Advertisements
Advertisements
Question
Why is the boiling point of o-dichlorobenzene higher than p-dichlorobenzene, but the melting point of para-isomer is higher than ortho-isomer?
Advertisements
Solution
Because o-dichlorobenzene has a greater dipole moment and a greater dipole-dipole interaction than p-dichlorobenzene, it has a higher boiling point.
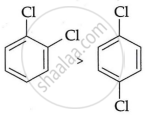
Boiling point
As opposed to ortho- and meta-isomer, p-dichlorobenzene has a higher melting point than that of o- and m-isomer due to its greater symmetry, which suits the crystal better.
APPEARS IN
RELATED QUESTIONS
For the same alkyl group, an alkyl bromide has a higher boiling point than alkyl fluoride because:
How many structural isomers are possible for a compound with the molecular formula C3H7Cl?
Which of the following halide is 2°?
Mg reacts with RBr best in ____________.
Arrange the following compounds in the increasing order of their densities.
(a)

(b)

(c)
(d)

Arrange the following compounds in increasing order of their boiling points.
(a) \[\begin{array}{cc}
\ce{CH3}\phantom{..................}\\
\backslash\phantom{...........}\\
\ce{CH - CH - CH2Br}\\
/\phantom{.............}\\
\ce{CH3}\phantom{..................}
\end{array}\]
(b) \[\ce{CH3CH2CH2CH2Br}\]
(c) \[\begin{array}{cc}
\phantom{...}\ce{CH3}\\
\phantom{}|\\
\ce{H3C - C - CH3}\\
\phantom{}|\\
\phantom{..}\ce{Br}
\end{array}\]
Which of the following compounds will have the highest melting point and why?
| (I) |  |
|
(II) |
 |
| (III) | 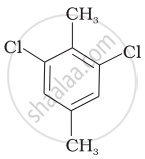 |
Arrange the isomeric dichlorobenzene in the increasing order of their boiling point and melting points.
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
