Definitions [34]
Coordination compounds are compounds in which a central metal atom or ion is linked to a number of ions or neutral molecules by coordinate bonds — i.e., by donation of lone pair of electrons by these ions or neutral molecules (called ligands) to the central metal atom.
Coordination compounds are those molecular compounds which retain their identity in solid as well as in aqueous solution. In these compounds, metals or atoms are bonded to a number of anions or neutral molecules by a coordinate bond.
Optical isomers are mirror images that cannot be superimposed on one another. These are called enantiomers.
Isomerism is the phenomenon in which compounds have the same molecular formula but differ in their physical or chemical properties due to a different arrangement of atoms or groups in space or structure.
Define the term Hydrated isomers.
Isomers in which there is exchange of solvent (water) ligands between coordination and ionization spheres are called hydrate isomers.
Define Distereoisomers.
Two or more coordination compounds which contain the same number and types of atoms, and bonds (i.e., the connectivity between atoms is the same), but which have different spatial arrangements of the atoms are called distereoisomers.
Define the term Co-ordination isomer.
Isomers which show interchange of ligands between cationic and anionic spheres of different metal ions are called co-ordination isomers.
In a disubstituted complex molecule/ion, when two same ligands are at right angles (90°), the geometrical isomer is known as a cis-isomer.
When two ligands are in opposite directions, i.e., at 180° to each other, the isomer formed is the trans-isomer.
Define the following term:
Anomers
Anomers are stereoisomers of sugars that differ only in the configuration of the hydroxyl group at the C1 position. They are not mirror images of each other.
The arrangement of ligands in order of increasing field strength is called spectrochemical series.
Ligands that produce large crystal field splitting and form low spin complexes are called strong field ligands.
A complex in which electrons occupy higher energy orbitals before pairing due to small Δ₀ is called high spin complex.
A complex in which electrons pair in lower energy orbitals due to large Δ₀ is called low spin complex.
A compound containing carbon monoxide ligand bonded to a transition metal is called metal carbonyl.
The mutual strengthening of σ-donation and π-back bonding between metal and ligand is called synergic bonding.
The spatial arrangement of ligands directly bonded to the central metal atom defines a geometry which is called coordination polyhedron.
A complex in which the central metal atom is bonded to only one kind of ligand is called homoleptic complex.
Ligands that produce small crystal field splitting and form high spin complexes are called weak field ligands.
An ion or molecule which donates one or more pairs of electrons to the central metal atom/ion is called ligand.
The number of donor atoms directly bonded to the central metal atom/ion in a complex is called coordination number.
A compound in which a central metal atom or ion is bonded to a fixed number of ions or molecules through coordinate bonds is called coordination compound.
The central metal atom/ion together with the ligands attached to it enclosed in square brackets is called coordination entity.
The atom or ion to which a fixed number of ligands are bonded in a definite geometrical arrangement is called central atom or ion.
The central metal atom/ion along with the ligands attached to it and enclosed in square brackets is called coordination sphere.
The isomerism arising due to difference in the number of solvent molecules inside and outside the coordination sphere is called solvate isomerism.
A complex in which the central metal atom is bonded to more than one kind of ligand is called heteroleptic complex.
The isomerism arising due to different spatial arrangement of ligands around the central metal atom is called geometrical isomerism.
The isomerism in which complexes are non-superimposable mirror images of each other is called optical isomerism.
The isomerism arising due to different modes of attachment of an ambidentate ligand is called linkage isomerism.
The isomerism arising due to exchange between a ligand inside the coordination sphere and an ion outside it is called ionisation isomerism.
A complex in which inner (n−1)d orbitals participate in hybridisation is called inner orbital complex.
A complex in which outer nd orbitals participate in hybridisation is called outer orbital complex.
The splitting of degenerate d-orbitals in the presence of ligands due to electrostatic interactions is called crystal field splitting.
Theorems and Laws [10]
Alfred Werner (1893) proposed the first systematic theory to explain the structure and bonding in coordination compounds. His key postulates are:
Main Postulates:
- In coordination compounds, metals show two types of valencies — Primary (ionisable) and Secondary (non-ionisable).
- Primary valences are normally ionisable. They are satisfied by negative ions (counter ions/anions). They correspond to the metal's oxidation state.
