Advertisements
Advertisements
Question
Write the equation for the following:
Oxidation of chloroform by air and light.
Advertisements
Solution
In the presence of light, chloroform is slowly oxidised by air to carbonyl chloride, also known as phosgene.
\[\ce{\underset{Chloroform}{CHCl3} + 1/2O2 ->[Light] \underset{Phosgene}{COCl2} + HCl}\]
APPEARS IN
RELATED QUESTIONS
Give the uses of freon 12.
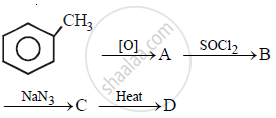
In the following sequence of the reactions, what is D?
Phosgene is a common name for ____________.
What are the IUPAC names of the insecticide DDT and benzene hexachloride? Why is their use banned in India and other countries?
How can you obtain iodoethane from ethanol when no other iodine-containing reagent except NaI is available in the laboratory?
The most stable conformation of 1, 2-dibromomethane among the following is
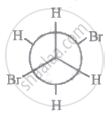 |
| I |
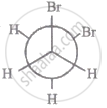 |
| II |
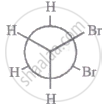 |
| III |
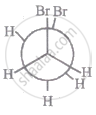 |
| IV |
Which among the following pairs has only herbicides?
Give the uses of DDT.
Give the uses of iodoform.
Strong reducing agent converts CHCl3 into:
