Advertisements
Advertisements
Question
Diphenyls are potential threat to the environment. How are these produced from arylhalides?
Advertisements
Solution
Diphenyl such as p,p ‘-dichlorodiphenyl trichloroethane (DDT) are posing serious environment problems because of its chemical stability and its fat solubility. Its residue accumulates in environment and its long-term effects could be highly dangerous.
Diphenyl can be prepared from aryl halides by the following two methods:
(i) Fittig reaction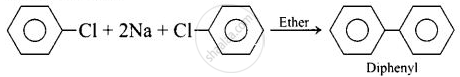
(ii) Ullmann biaryl synthesis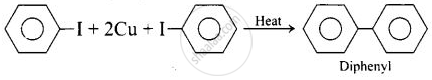
APPEARS IN
RELATED QUESTIONS
How the following conversion can be carried out?
Aniline to phenylisocyanide
Give balanced equations for the following reactions:
Acetic anhydride with phosphorus pentachloride.
Write balanced chemical equation for the following reaction:
Methyl magnesium iodide is treated with carbon dioxide and the product hydrolysed in acidic medium.
Write the formula of the following compound:
Triammine triaquachromium (III) chloride
Write the formula of the following compound:
Potassiumhexacyanoferrate(III)
Phosgene is a common name for ____________.
Chloroform and conc. HNO3 reacts to produce ____________.
Chloromethane on treatment with excess of ammonia yields mainly ______.
Name the alkene which will yield 1-chloro-1-methylcyclohexane by its reaction with \[\ce{HCl}\]. Write the reactions involved.
The most stable conformation of 1, 2-dibromomethane among the following is
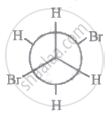 |
| I |
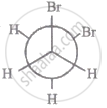 |
| II |
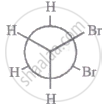 |
| III |
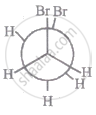 |
| IV |
Westrosol is:-
In the above reaction if we take methylene chloride and isopropylidene chloride then products are ______.
The compound that will not give iodoform on treatment with alkali and iodine is ______.
Phenyl magnesium bromide reacts with methanol to give ______.
Auto-oxidation of chloroform in air and sunlight produces a poisonous gas known as ______.
State the uses of iodoform (CHI3)
Write two uses of polyhalogen compounds.
Assertion (A): CHCI3 is stored in dark bottles. Reason (R): CHCl3 is oxidised in dark.
In the light of above statements, choose the most appropriate answer from the options given below
Which of these compounds is synthesised by chloral?