- Secondary valences are non-ionisable. They are satisfied by neutral molecules or negative ions (ligands). The secondary valency is equal to the coordination number and is constant for a metal.
- Ion groups bound by secondary valencies to the metal have a characteristic spatial arrangement (geometry). This geometry is decided by the secondary valences, not the primary valences.
Werner's Formula Examples:
- CrCl₃·6H₂O: In [Cr(H₂O)₆]Cl₃, all three Cl⁻ are outside the coordination sphere and hence ionisable.
- CrCl₃·5H₂O: [Cr(H₂O)₅Cl]Cl₂ — two Cl⁻ are ionisable, one is inside the coordination sphere.
- CrCl₃·4H₂O: [Cr(H₂O)₄Cl₂]Cl — one Cl⁻ ionisable.
- CrCl₃·3H₂O: [Cr(H₂O)₃Cl₃] — no ionisable Cl⁻, no precipitate with AgNO₃.
- Proposed by Heitler and London (1927), further developed by Pauling and Slater.
- A covalent bond is formed when half-filled valence atomic orbitals of similar energies overlap, each containing one unpaired electron.
- Greater the overlap → stronger the bond.
Types of Orbital Overlap:
| Type | Description | Bond Formed |
|---|---|---|
| Axial (Head-on) overlap | Orbitals overlap along the internuclear axis | Sigma (σ) bond |
| Sidewise (Lateral) overlap | Orbitals overlap parallel to each other, perpendicular to the internuclear axis | Pi (π) bond |
Hybridisation & Shapes:
| Hybridisation | Shape | Coordination No. |
|---|---|---|
| sp³ | Tetrahedral | 4 |
| dsp² | Square planar | 4 |
| sp³d | Trigonal bipyramidal | 5 |
| d²sp³ | Octahedral (inner) | 6 |
| sp³d² | Octahedral (outer) | 6 |
Limitations of VBT:
- Involves a number of assumptions.
- Does not give a quantitative interpretation of magnetic data.
- Does not explain the colour exhibited by coordination compounds.
- Does not give a quantitative interpretation of the thermodynamic or kinetic stabilities of coordination compounds.
- Does not make exact predictions regarding the tetrahedral and square planar structures of 4-coordinate complexes.
- Does not distinguish between weak and strong ligands.
Statement:
Coordination compounds are named by following additive nomenclature rules recommended by IUPAC.
Important Rules:
- Cation named first.
- Ligands named alphabetically before metal.
- Prefixes: di, tri, tetra, etc.
- Oxidation state written in Roman numerals.
- Metal name ends with “-ate” in anionic complexes.
Statement:
In an octahedral field, the five degenerate d-orbitals split into two sets of different energies due to electrostatic repulsion between ligand electrons and metal d-electrons.
Explanation:
-
dx2−y2,dz2 form eg (higher energy)
-
dxy,dxz,dyz form t2g (lower energy)
-
Energy difference = Δ₀
-
Splitting depends on ligand strength
Statement:
The electronic configuration of a complex depends on the relative magnitude of crystal field splitting energy (Δ₀) and pairing energy (P).
Explanation:
-
If Δ₀ < P → High spin complex
-
If Δ₀ > P → Low spin complex
-
Strong field ligands → Low spin
-
Weak field ligands → High spin
Statement:
The colour of coordination compounds arises due to d–d electronic transitions between split d-orbitals in presence of ligands.
Explanation:
-
Absorption of specific wavelength of visible light
-
Complementary colour observed
-
Greater Δ₀ → higher energy light absorbed
-
d⁰ and d¹⁰ complexes are colourless
Statement:
In metal carbonyls, bonding involves both σ-donation from CO to metal and π-back donation from metal to CO, strengthening the bond.
Explanation:
-
CO donates lone pair to metal (σ bond)
-
Metal donates electron density into π* orbital of CO
-
Back bonding strengthens M–C bond
-
Weakens C–O bond
Statement:
Coordination compounds exhibit isomerism due to different arrangement of ligands either in space or within the coordination sphere.
Types:
- Structural isomerism
- Stereoisomerism
Explanation:
Structural isomers differ in bonding, while stereoisomers differ in spatial arrangement.
