Advertisements
Advertisements
प्रश्न
Auto-oxidation of chloroform in air and sunlight produces a poisonous gas known as ______.
पर्याय
Tear gas
Mustard gas
Phosgene gas
Chlorine gas
Phosphine
Advertisements
उत्तर
Auto-oxidation of chloroform in air and sunlight produces a poisonous gas known as phosgene gas.
Explanation:
When chloroform \[\ce{(CHCl3)}\] combines with air and sunshine, it creates Phosgene gas \[\ce{(COCl2)}\] and hydrochloric acid \[\ce{(HCl)}\].
\[\ce{2CHCl3 + \underset{(Air)}{O2} ->[Light]\underset{(poisonous gas)}{\underset{Phosgene}{2COCl2}} + 2 HCl}\]
APPEARS IN
संबंधित प्रश्न
Write the environmental effects of DDT.
Give balanced equations for the following reactions:
Acetic anhydride with phosphorus pentachloride.
Write balanced chemical equation for the following reaction:
Methyl magnesium iodide is treated with carbon dioxide and the product hydrolysed in acidic medium.
Diphenyls are potential threat to the environment. How are these produced from arylhalides?
How can you obtain iodoethane from ethanol when no other iodine-containing reagent except NaI is available in the laboratory?
Assertion: It is difficult to replace chlorine by –OH in chlorobenzene in comparison to that in chloroethane.
Reason: Chlorine-carbon \[\ce{(C - Cl)}\] bond in chlorobenzene has a partial double bond character due to resonance.
The most stable conformation of 1, 2-dibromomethane among the following is
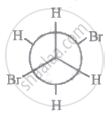 |
| I |
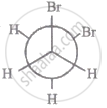 |
| II |
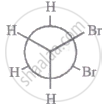 |
| III |
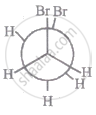 |
| IV |
Sodium ethoxide react with ethyl iodide to yield.
Methyl bromide is not used ______.
Iodoform can be prepared from all except:
