Advertisements
Advertisements
Question
How can you obtain iodoethane from ethanol when no other iodine-containing reagent except NaI is available in the laboratory?
Advertisements
Solution
Ethanol is converted to chloroethane and Cl can be replaced by I.
\[\ce{C2H5OH + HCl ->[ZnCl2] C2H5Cl ->[NaI] C2H5I}\]
APPEARS IN
RELATED QUESTIONS
Give balanced equations for the following reactions:
Acetic anhydride with phosphorus pentachloride.
In which of the following conversions, phosphorus pentachloride is used as the reagent?
Among the following, insecticide is:
Chloropicrin is formed by the reaction of ____________.
A dihalogen derivative ‘X’ of a hydrocarbon with three carbon atoms react with ale. KOH and produces hydrocarbon which forms red ppt. with ammonical Cu2Cl2. ‘X’ gives an aldehyde on reaction with aq. KOH. The compound ‘X’ is:
Major product obtained on reaction of 3-Phenyl propene with HBr in presence of organic peroxide.
Match the compounds given in Column I with the effects given in Column II.
| Column I | Column II | |
| (i) | Chloramphenicol | (a) Malaria |
| (ii) | Thyroxine | (b) Anaesthetic |
| (iii) | Chloroquine | (c) Typhoid fever |
| (iv) | Chloroform | (d) Goiter |
| (e) Blood substituent |
Assertion: It is difficult to replace chlorine by –OH in chlorobenzene in comparison to that in chloroethane.
Reason: Chlorine-carbon \[\ce{(C - Cl)}\] bond in chlorobenzene has a partial double bond character due to resonance.
The most stable conformation of 1, 2-dibromomethane among the following is
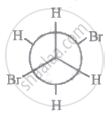 |
| I |
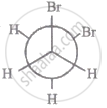 |
| II |
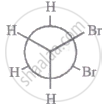 |
| III |
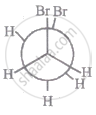 |
| IV |
Chloroform on reduction with Zn an HCI (all) gives:-
Westrosol is:-
In the above reaction if we take methylene chloride and isopropylidene chloride then products are ______.
Which among the following pairs has only herbicides?
Give the IUPAC name of the following compound:
\[\ce{(CCl3)3CCl}\]
Give the IUPAC names of the following componus:
\[\ce{CICH2C =CCH2Br}\]
Convert the following iodomethane to methoxy methane.
Give the uses of iodoform.
Methyl bromide is not used ______.
Iodoform can be prepared from all except:
