Advertisements
Advertisements
प्रश्न
Vapour pressure of pure acetone and chloroform at 328 K are 741.8 mm Hg and 632.8 mm Hg respectively. Assuming that they form ideal solution over the entire range of composition, plot `p_"total"`, `p_"chloroform"` and `p_"acetone"` as a function of `chi_"acetone"`. The experimental data observed for different compositions of mixtures is:
| `bb(100 xx chi_"acetone")` | `bb(P_"acetone"//"mm Hg")` | `bb(p_"chloroform"//"mm Hg")` |
| 0 | 0 | 632.8 |
| 11.8 | 54.9 | 548.1 |
| 23.4 | 110.1 | 469.4 |
| 36 | 202.4 | 359.7 |
| 50.8 | 322.7 | 257.7 |
| 58.2 | 405.9 | 193.6 |
| 64.5 | 454.1 | 161.2 |
| 72.1 | 521.1 | 120.7 |
Plot this data also on the same graph paper. Indicate whether it has a positive or negative deviation from the ideal solution.
Advertisements
उत्तर
| `bb(100 xx chi_"acetone")` | `bb(P_"acetone"//"mm Hg")` | `bb(p_"chloroform"//"mm Hg")` | `bb("P"_"total" ("mm Hg"))` |
| 0 | 0 | 632.8 | 632.8 |
| 11.8 | 54.9 | 548.1 | 603.0 |
| 23.4 | 110.1 | 469.4 | 579.5 |
| 36 | 202.4 | 359.7 | 562.1 |
| 50.8 | 322.7 | 257.7 | 580.4 |
| 58.2 | 405.9 | 193.6 | 595.5 |
| 64.5 | 454.1 | 161.2 | 615.3 |
| 72.1 | 521.1 | 120.7 | 641.8 |
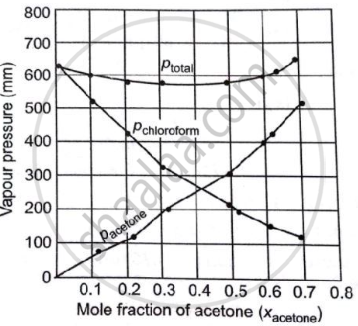
It can be observed from the graph that the plot for the ptotal of the solution curves downward. Therefore, the solution shows a negative deviation from the ideal behaviour.
APPEARS IN
संबंधित प्रश्न
What role does the molecular interaction play in a solution of alcohol and water?
Fill in the blanks by choosing the appropriate word/words from those given in the brackets:
Ideal solutions obey ………. law and they …………. form azeotropic mixtures.
(Henry’s, aldol condensation, absence, do not, ohm, Raoult’s, increases, common ion effect, easily, three, solubility product, ohm-1, two, four, ohm-1, cm2, Cannizzaro, ohm-1 cm-1, zero, decreases, presence)
Two liquids X and Y on mixing gives a warm solution. The solution is ______.
The observed depression in the freezing point of water for a particular solution is 0.093°C calculate the concentration of the solution in molality. Given that molal depression constant for water is 1.86 K Kg mol-1.
For ideal solution the enthalpy of mixing of the pure components to form the solution is ____________.
The rate at which a solid dissolves in liquid does not depend on ____________.
Which of the following condition is not satisfied by an ideal solution?
If two liquids A and B form minimum boiling azeotrope at some specific composition then ______.
What is “semi permeable membrane”?
The pH of a solution of hydrochloric acid is 1.5. Calculate the concentration of the acid.
On increase in pressure for dissociation ofN2O4 into NO2, equilibrium shift towards
Lowering of vapour pressure of an aqueous solution of a non-volatile, non-electrolyte 1 molal aqueous solution at 100°C is ______.
If liquids A and B form an ideal solution, the ______.
1 mole of liquid A and 2 moles of liquid B make a solution having a total vapour pressure of 40 torr. The vapour pressure of pure A and pure B are 45 torr and 30 torr, respectively. The above solution ______.
What type of deviation from Roult’s Law is expected when phenol and aniline are mixed with each other? What change in the net volume of the mixture is expected? Graphically represent the deviation.
Suggest the most important type of intermolecular attractive interaction in the following pair.
n-hexane and n-octane
Suggest the most important type of intermolecular attractive interaction in the following pair.
NaClO4 and water
5 moles of liquid X and 10 moles of liquid Y make a solution having a vapour pressure of 70 torr. The vapour pressures of pure X and Y are 63 torr and 78 torr respectively. Which of the following is true regarding the described solution?
