Advertisements
Advertisements
Question
Write the main products when methyl chloride is treated with AgCN.
Advertisements
Solution
When methyl chloride is treated with AgCN, methyl cyanide is formed.

APPEARS IN
RELATED QUESTIONS
Out of  , which is more reactive towards SN1 reaction and why?
, which is more reactive towards SN1 reaction and why?
Write the structure of the major product in each of the following reaction :

SN1 reaction of alkyl halides lead to ___________.
The correct order of increasing the reactivity of C–X bond towards nucleophile in following compounds.

(I)

(II)
(CH3)3CCl
(III)
(CH3)2CHCl
(IV)
Which of the following compound will undergo racemisation when reacts with aq. KOH?
(i)
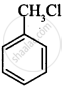
(ii)
CH3CH2CH2Cl
(iii)
\[\begin{array}{cc}
\ce{CH3}\phantom{..}\\
|\phantom{....}\\
\ce{CH3-CH-CH2Cl}
\end{array}\]
(iv)
\[\begin{array}{cc}
\phantom{..}\ce{H}\\
\phantom{..}|\\
\ce{CH3-C-Cl}\\
\phantom{..}|\\
\phantom{.....}\ce{C2H5}
\end{array}\]
In which reaction mechanism carbocation is formed?
The number of chiral carbons present in the molecule given below is ______.

Complete the reaction with the main product formed:

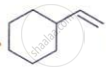 HCI, Major product ______.
HCI, Major product ______.
The compound that will undergo SN1 reaction with the fastest rate is:
