Advertisements
Advertisements
प्रश्न
Diphenyls are potential threat to the environment. How are these produced from arylhalides?
Advertisements
उत्तर
Diphenyl such as p,p ‘-dichlorodiphenyl trichloroethane (DDT) are posing serious environment problems because of its chemical stability and its fat solubility. Its residue accumulates in environment and its long-term effects could be highly dangerous.
Diphenyl can be prepared from aryl halides by the following two methods:
(i) Fittig reaction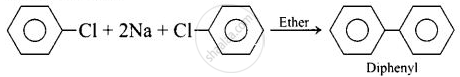
(ii) Ullmann biaryl synthesis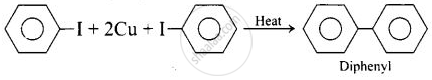
APPEARS IN
संबंधित प्रश्न
How can the following conversions be brought about:
Methyl chloride to acetone.
Give balanced equations for the following reactions:
Acetic anhydride with phosphorus pentachloride.
Write the formula of the following compound:
Triammine triaquachromium (III) chloride
Chloroform and conc. HNO3 reacts to produce ____________.
Fitting reaction can be used to prepare ____________.
Major product obtained on reaction of 3-Phenyl propene with HBr in presence of organic peroxide.
Why iodoform has appreciable antiseptic property?
What are the IUPAC names of the insecticide DDT and benzene hexachloride? Why is their use banned in India and other countries?
How will you obtain monobromobenzene from aniline?
Convert the following:
Chloroethane to methanamine.
The most stable conformation of 1, 2-dibromomethane among the following is
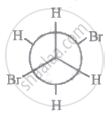 |
| I |
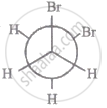 |
| II |
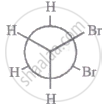 |
| III |
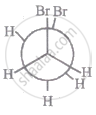 |
| IV |
The shape of methyl carbanion is similar to that of–
Westrosol is:-
The compound that will not give iodoform on treatment with alkali and iodine is ______.
Auto-oxidation of chloroform in air and sunlight produces a poisonous gas known as ______.
Give the uses of carbon tetrachloride.
Strong reducing agent converts CHCl3 into:
Assertion (A): CHCI3 is stored in dark bottles. Reason (R): CHCl3 is oxidised in dark.
In the light of above statements, choose the most appropriate answer from the options given below
