Definitions [19]
Compounds in which one or more hydrogen atom(s) directly bonded to an aromatic ring is (are) replaced by halogen.
General formula: Ar–X.
Compounds in which one or more hydrogen atom(s) of an alkane is (are) replaced by halogen.
General formula: R–X.
Define racemic mixture.
A mixture containing two enantiomers in equal proportions will have zero optical rotation, as the rotation due to one isomer will be cancelled by the rotation due to the other isomer. Such a mixture is called a racemic mixture or a racemic modification.
The reaction in which diazonium salt is treated with cuprous halide to form haloarene is called Sandmeyer reaction.
The reaction in which halogen is added across a carbon-carbon double bond is called halogenation reaction.
The reaction in which alkyl chloride or bromide is converted into alkyl fluoride using metallic fluorides is called Swarts reaction.
The organic compounds in which one or more hydrogen atoms of hydrocarbons are replaced by halogen atoms are called halogen derivatives.
The compounds in which halogen atom is bonded to an sp³ hybridised carbon atom of an alkyl group are called haloalkanes.
The compounds in which halogen atom is directly bonded to an sp² hybridised carbon atom of an aromatic ring are called haloarenes.
The alkyl halide in which halogen is attached to a secondary carbon atom is called secondary alkyl halide.
The alkyl halide in which halogen is attached to a tertiary carbon atom is called tertiary alkyl halide.
The compounds in which halogen is bonded to an sp³ carbon adjacent to a carbon-carbon double bond are called allylic halides.
The compounds in which halogen is bonded to an sp³ carbon attached to an aromatic ring are called benzylic halides.
The compounds in which halogen is bonded to an sp² carbon of a carbon-carbon double bond are called vinylic halides.
The reaction in which a halogen atom in an alkyl halide is replaced by a nucleophile is called nucleophilic substitution reaction.
The reaction in which a halogen atom is removed along with a hydrogen atom forming a double bond is called elimination reaction.
The reaction in which alkyl chloride or bromide reacts with sodium iodide in acetone to form alkyl iodide is called Finkelstein reaction.
The formation of equal amounts of two enantiomers resulting in optically inactive mixture is called racemisation.
The alkyl halide in which halogen is attached to a primary carbon atom is called primary alkyl halide.
Key Points
On the Basis of Number of Halogen Atoms-
| Type | Description | Example |
|---|---|---|
| Monohalogen | One H replaced by one halogen | R–CH₂X |
| Dihalogen | Two H replaced (gem or vic) | R–CHX₂ |
| Trihalogen | Three H replaced | R–CX₃ |
| Tetrahalogen | Four H replaced | CX₄ |
Gem dihalide: Both halogens on the same carbon (e.g., 1,1-dichloroethane).
Vic dihalide: Halogens on adjacent carbons (e.g., 1,2-dichloroethane).
On the Basis of Type of Carbon Bearing the Halogen:
| Type | Description | Example |
|---|---|---|
| Primary (1°) | –X bonded to 1° carbon | RCH₂X |
| Secondary (2°) | –X bonded to 2° carbon | R₂CHX |
| Tertiary (3°) | –X bonded to 3° carbon | R₃CX |
| Allylic | –X bonded to allylic carbon | CH₂=CH–CH₂X |
| Benzylic | –X bonded to benzylic carbon | C₆H₅–CH₂X |
| Vinylic | –X bonded to sp² carbon of C=C | CH₂=CH–X |
| Aryl halide | –X bonded directly to aromatic ring | C₆H₅–X (Ar–X) |
Alkyl halide carbon (with X) is sp³ hybridised; aryl halide carbon is sp² hybridised — this is why aryl C–X bond is shorter and stronger.
Common Names:
- Alkyl halide or aryl halide
- e.g., CH₃Cl → Methyl chloride; CH₂=CHCl → Vinyl chloride
IUPAC Names:
- Haloalkane or arylhalide
- Rule 1: Find the longest carbon chain containing the halogen. If a double/triple bond is present, give it priority.
- Rule 2: Number from the end nearer the first substituent. Assign each substituent a position number.
- Multiple same halogens → di-, tri-, tetra- prefix.
- Different halogens → list alphabetically and number to give the alphabetically first halogen the lowest possible number.
