmotic pressure of 6.8 atm at 310 K. Calculate the degree of dissociation of KCl. (R = 0.0821 L atm K−1 mol−1).
A solution containing 8.44 g of sucrose in 100 g of water has a vapour pressure 4.56 mm of Hg at 273 K. If the vapour pressure of pure water is 4.58 mm of Hg at the same temperature, calculate the molecular weight of sucrose.
Choose the correct alternative:
An aqueous solution of urea freezes at –0.186°C, Kf for water = 1.86 K kg mol–1, Kb for water = 0.512 K kg mol–1. The boiling point of urea solution will be ______.
373.065 K
373.186 K
373.512 K
373.0512 K
Determine the osmotic pressure of a solution prepared by dissolving 25 mg of K2SO4 in 2 liter of water at 25°C, assuming that it is completely dissociated.
An aqueous solution containing 12.48 g of barium chloride (BaCl2) in 1000 g of water, boils at 100.0832°C. Calculate the degree of dissociation of barium chloride.
(Kb for water = 0.52 K kg mol−1, at. wt. Ba = 137, Cl = 35.5)
The freezing point of a solution containing 5.85 g of NaCl in 100 g of water is −3.348°C. Calculate Van’t Hoff factor ‘i’ for this solution. What will be the experimental molecular weight of NaCl?
(Kf for water = 1.86 K kg mol−1, at. wt. Na = 23, Cl = 35.5)
Calculate the boiling point of urea solution when 6 g of urea is dissolved in 200 g of water.
(Kb for water = 0.52 K kg mol–1, boiling point of pure water = 373 K, mol. wt. of urea = 60)
When 0.4 g of acetic acid is dissolved in 40 g of benzene, the freezing point of the solution is lowered by 0.45 K. Calculate the degree of association of acetic acid. Acetic acid forms dimer when dissolved in benzene.
(Kf for benzene = 5.12 K kg mol−1, at. wt. C = 12, H = 1, O = 16)
A solution is prepared by dissolving 9.25 g of non-volatile solute in 450 mL of water. It has an osmotic pressure of 350 mm of Hg at 27°C. Assuming the solute is non-electrolyte, determine its molecular mass. (R = 0.0821 Lit-atm K−1 mol−1)
The elevation in boiling point when 0.30 g of acetic acid is dissolved in 100 g of benzene is 0.0633°C. Calculate the molecular weight of acetic acid from this data. What conclusion can you draw about the molecular state of the solute in the solution?
(Given Kb for benzene = 2.53 K kg mol−1, at. wt. of C = 12, H = 1, O = 16)
Determine the osmotic pressure of a solution prepared by dissolving 0.025 g of K2SO4 in 2 litres of water at 25°C, assuming that K2SO4 is completely dissociated.
(R = 0.0821 Lit-atm K−1 mol−1, mol. wt. of K2SO4 = 174 g mol−1)
An aqueous solution of a non-volatile solute freezes at 272.4 K, while pure water freezes at 273.0 K. Determine the following:
(Given Kf = 1.86 K kg mol−1, Kb = 0.512 K kg mol−1 and vapour pressure of water at 298 K = 23.756 mm of Hg)
- The molality of solution
- Boiling point of solution
- The lowering of vapour pressure of water at 298 K.
A solution containing 1.23 g of calcium nitrate in 10 g of water, boils at 100.975°C at 760 mm of Hg. Calculate the van’t Hoff factor for the salt at this concentration.
(Kb for water = 0.52 K kg mol−1, mol. wt. of calcium nitrate = 164 g mol−1)
Select and write the correct alternative from the choices given below
A solution containing components A and B follows Raoult’s law. With reference to the statement, which of the following options is correct?
A-B attraction force is greater than A-A and B-B.
A-B attraction force is less than A-A and B-B.
A-B attraction force remains the same as A-A and B-B.
Total volume of solution is different from sum of volumes of both components A and B.
A solution of urea (molecular mass = 60) contains 8.6 g per litre. It is isotonic with a 5% solution of a non-volatile and non-electrolytic solute. What will be the molecular mass of the solute?
34.9 g mol−1
349 g mol−1
861 g mol−1
3490 g mol−1
The value of molal depression constant or cryoscopic constant (Kf) depends on which of the following?
Nature of solvent
Heat of the solution of the solute in the solvent
Nature of solute
Vapour pressure of the solution
According to which law the solubility of a gas at equilibrium and constant temperature varies directly with the pressure of the gas, provided the gas does not undergo any chemical change during the dissolution?
Raoult’s law
Nernst distribution law
Henry’s law
van’t hoff law
0.76 g of glucose (molecular mass = 180 g mol−1) is dissolved in 20 ml of aqueous solution at 298 K (R = 0.0821 L atm K−1 mol−1).
What is the osmotic pressure of solution at 298 K?
8.41 atm
0.48 atm
4.41 atm
5.16 atm
0.76 g of glucose (molecular mass = 180 g mol−1) is dissolved in 20 ml of aqueous solution at 298 K (R = 0.0821 L atm K−1 mol−1).
What is the molarity of glucose solution?
0.42 M
0.21 M
4.01 M
2.02 M
5 moles of sucrose (molecular mass = 342 g mol−1) dissolved in 1000 g of water (molecular mass = 18 g mol−1). Vapour pressure of pure water at 298 K = 4.57 mm Hg.
What will be the vapour pressure of sucrose solution at the same temperature?
0.419 mm Hg
6.570 mm Hg
4.190 mm Hg
0.657 mm Hg
5 moles of sucrose (molecular mass = 342 g mol−1) dissolved in 1000 g of water (molecular mass = 18 g mol−1). Vapour pressure of pure water at 298 K = 4.57 mm Hg.
What will be the mole fraction of sucrose in water?
0.8261
0.0826
0.4376
0.0435
A solution containing 54 g of glucose (molecular mass = 180 g mol−1) in 250 g of water (Kf for water = 1.86 K kg mol−1).
What will be the freezing point of this glucose solution?
276.402 K
270.768 K
370.402 K
272.563 K
A solution containing 54 g of glucose (molecular mass = 180 g mol−1) in 250 g of water (Kf for water = 1.86 K kg mol−1).
What will be the molality of this glucose solution?
1.20 m
0.12 m
2.40 m
0.24 m
Solutions for 2: Solutions
![Nootan solutions for केमिस्ट्रि [इंग्रजी] इयत्ता १२ आयएससी chapter 2 - Solutions Nootan solutions for केमिस्ट्रि [इंग्रजी] इयत्ता १२ आयएससी chapter 2 - Solutions - Shaalaa.com](/images/chemistry-english-class-12-isc_6:a6407550bf9a46188b122e0d354548f8.png)
Nootan solutions for केमिस्ट्रि [इंग्रजी] इयत्ता १२ आयएससी chapter 2 - Solutions
Shaalaa.com has the CISCE Mathematics केमिस्ट्रि [इंग्रजी] इयत्ता १२ आयएससी CISCE solutions in a manner that help students grasp basic concepts better and faster. The detailed, step-by-step solutions will help you understand the concepts better and clarify any confusion. Nootan solutions for Mathematics केमिस्ट्रि [इंग्रजी] इयत्ता १२ आयएससी CISCE 2 (Solutions) include all questions with answers and detailed explanations. This will clear students' doubts about questions and improve their application skills while preparing for board exams.
Further, we at Shaalaa.com provide such solutions so students can prepare for written exams. Nootan textbook solutions can be a core help for self-study and provide excellent self-help guidance for students.
Concepts covered in केमिस्ट्रि [इंग्रजी] इयत्ता १२ आयएससी chapter 2 Solutions are Overview of Solutions, Introduction to Solutions, Types of Solutions, Composition of a Solution, Intensive and Extensive Properties, Colligative Properties, Non-Volatile, Non-Electrolytic and Electrolytic Solutes, Dissociation and Association, Solutions of Gases in Liquids, Solid Solutions, Solutions of Solids in Liquids, Ideal Solutions, Non-Ideal Solutions, Types of Non-Ideal Solutions, Principle of Fractional Distillation and Azeotropic (Constant Boiling) Mixtures, Relative Lowering of Vapour Pressure, Elevation of Boiling Point, Depression of Freezing Point, Osmosis and Osmotic Pressure, Abnormal Molecular Masses, Association and Dissociation of Solute Molecules : Cause of Abnormal Molecular Masses, Van’t Hoff Factor, Calculation of Extent of Association or Dissociation of a Solute in Solution.
Using Nootan केमिस्ट्रि [इंग्रजी] इयत्ता १२ आयएससी solutions Solutions exercise by students is an easy way to prepare for the exams, as they involve solutions arranged chapter-wise and also page-wise. The questions involved in Nootan Solutions are essential questions that can be asked in the final exam. Maximum CISCE केमिस्ट्रि [इंग्रजी] इयत्ता १२ आयएससी students prefer Nootan Textbook Solutions to score more in exams.
Get the free view of Chapter 2, Solutions केमिस्ट्रि [इंग्रजी] इयत्ता १२ आयएससी additional questions for Mathematics केमिस्ट्रि [इंग्रजी] इयत्ता १२ आयएससी CISCE, and you can use Shaalaa.com to keep it handy for your exam preparation.