Statement:
According to Valence Bond Theory, metal ions undergo hybridisation of atomic orbitals to form equivalent hybrid orbitals which overlap with ligand orbitals to form coordinate bonds.
Explanation:
-
Explains geometry (tetrahedral, square planar, octahedral)
-
Predicts magnetic behaviour
-
Inner orbital and outer orbital complexes possible
Statement:
Werner proposed that metals in coordination compounds exhibit two types of valencies — primary and secondary valencies.
Explanation:
- Primary valency corresponds to oxidation state.
- Secondary valency corresponds to coordination number.
- Primary valencies are ionisable.
- Secondary valencies are non-ionisable and have definite geometry.
- Secondary valencies are directed in space.
Key Points
Ligands:
Ligands are the donor atoms, molecules, or anions that donate a pair of electrons to the metal atom or ion and form a coordinate bond. The number of coordinating or ligating groups present in a ligand is called the denticity of that ligand.
| Type | Description | Examples |
|---|---|---|
| (i) Unidentate / Monodentate | Binds through one donor atom | Cl⁻, H₂O, NH₃, NO |
| (ii) Didentate / Bidentate | Binds through two donor atoms | en (ethane-1,2-diamine), C₂O₄²⁻ (oxalate) |
| (iii) Polydentate | Several donor atoms in a single ligand | N(CH₂CH₂NH₂)₃ |
| (iv) Ambidentate | Two different donor atoms, either of which can coordinate | NO₂⁻ (through N or O), SCN⁻ (through S or N), NCS⁻ |
| (v) Chelating | Forms a ring structure with the central atom; polydentate chelate complexes are more stable than monodentate analogues | EDTA (hexadentate), en (bidentate), C₂O₄²⁻ |
NH₄⁺ is NOT a ligand — the N atom in NH₄⁺ has no lone pair of electrons to donate (lone pair has been donated to H⁺). Thiosulphato (S₂O₃²⁻) is an ambidentate ligand, NOT a chelating ligand.
Coordination Number:
The coordination number (CN) of a metal ion in a complex is the total number of unidentate ligands (plus double the number of didentate ligands if any) attached to the central metal ion through coordinate bonds.
Homoleptic vs Heteroleptic Complexes:
- Homoleptic complexes: Metal is bound to only one kind of donor group. e.g., [Co(NH₃)₆]³⁺
- Heteroleptic complexes: Metal is bound to more than one kind of donor group. e.g., [Co(NH₃)₄Cl₂]⁺
Rules for Writing Formulae:
- The cation is written first, then the anion.
- In the formula of the complex ion/entity, the central metal atom is written first, then the ligands in alphabetical order.
- The formula of the entire coordination entity is enclosed in square brackets.
Rules for Naming:
Rule 1: Names of neutral coordination complexes are given without spaces. Cation is named first, separated by a space from the anion.
Rule 2 (Naming ligands first):
- Ligands that act as anions end in –o: Cl⁻ = chlorido, Br⁻ = bromido, I⁻ = iodido
- Anions ending in –ite and –ate are replaced with –ito and –ato: SO₄²⁻ = sulphato, CO₃²⁻ = carbonato, NO₂⁻ = nitrito, CH₃COO⁻ = acetato
- Neutral ligands get the same name as the uncoordinated molecule (with spaces omitted): C₅H₅N = pyridine, (CH₃)₂SO = dimethylsulfoxide (DMSO)
Exceptions — neutral ligands with special names:
| Molecule | Special Name |
|---|---|
| H₂O | aqua |
| NH₃ | ammine |
| CS | thiocarbonyl |
| CO | carbonyl |
| NO | nitrosyl |
Rule 3 (Prefixes): Greek prefixes (di, tri, tetra) are used for simple ligand names. For polydentate ligands (i.e., those with a binding site name containing di/tri already): bis-, tris-, tetrakis-, pentakis-, hexakis- are used instead. e.g., bis(ethane-1,2-diamine) not "diethylenediamine".
Rule 4: Oxidation state of the metal is indicated by a Roman numeral in parentheses after the metal name. NO = nitrosyl.
Rule 5 (Complex ion is a cation): Metal is named same as the element. e.g., Co in a cationic complex = cobalt. Name = Ligands + Metal name (with OS)
Rule 6 (Complex ion is an anion): Metal name ends in –ate + oxidation number.
Anionic Complex Metal Names:
| Metal | Name in Anionic Complex |
|---|---|
| Iron | Ferrate |
| Lead | Plumbate |
| Gold | Aurate |
| Chromium (Cr) | Chromate |
| Palladium (Pd) | Palladinate |
| Mercury (Hg) | Mercurate |
| Zinc (Zn) | Zincate |
| Nickel (Ni) | Nickelate |
| Copper | Cuprate |
| Silver | Argentate |
| Tin | Stannate |
| Cobalt (Co) | Cobaltate |
| Platinum (Pt) | Platinate |
| Cadmium (Cd) | Cadmate |
| Aluminium (Al) | Aluminate |
IUPAC Name Examples
- Na₂[Fe(CN)₅NO]: Sodium pentacyanonitrosatoferrate(II) (Note: pentacyanonitrosylferrate(II))
- [Fe(CN)₆]³⁻: hexacyanidoferrate(III) ion
- [Pt(NH₃)₂(Br)(NO₂)Cl]Cl: triamminebromochloronitroplatinum(IV) chloride
- K₃[Cr(C₂O₄)₃]: potassium trioxalatochromate(III)
Order of naming ions: Positive ion (cation) first, then negative ion (anion). In naming the complex ion, ligands first (alphabetically), then metal.
1. Order
- Ligand → Metal
- Cation first, then anion
2. Ligand Names
- Cl⁻ → chloro
- CN⁻ → cyano
- OH⁻ → hydroxo
- NH₃ → ammine
- H₂O → aqua
- CO → carbonyl
3. Number Prefix
- di, tri, tetra, penta, hexa
- Special: bis, tris (if ligand has number)
4. Order of Ligands
-
Alphabetical order
5. Metal Name
- Neutral/cation → normal name
- Anionic complex → ends with “-ate”
Fe → ferrate
Cu → cuprate
Co → cobaltate
6. Oxidation State
-
Write in Roman (II), (III)
7. Important
- Counter ions not named
- Complex always in [ ]
8. Examples
Neutral complexes
- [Co(NO₂)₃(NH₃)₃] → Triamminetrinitrocobalt(III)
- Fe(CO)₅ → Pentacarbonyliron(0)
Cationic complexes
- [Cu(NH₃)₄]²⁺ → Tetraamminecopper(II) ion
- [Fe(H₂O)₅(NCS)]²⁺ → Pentaaquathiocyanatoiron(III) ion
Anionic complexes
- [Ni(CN)₄]²⁻ → Tetracyanonickelate(II) ion
- [Fe(CN)₆]⁴⁻ → Hexacyanoferrate(II) ion
Compounds (Very Important)
- [Co(NH₃)₅Cl]Cl₂ → Pentaamminechlorocobalt(III) chloride
- K₃[Al(C₂O₄)₃] → Potassium trioxalatoaluminate(III)
- Na₃[Co(NO₂)₆] → Sodium hexanitrocobaltate(III)
| Main Type | Subtype | Condition / Description | Key Rule | Example |
|---|---|---|---|---|
| Stereoisomerism | Geometrical (cis–trans) | Different spatial arrangement | cis = 90°, trans = 180° | [Pt(NH₃)₂Cl₂] |
| Optical | Non-superimposable mirror images | No plane of symmetry | [Co(en)₃]³⁺ | |
| Structural Isomerism | Ionisation | Exchange of ions inside/outside coordination sphere | Counter ion acts as ligand | [Co(NH₃)₅SO₄]Br / [Co(NH₃)₅Br]SO₄ |
| Linkage | Ambidentate ligand attaches via different atoms | NO₂⁻, SCN⁻ | [Co(NH₃)₅NO₂]Cl₂ / [Co(NH₃)₅ONO]Cl₂ | |
| Coordination | Ligand exchange between metal complexes | Two metal centers involved | [Co(NH₃)₆][Cr(CN)₆] | |
| Solvate (Hydrate) | Solvent inside vs outside coordination sphere | Crystal water difference | [Cr(H₂O)₆]Cl₃ / [Cr(H₂O)₅Cl]Cl₂·H₂O |
Importance:
- Successfully explains the formation, directional nature, and geometry of covalent bonds
- Explains why bond angle in water (104.5°) is less than tetrahedral angle through lone pair repulsion
- Explains why bond strength increases with greater orbital overlap
Limitations:
- Cannot satisfactorily explain the paramagnetic nature of O₂ (O₂ has 2 unpaired electrons, but VBT shows all electrons paired in a double bond)
- Fails to explain the electronic spectra of molecules
- Cannot account for the equal bond lengths in resonance structures (e.g., benzene)
- Does not explain why some molecules are coloured
These limitations led to the development of Molecular Orbital Theory (MOT).
CFT is an electrostatic model that considers the metal-ligand bond to be ionic, arising purely from electrostatic interactions between the metal ion and the ligand (treated as point charges for anions, or point dipoles for neutral molecules).
CFT considers the effect of ligands on the relative energies of the d-orbitals of the central metal atom/ion.
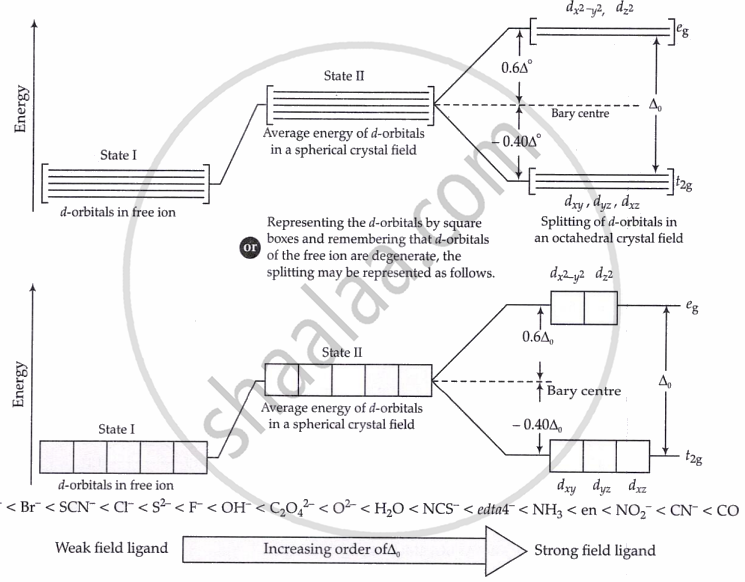
If Δ₀ < P, 4th electron will enter eg giving the configuration \[t_{2g}^3e_{g}^1.\] Ligands for which Δ₀ < P are called weak field ligands.
If Δ₀ > P, pairing will occur in the t2g orbitals and eg orbitals will remain vacant. So, the configuration for 4th e⁻ will be \[t_{2g}^4e_{g}^0.\]. For Δ₀ > P, ligands are strong field ligands.
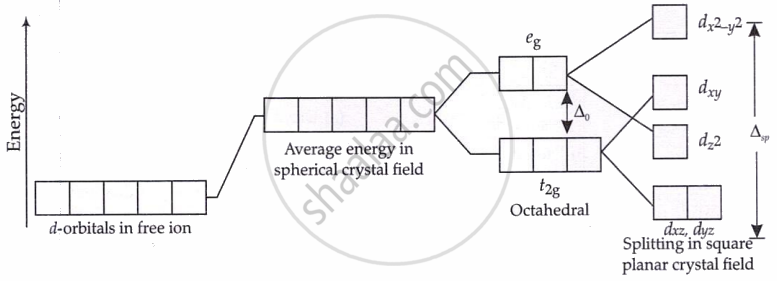
Splitting of d-orbitals in a square planar crystal field:
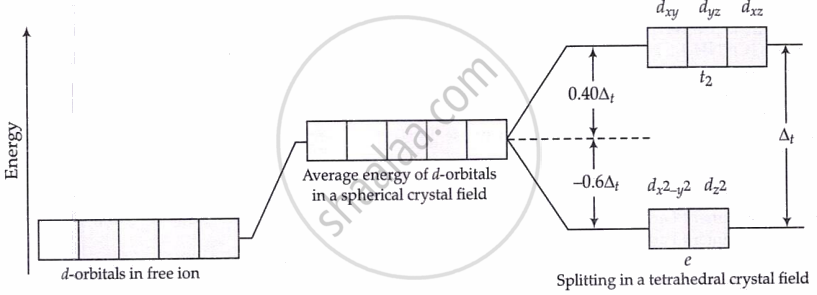
Splitting of d-orbital in a tetrahedral crystal field:
- In coordination compounds, d-orbitals split into t₂g (lower) and eg (higher) energy levels due to the ligand field.
- The energy difference between them is called the crystal field splitting energy (Δ₀).
- This Δ₀ lies in the visible region, so these compounds absorb visible light.
- When light is absorbed, an electron jumps from t₂g → eg, called a d–d transition.
- The observed colour is complementary to the colour of light absorbed.
- The energy relation is: E = hν = Δ₀.
- Metal ions with d¹–d⁹ configuration are coloured, while d⁰ and d¹⁰ are colourless.
- Some compounds (e.g., KMnO₄) show colour due to charge transfer (LMCT), not d–d transition.
- Ligand strength affects colour: strong field ligands ↑ Δ₀, weak ligands ↓ Δ₀.
- Geometry affects splitting: tetrahedral complexes have smaller splitting
Δₜ = 4/9 Δ₀ (e.g., Co²⁺: pink → blue change).
Metal carbonyls are coordination compounds of transition metals with CO as the ligand.
Synergic Bonding (Synergistic Effect):
The M–CO bond in metal carbonyls involves two components acting together (hence "synergic"):
- σ-bond component: CO acts as a σ-donor — the lone pair on C of CO donates into an empty metal orbital → forms a σ bond (M ← C=O).
- π-bond component (back donation): Metal acts as a π-donor — filled metal d-orbitals back-donate electron density into the empty π* (antibonding) orbital of CO → forms a π back-bond (M → C≡O).
This mutual reinforcement of both donation and back-donation strengthens the M–C bond and is called the synergic effect. This also weakens the C≡O bond (back donation into π*) slightly compared to free CO.
The C≡O bond order in metal carbonyls is less than 3 (slightly less than free CO) due to back donation.
Examples:
- Ni(CO)₄ — tetracarbonylnickel(0); d¹⁰; tetrahedral
- Fe(CO)₅ — pentacarbonyliron(0); d⁸; trigonal bipyramidal
- Cr(CO)₆ — hexacarbonylchromium(0); d⁶; octahedral
| No. | Application | Description / Key Point | Example |
|---|---|---|---|
| 1 | Qualitative Analysis | Used for the detection of metal ions | Ni²⁺ + DMG → Ni-DMG (red ppt) |
| 2 | Gravimetric Analysis | Metal ions are estimated by forming stable complexes | Conversion into stable coordination compounds |
| 3 | Volumetric Analysis | EDTA is used as a chelating agent in titrations | Estimation of Ca²⁺, Mg²⁺, Zn²⁺ |
| 4 | Biological Systems | Essential role in living organisms | Haemoglobin (Fe²⁺), Chlorophyll (Mg), Vitamin B₁₂ (Co) |
| 5 | Medicinal Uses | Used in the treatment of diseases | Cis-platin [Pt(NH₃)₂Cl₂] (anti-cancer) |
| 6 | Qualitative Separation | Separation based on the stability of complexes | Cu²⁺ & Cd²⁺ via cyanide complexes |
| 7 | Photography | Dissolution of AgBr using complex formation | [Ag(S₂O₃)₂]³⁻ (hypo solution) |
| 8 | Hydrometallurgy | Extraction of metals using complexes | [Ag(CN)₂]⁻, [Au(CN)₂]⁻ |
| 9 | Electroplating | Provides smooth and uniform coating | Metal deposition using complexes |
Coordination compounds are important due to their roles in:
-
Analytical chemistry (EDTA titrations)
-
Metallurgy (cyanide extraction of gold)
-
Medicine (cis-platin in cancer therapy)
-
Biology (haemoglobin, chlorophyll, vitamin B₁₂)
-
Catalysis (Wilkinson catalyst)
Important Questions [89]
- Write the formula for the following coordination compound: Iron (III) hexacyanoferrate (II)
- Arrange the Following Compound Groups in the Increasing Order of Their Property Indicated : Propanol, Propane, Propanal (Boiling Point)
- Arrange the Following Compound Groups in the Increasing Order of Their Property Indicated : P-nitrophenol, Ethanol, Phenol (Acidic Character)
- Which of the Following is a More Stable Complex and Why ? [ C O ( N H 3 ) 6 ] 3 + a N D [ C O ( E N ) 3 ] 3 +
- What is the secondary valency of cobalt in [Co(en2)Cl2]+?