Examples:
- CH₃Cl → Chloromethane
- CH₂=CHCl → Chloroethene
- (CH₃)₃CCl → 2-Chloro-2-methylpropane (common: tert-butyl chloride)
- 2-Chloro-1-methylbenzene → o-Chlorotoluene → IUPAC: 1-Chloro-2-methylbenzene
Formation of Alkyl Halide from Alcohols:
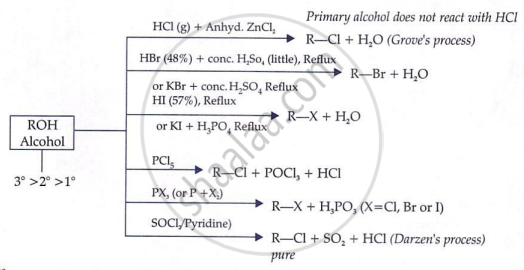
From Alkenes
(a) Addition of halogen acids:
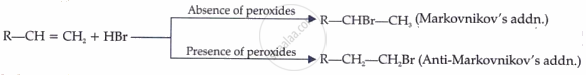
(b) Allylic halogenation:
\[\ce{\underset{Propene}{CH3 - CH} = CH2 + Cl2 ->[775K] \underset{\underset{(Allyl chloride)}{3-Chloro-1-propene}}{ClCH2 - CH = CH2 + HCl}}\]
From alkanes (Swarts reaction)
Alkyl chloride or bromide react with AgF, SbF3, or Hg2F2 give alkyl fluoride. This reaction is known Swarts reaction. Antimony trifluoride (SbF2) is commonly used in this reaction.
2CH3CH3CI + Hg2F2→ 2CH3CH2F+ Hg2Cl2

From the halide exchange method (Finkelstein reaction)
Alkyl chloride or bromide react with Nal or KI in presence of acetone give alkyl iodide. It is halide exchange reaction or Finkelstein reaction. It is a SN2 reaction.
\[\ce{R - Cl + Nal ->[Acetone or Methanol] R - I + NaCl}\]
\[\begin{array}{cc}
\phantom{..........}\ce{Cl}\phantom{................................................}\ce{I}\phantom{..}\\
\phantom{..........}|\phantom{..................................................}|\phantom{.}\\
\phantom{}\ce{CH3 - CH2 - CH - CH3 ->[Nal][Acetone] CH3 - CH2 - CH - CH3}\phantom{}
\end{array}\]
Borodine-Hunsdiecker reaction
Silver salt of carboxylic acid on heating with Br2 + carbon tetrachloride give alkyl bromide
\[\ce{\underset{Silver acetate}{CH3COOAg} + Br2 ->[CCl4/Reflux] \underset{Methyl bromide}{CH3 - Br} + AgBr + CO2}\]
Note: If silver salt of carboxylic acid is heated with I2 in presence of CCI4 ester is obtained. This reaction is known as simonini reaction.
\[\ce{2RCOOAg + I2 ->[CCl4] \underset{Ester}{RCOOR} + 2CO2 + 2AgI}\]
If silver salt of carboxylic acid is heated with I2 in presence of HgO or lead tetra acetate than alkyl iodide is obtained
\[\ce{2RCOOAg ->[I2/HgO][Δ] 2R - X + 2CO2 + HgI2 + H2O}\]
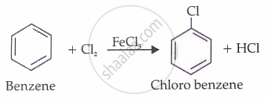
Direct halogenations
Chlorobenzene is prepared by direct chlorination of benzene in the presence of Lewis acid catalysts such as FeCl3.
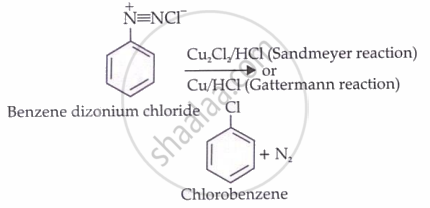
From benzene diazonium chloride
Chlorobenzene is prepared by the Sandmeyer reaction or the Gattermann reaction using benzene diazonium chloride.
Sandmeyer reaction
When an aqueous solution of benzene diazonium chloride is warmed with Cu2Cl2 in HCl, chlorobenzene is formed.
Preparation of iodobenzene
Iodobenzene is prepared by warming benzene diazonium chloride with aqueous KI solution.
\[\ce{\underset{(\underset{\underset{chloride}{benzene diazonium}}{Sandmeyer rection)}}{C6H5N2Cl + Kl} ->[warm] \underset{Iodo benzene}{C6H5I + N2 + KCl}}\]
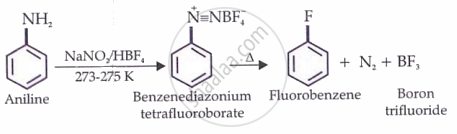
Preparation of fluorobenzene
Aryl fluorides are prepared by
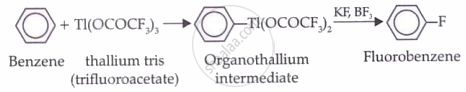
When a diazonium salt is treated with fluoroboric acid (HBF), the diazonium fluoro borate precipitates out of solution. If this precipitated salt is filtered and then heated, it decomposes to give the aryl fluoride.
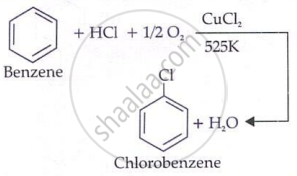
Commercial preparation of chloro benzene (Raschig process)
Chlorobenzene is commercially prepared by passing a mixture of benzene vapour, air and HCl over heated cupric chloride. This reaction is called the Raschig process.
Melting and Boiling Points
- Depend on Van der Waals dispersion forces and dipole–dipole interactions.
- Boiling point ∝ size of halogen atom and number of electrons: R–I > R–Br > R–Cl > R–F (for the same carbon chain)
- Boiling point ∝ surface area ∝ no. of carbons in chain (longer chain → higher B.P.)
- Branching reduces B.P.: B.P. ∝ 1/branching (isomers go from primary → tertiary, B.P. falls)
- Para-isomers of dihalobenzenes have higher melting points than ortho- and meta-isomers due to symmetry fitting better in the crystal lattice.
Density
- Density ∝ no. of halogen atoms / molecular mass.
- Bromo, iodo, and polychloro derivatives are heavier than water: Density: R–I > R–Br > R–Cl > R–F
- For isomers of chlorobenzene: density ∝ molecular mass → benzene < chlorobenzene < dichlorobenzene < bromochlorobenzene.
Solubility
- Haloalkanes are very slightly soluble in water (attraction between alkyl halide molecules is stronger than attraction between alkyl halide and water, and they fail to form H-bonds with water).
- Solubility order in water: R–F > R–Cl > R–Br > R–I
- Haloalkanes dissolve readily in organic solvents (due to similar intermolecular forces).
Alkyl iodide is so unstable that it decomposes in sunlight: 2R–I → 2R + I₂ (violet vapours)
The C–X bond in alkyl halides is polarised (Cδ+–Xδ–), making alkyl halides reactive towards nucleophiles.
Two Types of SN Reactions
SN1 (Unimolecular Nucleophilic Substitution):
- First-order kinetics: Rate = k[RX] (depends only on substrate concentration)
- Two-step mechanism: Step 1 (slow) — ionisation to form carbocation; Step 2 (fast) — attack by nucleophile.
- Intermediate: Trigonal planar carbocation.
- More substituted alkyl halides react faster (more stable carbocation).
- Reactivity order: R₃CX > R₂CHX > RCH₂X (3° > 2° > 1°)
- Gives a racemic mixture (optically inactive product) because the nucleophile can attack from both faces.
- For aryl/vinyl halides: Ar₂CX > Ar₂CHX > ArCH₂X = CH₂=CHX > CH₂=CHCH₂X
SN2 (Bimolecular Nucleophilic Substitution):
- Second-order kinetics: Rate = k[RX][Nu] (depends on both substrate and nucleophile concentration)
- One-step mechanism (concerted): Nucleophile attacks from the back side as leaving group departs simultaneously → Transition State is formed.
- Results in Walden Inversion (inversion of configuration at the carbon — stereochemistry inverted).
- Reactivity order: Methyl halide > Primary > Secondary > Tertiary (CH₃X > 1° > 2° > 3°)
- The SN2 reaction rate depends on the concentration of both alkyl halide and nucleophile.
β-Elimination Reaction:
- When alkyl halides are heated with alcoholic KOH or KNH₂, they undergo β-elimination of HX to form an alkene (new π bond).
- The carbon directly attached to X = α-carbon; the carbon adjacent to it = β-carbon.
- Order of reactivity in elimination: R–Cl < R–Br < R–I
Saytzeff's Rule (Zaitsev's Rule):
- In unsymmetrical alkyl halides, hydrogen is preferentially eliminated from the β-carbon with fewer hydrogen atoms → forms the more highly substituted alkene (major product).
