Advertisements
Advertisements
प्रश्न
Vapour pressure of pure acetone and chloroform at 328 K are 741.8 mm Hg and 632.8 mm Hg respectively. Assuming that they form ideal solution over the entire range of composition, plot `p_"total"`, `p_"chloroform"` and `p_"acetone"` as a function of `chi_"acetone"`. The experimental data observed for different compositions of mixtures is:
| `bb(100 xx chi_"acetone")` | `bb(P_"acetone"//"mm Hg")` | `bb(p_"chloroform"//"mm Hg")` |
| 0 | 0 | 632.8 |
| 11.8 | 54.9 | 548.1 |
| 23.4 | 110.1 | 469.4 |
| 36 | 202.4 | 359.7 |
| 50.8 | 322.7 | 257.7 |
| 58.2 | 405.9 | 193.6 |
| 64.5 | 454.1 | 161.2 |
| 72.1 | 521.1 | 120.7 |
Plot this data also on the same graph paper. Indicate whether it has a positive or negative deviation from the ideal solution.
Advertisements
उत्तर
| `bb(100 xx chi_"acetone")` | `bb(P_"acetone"//"mm Hg")` | `bb(p_"chloroform"//"mm Hg")` | `bb("P"_"total" ("mm Hg"))` |
| 0 | 0 | 632.8 | 632.8 |
| 11.8 | 54.9 | 548.1 | 603.0 |
| 23.4 | 110.1 | 469.4 | 579.5 |
| 36 | 202.4 | 359.7 | 562.1 |
| 50.8 | 322.7 | 257.7 | 580.4 |
| 58.2 | 405.9 | 193.6 | 595.5 |
| 64.5 | 454.1 | 161.2 | 615.3 |
| 72.1 | 521.1 | 120.7 | 641.8 |
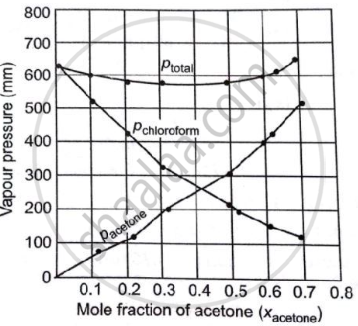
It can be observed from the graph that the plot for the ptotal of the solution curves downward. Therefore, the solution shows a negative deviation from the ideal behaviour.
APPEARS IN
संबंधित प्रश्न
How is the sign of ΔmixH related to positive deviations from Raoult’s law?
Suggest the most important type of intermolecular attractive interaction in the following pair.
acetonitrile (CH3CN) and acetone (C3H6O)
What type of intermolecular attractive interaction exists in the pair of methanol and acetone?
The molality of a solution containing 1.8 g of glucose dissolved in 250 g of water is
Which of the following concentration terms is/are independent of temperature
Which one of the following is incorrect for an ideal solution?
Assertion: An ideal solution obeys Raoults Law.
Reason: In an ideal solution, solvent – solvent as well as solute – solute interactions are similar to solute-solvent interactions.
Which solution has the lower freezing point? 10 g of methanol (CH3OH) in 100g g of water (or) 20 g of ethanol (C2H5OH) in 200 g of water.
For ideal solution the volume of mixing of the pure components to form the solution is ____________.
The rate at which a solid dissolves in liquid does not depend on ____________.
Intermolecular forces between two benzene molecules are nearly of same strength as those between two toluene molecules. For a mixture of benzene and toluene, which of the following are not true?
(i) ∆mix H = zero
(ii) ∆mix V = zero
(iii) These will form minimum boiling azeotrope.
(iv) These will not form ideal solution.
Concentration terms such as mass percentage, ppm, mole fraction and molality are independent of temperature, however molarity is a function of temperature. Explain.
Why soda water bottle kept at room temperature fizzes on opening?
Explain the terms ideal and non-ideal solutions in the light of forces of interactions operating between molecules in liquid solutions.
Upon increase in pressure for dissociation N2O4 into NO2, equilibrium shift towards
On increase in pressure for dissociation ofN2O4 into NO2, equilibrium shift towards
Lowering of vapour pressure of an aqueous solution of a non-volatile, non-electrolyte 1 molal aqueous solution at 100°C is ______.