- Assertion (A): [Co(NH3)5SO4]Cl gives a white precipitate with silver nitrate solution. Reason (R): The complex dissociates to give Cl− and SOA42− ions.
- What is a chelate complex?
- How many ions are produced from the complex Co(NH3)6]Cl2 in solution?
- What are Heteroleptic complexes?
- Why chelate complexes are more stable than complexes with unidentate ligands?
- Assertion (A): EDTA is a hexadentate ligand. Reason (R): EDTA has 2 nitrogen and 4 oxygen donor atoms.
- Which of the following species cannot act as a ligand? Give reason.
- What are Homoleptic complexes?
- What is meant by unidentate ligand?
- What Happens When Pcl5 is Heated?
- Write Structures of Compounds A, B and C in of the Following Reactions - 1
- Write Structures of Compounds A, B and C in of the Following Reactions 2
- Following Compounds Are Given to You : 2-bromopentane, 2-bromo-2-methylbutane, 1-bromopentane Write the Compound Which is Most Reactive Towards Sn2 Reaction.
- Write Structures of Compounds a and B of the Following Reaction :
- Write Iupac Name of the Following Complex [Cr(Nh3)3cl3]
- Write Structures of Compounds a and B of the Following Reaction :
- Write the IUPAC name of the following complex: [Co(NH3)5(CO3)]Cl
- Complete the Following Reactions Nh3+3cl2(Excess) ---->
- Write the Structures of Compounds A, B and C in the Following Reactions - 2
- Write the Structures of Compounds A, B and C in the Following Reactions: -1
- What is meant by a didentate ligand?
- Why Dextro and Laevorotatory Isomers of Butan-2-ol Are Difficult to Separate by Fractional Distillation?
- Oh 1 234 * 3-methyl Butan-2-ol is Optically Active as the 2nd Carbon is Connected to an Hydroxyl Group, Methyl Group, Hydrogen and an Alkyl Group. It Has a Chiral Carbon.
- Give One Chemical Test as an Evidence to Show that Co (Nh3)5cl Are Ionisation Isomers.
- Answer the Following Question. Write Iupac Name of the Complex Pt(En)2ci2. Draw Structures of Geometrical Isomers for this Complex.
- Draw one of the geometrical isomers of the complex [Pt (en)2Cl2] +2 which is optically inactive
- Draw the geometrical isomers of complex [Co(en)A2ClA2]A+.
- Draw one of the geometrical isomers of the complex [Pt(en)2Cl2]2+ which is optically active.
- Draw the geometrical isomers of complex [Pt(NH3)2Cl2].
- What type of isomerism is exhibited by the complex [Co(en)3]3+?
- Define the following term: Anomers
- Why is Butan-1-ol Optically Inactive but Butan-2-ol is Optically Active?
- Draw the geometrical isomers of complex [Pt(en)2Cl2]2+.
- When a coordination compound CoCl3.6NH3 is mixed with AgNO3, 3moles of AgCl are precipitated per mole of the compound. Write the structural formula of the coordination compound.
- Draw the geometrical isomers of [Co(en)2Cl2]2+. Which geometrical isomer of [Co(en)2Cl2]2+ is not optically active and why?
- Which of the following molecules has a chiral centre correctly labelled with an asterisk (*)?
- Assertion (A): Trans [CrCl2(ox)2]3− shows optical isomerism. Reason (R): Optical isomerism is common in octahedral complexes involving didentate ligands.
- Write IUPAC name of the complex [Pt(en)2Cl2]. Draw structures of geometrical isomers for this complex.
- Write the Iupac Name of the Complex [Cr(Nh3)4cl2]+. What Type of Isomerism Does It Exhibit?
- What type of isomerism is shown by the complex [Co(NH3)6] [Cr(CN)6]?
- What Type of Isomerism is Shown by the Complex [Co(En)3]Cl3
- Which isomer of C5H10 gives a single monochloro compound C5H9Cl in bright sunlight?
- The complex [Co(NH3)5(NO2)]Cl2 is red in colour. Give IUPAC the name of its linkage isomer.
