Advertisements
Advertisements
प्रश्न
What type of deviation from Roult’s Law is expected when phenol and aniline are mixed with each other? What change in the net volume of the mixture is expected? Graphically represent the deviation.
Advertisements
उत्तर
Negative Deviation is expected when phenol and aniline are mixed with each other. The net volume of the mixture will decrease, ΔV < 0 due to stronger intermolecular interactions.
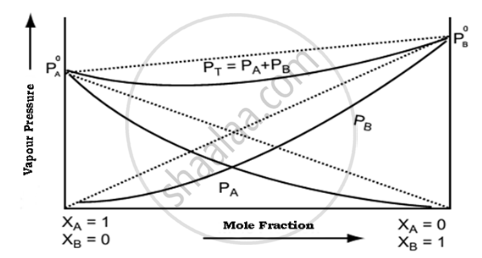
Diagram showing negative deviation from Raoult’s law
APPEARS IN
संबंधित प्रश्न
What role does the molecular interaction play in a solution of alcohol and water?
Define an ideal solution and write one of its characteristics.
Two liquids X and Y on mixing gives a warm solution. The solution is ______.
Assertion: An ideal solution obeys Raoults Law.
Reason: In an ideal solution, solvent – solvent as well as solute – solute interactions are similar to solute-solvent interactions.
State Raoult law and obtain the expression for lowering of vapour pressure when the nonvolatile solute is dissolved Insolvent.
Which of the following is an example of a solid solution?
If two liquids A and B form minimum boiling azeotrope at some specific composition then ______.
Concentration terms such as mass percentage, ppm, mole fraction and molality are independent of temperature, however molarity is a function of temperature. Explain.
On increase in pressure for dissociation ofN2O4 into NO2, equilibrium shift towards
Suggest the most important type of intermolecular attractive interaction in the following pair.
n-hexane and n-octane
