Advertisements
Advertisements
Question
Auto-oxidation of chloroform in air and sunlight produces a poisonous gas known as ______.
Options
Tear gas
Mustard gas
Phosgene gas
Chlorine gas
Phosphine
Advertisements
Solution
Auto-oxidation of chloroform in air and sunlight produces a poisonous gas known as phosgene gas.
Explanation:
When chloroform \[\ce{(CHCl3)}\] combines with air and sunshine, it creates Phosgene gas \[\ce{(COCl2)}\] and hydrochloric acid \[\ce{(HCl)}\].
\[\ce{2CHCl3 + \underset{(Air)}{O2} ->[Light]\underset{(poisonous gas)}{\underset{Phosgene}{2COCl2}} + 2 HCl}\]
APPEARS IN
RELATED QUESTIONS
Draw structure of DDT.
Among the following, insecticide is:
Diphenyls are potential threat to the environment. How are these produced from arylhalides?
How can you obtain iodoethane from ethanol when no other iodine-containing reagent except NaI is available in the laboratory?
Some halogen containing compounds are useful in daily life. Some compounds of this class are responsible for exposure of flora and fauna to more and more of UV light which causes destruction to a great extent. Name the class of these halocompounds. In your opinion, what should be done to minimise harmful effects of these compounds.
The most stable conformation of 1, 2-dibromomethane among the following is
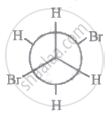 |
| I |
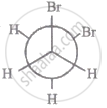 |
| II |
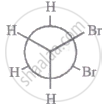 |
| III |
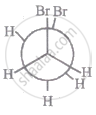 |
| IV |
CH3NH2 react with CH3mg x to give
Give the IUPAC name of the following compound:
\[\ce{(CCl3)3CCl}\]
Write names of two polyhalogen compounds.
Convert the following iodomethane to methoxy methane.