- e.g., 2-bromopentane → pent-2-ene (81%) [major] + pent-1-ene (19%) [minor]
Types of Elimination:
- α-elimination: Atom or group lost from the same carbon (gives carbene intermediates).
- β-elimination: H from β-carbon, X from α-carbon → alkene.
- E₁ reaction: Two steps (similar mechanism to SN1)
- E₂ reaction: One step (concerted, anti-periplanar geometry required — similar to SN2 but gives alkene)
Dehydrohalogenation:
- Loss of HX from alkyl halide with alc. KOH → alkene.
\[\ce{\underset{Alky halide}{C_{n}H_{2n + 1}X} ->[Alcholic KOH] \underset{Alkene}{C_{n}H_{2n}} + KX + H2O}\]
- With NaOH, Con. NH₃, t-BuONa, KNH₂, NaNH₂: elimination also occurs.
With magnesium: \[\ce{RX + Mg ->[Dry][Ether] RMgX (Grignard reagent)}\]
With sodium (Wurtz Reaction) \[\ce{-> RX + 2Na + XR ->[Dry ether] R - R + 2NaX}\]
Reduction: \[\ce{RX + 2H ->[Zn/HCl (conc)][or Zn-Cu/C2H5OH]RH + HX}\]
Aryl halides are less reactive than alkyl halides in nucleophilic substitution. Due to resonance effect, lone pair on halogen is delocalized into benzene ring.
This gives partial double bond character to C–X bond → bond becomes shorter & stronger.
Strong electron-withdrawing groups (EWGs) like –NO₂ increase reactivity.
EWGs must be at ortho or para positions for effective substitution.
Example reaction:
p-chloronitrobenzene + OH⁻ → p-nitrophenol (Cl replaced by OH).
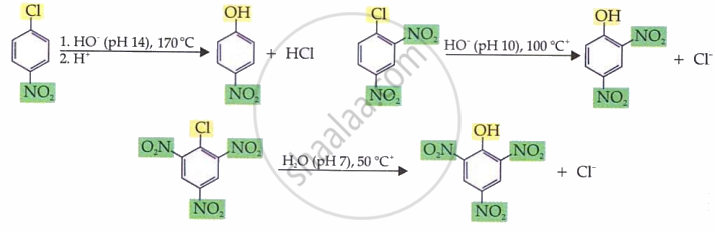
Mechanism is SNAr (Addition–Elimination).
- Step 1: Nucleophile attacks carbon bearing halogen → forms intermediate.
- Step 2: Leaving group (Cl⁻) departs → aromaticity restored.
- Reactivity order:
More –NO₂ groups = higher reactivity
(Tri-NO₂ > Di-NO₂ > Mono-NO₂ > no EWG)
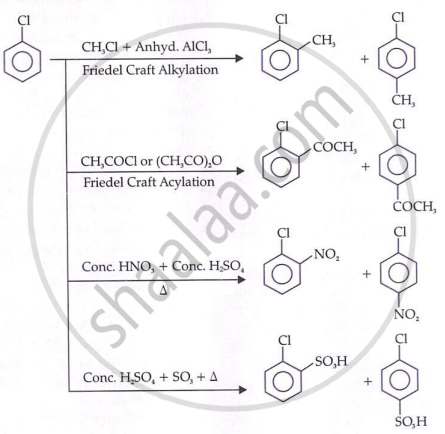
- Haloarenes undergo electrophilic substitution at a slower rate than benzene (halogen is deactivating due to –I effect).
- However, halogen is an ortho/para director (due to +M/resonance effect — lone pair donation to ring at ortho and para positions increases electron density there).
- Chlorine's single electron pair engages in resonance with the ring → electron density rises at ortho and para positions → electrophile attacks there.
Statement:
Alkyl chlorides or bromides react with metallic fluorides (AgF, Hg₂F₂, CoF₂) to form alkyl fluorides.
Equation:
R–Cl + AgF → R–F + AgCl
Importance:
Used for preparation of alkyl fluorides.
Statement:
Aryl diazonium salts react with cuprous halide (CuCl/CuBr) to give haloarenes.
Equation:
Ar–N₂⁺Cl⁻ + CuCl → Ar–Cl + N₂
Importance:
Used to introduce halogen into aromatic ring.
- Halogen is more electronegative than carbon.
- Bond becomes polar.
- Carbon carries partial positive charge.
- Reactivity depends on bond strength.
- Generally colourless liquids or gases.
- Boiling point increases with molecular mass.
- Slightly soluble in water.
- Density increases with number of halogen atoms.
A bimolecular nucleophilic substitution reaction in which nucleophile attacks carbon from backside and displaces leaving group in a single step.
Characteristics:
- One-step reaction
- No intermediate
- Transition state formed
- Inversion of configuration
- Rate depends on both reactants
Order:
Methyl > 1° > 2° > 3°
Reason:
Steric hindrance affects reaction.
A unimolecular nucleophilic substitution reaction in which leaving group departs first forming carbocation intermediate.
Characteristics:
- Two-step reaction
- Carbocation intermediate
- Rate depends only on substrate
- Racemisation possible
- Favoured by 3° halides
Order:
3° > 2° > 1°
Reason:
Stability of carbocation.
Statement:
In elimination reactions, the preferred product is the alkene having greater number of alkyl groups attached to the doubly bonded carbon atoms.
Example:
2-Bromopentane → Pent-2-ene (major)
Reason:
More substituted alkene is more stable.
Statement:
Alkyl halides react with sodium metal in dry ether to form higher alkanes.
Reaction:
2R–X + 2Na → R–R + 2NaX
Limitation:
Best for primary alkyl halides.
Statement:
Alkyl halides react with magnesium in dry ether to form Grignard reagents (R–MgX).
Nature:
Highly reactive, reacts with water.
Important:
Reaction must be moisture free.
Statement:
Alkyl chlorides or bromides react with sodium iodide in dry acetone to form alkyl iodides.
Equation:
R–Cl + NaI → R–I + NaCl↓
Reason:
NaCl precipitates in acetone driving the reaction forward.
Statement:
Aryl halide reacts with alkyl halide in presence of sodium in dry ether forming alkyl arene.
Mechanism:
Free radical coupling reaction.
Limitation:
Mixture of products may form.
Important Questions [66]
- Draw the Structure of Major Monohalo Product in Each of the Following Reactions :
- Which of the following belongs to the class of vinyl halides?
- Write the Product(S) in the Following Reactions: O+Hcn
- Write the product(s) in the following reaction: COONa + NaOH ->[CaO][\Delta] ?
- Out of Which is an Example of a Benzylic Halide?
- Complete the Following Reaction: \\Ce{(C6h5ch2)2cd + 2ch3cocl}\
- Write the Structure of 1-bromo-4-chlorobut-2-ene
- Write the Structures of the Main Products in the Following Reactions : - 2
- Write the Product(S) in the Following Reactions: Ch3−Ch=Ch−Cn Dibal−H
- Write the mechanism (using curved arrow notation) of the following reaction:
- Give Reasons : N-butyl Bromide Has Higher Boiling Point than T-butyl Bromide.
- Arrange the following compounds in increasing order of their boiling points: CH3CH2OH, CH3−CHO, CH3−COOH
- P-Dichlorobenzene has higher m.p. and lower solubility than those of o- and m-isomers. Discuss.
- Why is the boiling point of o-dichlorobenzene higher than p-dichlorobenzene, but the melting point of para-isomer is higher than ortho-isomer?
- Arrange the following compounds in increasing order of their boiling points. (a) CH3-CH(CH3)-CH2Br (b) CHA3CHA2CHA2CHA2Br
- Arrange the set of compounds in order of increasing boiling points. Bromomethane, Bromoform, Chloromethane, Dibromomethane.
- Why Dextro and Laevorotatory Isomers of Butan-2-ol Are Difficult to Separate by Fractional Distillation?
- Define racemic mixture.
- What happens when ethyl chloride is treated with aqueous KOH?
- C–Cl bond length in chlorobenzene is shorter than C–Cl bond length in CH3–Cl.
- Sn1 Reactions Are Accompanied by Racemization in Optically Active Alkyl Halides.
- Given Reasons: Sn1 Reactions Are Accompanied by Racemization in Optically Active Alkyl Halides.
- Which Would Undergo Sn2 Reaction Faster in the Following Pair and Why ? Ch3 – Ch2 – Br and Ch3 – Ch2 – I
- Give the mechanism of the following reaction: CHA3CHA2OH→413KHA2SOA4CHA3CHA2−O−CHA2CHA3+HA2O
- Nucleophilic Substitution: Nucleophilic substitution reaction of haloalkane can be conducted according to both SN1 and SN2 mechanisms. SN1 is a two-step reaction, while SN2 is a single-step reaction.
- Write the Major Products(S) in the Following
- Which Would Undergo Sn1 Reaction Faster in the Following Pair and Why?
- Answer the Following Question. Write One Stereochemical Difference Between Sn1 and Sn2 Reactions.
- Racemisation occurs in ______.
- Arrange the following compounds in increasing order of reactivity towards SN2 reaction. 2-Bromopentane, 1-Bromopentane, 2-Bromo-2-methylbutane
- Retention of configuration is observed in ______.
- Convert bromoethane to propanamine.
- Complete the reaction with the main product formed: N2+Cl- ->[CH3CH2OH]
- Inversion of configuration occurs in ______.
- How do you convert the following: Ethanol to propanenitrile
- Write the main products when methyl chloride is treated with AgCN.
- Give reasons for the following: (CH3)3C–O–CH3 on reaction with HI gives (CH3)3C–I and CH3–OH as the main products and not (CH3)3C–OH and CH3–I.
- which is more reactive towards SN1 reaction and why?
- Given reasons: C–Cl bond length in chlorobenzene is shorter than C–Cl bond length in CH3–Cl.
- Which Would Undergo SN2 Reaction Faster in the Following Pair and Why?
- Write the Structure of an Isomer of Compound C4h9br Which is Most Reactive Towards Sn1 Reaction
- Write the Structure of the Major Product in Each of the Following Reaction
- Write the Structures of A, B and C in the Following
- In the following pair of halogen compounds, which compound undergo a faster SN1 reaction? Cl and Cl
- What are ambident nucleophiles? Explain with an example.
- Arrange the compounds of the following set in order of reactivity towards SN2 displacement: 2-Bromo-2-methylbutane, 1-Bromopentane, 2-Bromopentane
- Assertion (A) : Nucleophilic substitution of iodoethane is easier than chloroethane. Reason (R) : Bond enthalpy of C-I bond is less than that of C-Cl bond.
- How do you convert: 2-bromobutane to but-2-ene
- Write the main products when n-butyl chloride is treated with alcoholic KOH.
- The conversion of an alkyl halide into an alkene by alcoholic KOH is classified as ______.
- Write the Structure of Main Compounds a and B in the Following Reaction:
- Write Chemical Equation in Support of Your Answer. Out of Cl and Ch2- Cl, Which One is More Reactive Towards Nucleophilic Substitution Reaction and Why?
- Chlorobenzene is extremely less reactive towards a nucleophilic substitution reaction. Give two reasons for the same.
- Write the final product(s) in each of the following reactions
- The presence of nitro group (−NO2) at o/p positions increases the reactivity of haloarenes towards nucleophilic substitution reactions.
- Give reasons: The dipole moment of chlorobenzene is lower than that of cyclohexyl chloride.
- Write the Product Formed on Reaction of D-glucose with Br2 Water.
- What happens when CHA3−Br is treated with KCN?
- Out of (Ch3)3 C-br and (Ch3)3 C-i, Which One is More Reactive Towards Sn1 and Why?
- Assertion: Chlorobenzene is resistant to nucleophilic substitution reaction at room temperature. Reason (R): C–Cl bond gets weaker due, to resonance.
- Why haloarenes are not reactive towards nucleophilic substitution reaction? Give two reactions.
- Assertion (A): Chlorobenzene is resistant to electrophilic substitution reaction. Reason (R): C-Cl bond in chlorobenzene acquires partial double bond characters due to resonance.
- How Would You Account the Following : Transition Metals and Their Compounds Show Catalytic Properties.
- Write the Major Products(S) in the Following
- Write the equation for the following: Oxidation of chloroform by air and light.
- Auto-oxidation of chloroform in air and sunlight produces a poisonous gas known as ______.
Concepts [15]
- Concept of Haloalkanes and Haloarenes
- Classification of Haloalkanes and Haloarenes
- Nomenclature of Haloalkanes and Haloarenes
- Nature of C-X Bond of Haloalkanes and Haloarenes
- Methods of Preparation of Haloalkanes
- Methods of Preparation of Haloarenes
- Physical Properties of Haloalkanes and Haloarenes
- Reactions of Haloalkanes - Nucleophilic Substitution Reactions
- Reactions of Haloalkanes - Elimination Reactions
- Reactions of Haloalkanes - Reaction with Metals
- Reactions of Haloarenes - Nucleophilic Substitution
- Reactions of Haloarenes - Electrophilic Substitution Reactions
- Reactions of Haloarenes - Reaction with Metals
- Polyhalogen Compounds
- Overview of Haloalkanes and Haloarenes
