Advertisements
Advertisements
Question
What type of deviation from Roult’s Law is expected when phenol and aniline are mixed with each other? What change in the net volume of the mixture is expected? Graphically represent the deviation.
Advertisements
Solution
Negative Deviation is expected when phenol and aniline are mixed with each other. The net volume of the mixture will decrease, ΔV < 0 due to stronger intermolecular interactions.
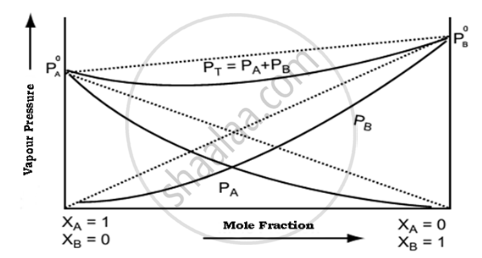
Diagram showing negative deviation from Raoult’s law
APPEARS IN
RELATED QUESTIONS
How is the sign of ΔmixH related to positive deviations from Raoult’s law?
Vapour pressure of pure acetone and chloroform at 328 K are 741.8 mm Hg and 632.8 mm Hg respectively. Assuming that they form ideal solution over the entire range of composition, plot ptotal, `p_"chloroform"` and `p_"acetone"` as a function of `chi_"acetone"`. The experimental data observed for different compositions of mixtures is:
| `bb(100 xx chi_"acetone")` | `bb(P_"acetone"//"mm Hg")` | `bb(p_"chloroform"//"mm Hg")` |
| 0 | 0 | 632.8 |
| 11.8 | 54.9 | 548.1 |
| 23.4 | 110.1 | 469.4 |
| 36.0 | 202.4 | 359.7 |
| 50.8 | 322.7 | 257.7 |
| 58.2 | 405.9 | 193.6 |
| 64.5 | 454.1 | 161.2 |
| 72.1 | 521.1 | 120.7 |
Plot this data also on the same graph paper. Indicate whether it has a positive or negative deviation from the ideal solution.
Two liquids X and Y on mixing gives a warm solution. The solution is ______.
For ideal solution the enthalpy of mixing of the pure components to form the solution is ____________.
Which of the following is an example of a solid solution?
Intermolecular forces between two benzene molecules are nearly of same strength as those between two toluene molecules. For a mixture of benzene and toluene, which of the following are not true?
(i) ∆mix H = zero
(ii) ∆mix V = zero
(iii) These will form minimum boiling azeotrope.
(iv) These will not form ideal solution.
Assertion: When a solution is separated from the pure solvent by a semipermeable membrane, the solvent molecules pass through it from pure solvent side to the solution side.
Reason: Diffusion of solvent occurs from a region of high concentration solution to a region of low concentration solution.
On increase in pressure for dissociation ofN2O4 into NO2, equilibrium shift towards
While titration dilute HCl solution with aqueous NaOH, which of the following will not be required?
Suggest the most important type of intermolecular attractive interaction in the following pair.
n-hexane and n-octane
