Advertisements
Advertisements
प्रश्न
How can you obtain iodoethane from ethanol when no other iodine-containing reagent except NaI is available in the laboratory?
Advertisements
उत्तर
Ethanol is converted to chloroethane and Cl can be replaced by I.
\[\ce{C2H5OH + HCl ->[ZnCl2] C2H5Cl ->[NaI] C2H5I}\]
APPEARS IN
संबंधित प्रश्न
Give balanced equations for the following reaction:
Acetaldehyde with hydroxylamine.
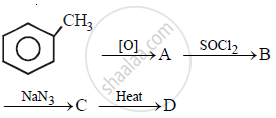
In the following sequence of the reactions, what is D?
Phosgene is a common name for ____________.
B.H.C. is used as an ____________.
Gammexane is ____________.
\[\ce{CH3CH2CH2Br + NaCN -> CH3CH2CH2CN + NaBr}\], will be fastest in:
A dihalogen derivative ‘X’ of a hydrocarbon with three carbon atoms react with ale. KOH and produces hydrocarbon which forms red ppt. with ammonical Cu2Cl2. ‘X’ gives an aldehyde on reaction with aq. KOH. The compound ‘X’ is:
Major product obtained on reaction of 3-Phenyl propene with HBr in presence of organic peroxide.
Name the alkene which will yield 1-chloro-1-methylcyclohexane by its reaction with \[\ce{HCl}\]. Write the reactions involved.
Diphenyls are potential threat to the environment. How are these produced from arylhalides?
How will you obtain monobromobenzene from aniline?
Ethyl chloride is converted into diethyl ether by
Westrosol is:-
Auto-oxidation of chloroform in air and sunlight produces a poisonous gas known as ______.
Write names of two polyhalogen compounds.
Give the uses of DDT.
Give the uses of carbon tetrachloride.
DDT is obtained by the reaction of chlorobenzene with ______.
Methyl bromide is not used ______.