- What Type of Isomerism is Shown by the Complex [Co(Nh3)5(Scn)]2+?
- What type of isomerism is exhibited by the complex [Co(NH3)5Cl]SO4?
- Nicl42- is Paramagnetic While Ni(Co)4 is Diamagnetic Though Both Are Tetrahedral. Why? (Atomic No. Ni = 28)
- Write the hybridisation and magnetic behaviour of [CoF6]3−. [Given: Atomic number of Co = 27]
- The magnetic moment of [NiCl4]2− is ______. [Atomic number: Ni = 28]
- [NiCl4]2− is paramagnetic, while [Ni(CO)4] is diamagnetic, though both are tetrahedral. Why? (Atomic number of Ni = 28)
- [Ni(CO)4] has tetrahedral geometry while [Ni(CN)4]2− has square planar, yet both exhibit diamagnetism. Explain.[Atomic number: Ni = 28]
- Write the Hybridisation and Number of Unpaired Electrons in the Complex [Cof_6]^(3-)`. (Atomic No. of Co = 27)
- Using valence bond theory, predict the hybridization and magnetic character of the following: [CoF6]3– [Atomic number of Co = 27]
- For the complex [Fe(H2O)6]+3, write the hybridisation, magnetic character and spin of the complex.
- Write the hybridization and shape of the following complexe : [CoF6]3–
- Why is [Nicl4]2− Paramagnetic While [Ni(Cn)4]2− is Diamagnetic? (Atomic Number of Ni = 28)
- Write the Hybridization and Magnetic Behaviour of the Complex [Ni(Co)4].
- Explain [Co(NH3)6]3+ is an inner orbital complex, whereas [Ni(NH3)6]2+ is an outer orbital complex. [At. No.: Co = 27, Ni = 28]
- Write the hybridization and shape of the following complexe : [Ni(CN)4]2–
- Write the Hybridization Type and Magnetic Behaviour of the Complex [Ni(Cn)4]2−. (Atomic Number of Ni = 28)
- Explain [Fe(CN)6]3− is an inner orbital complex, whereas [FeF6]3− is an outer orbital complex. [Atomic number: Fe = 26]
- Write the Hybridization and Magnetic Character of the Following Complexes: (I) Fe(H2o)62+
- For the complex [Fe(CN)6]3–, write the hybridization type, magnetic character and spin nature of the complex.
- Write the Hybridisation and Magnetic Character of [Co(C2o4)3]3–. (At. No. of Co = 27)
- Write the Hybridization and Magnetic Character of the Following Complexes: Fe(Co)5
- On the basis of crystal field theory, write the electronic configuration for d4 ion if ∆0 < P.
- Write the Electronic Configuration of Fe(Iii) on the Basis of Crystal Field Theory When It Forms an Octahedral Complex in the Presence of (I) Strong Field, and (Ii) Weak Field Ligand. (Atomic
- Why Are Low Spin Tetrahedral Complexes Rarely Observed?
- On the basis of crystal field theory, write the electronic configuration for d4 with a strong field ligand for which Δ0 > P.
- Why are low spin tetrahedral complexes not formed?
- Using crystal field theory, write the electronic configuration of d5 ion, if Δ0 > P.
- What is the spectrochemical series?
- What is the difference between a weak field ligand and a strong field ligand?
- What is crystal field splitting energy?
- On the basis of Crystal Field Theory, write the electronic configuration of d4 ion if Δ0 > P
- On the basis of Crystal Field theory, write the electronic configuration for the d5 ion with a strong field ligand for which Δ0 > P.
- On the basis of crystal field theory, write the electronic configuration for d4 ion if Δ0 > P.
- On the basis of crystal field theory, write the electronic configuration for the d5 ion with a weak ligand for which Δ0 < P.
- Why a solution of [Ni(H2O)6]2+ is green while a solution of [Ni(CN)4]2− is colourless? (At. no. of Ni = 28)
- On adding NaOH to ammonium sulphate, a colourless gas with pungent odour is evolved, which forms a blue-coloured complex with Cu2+ ion. Identify the gas.
- A solution of [Ni(H2O)6]2+ is green, but a solution of [Ni(CO)4] is colourless. Explain.[Atomic number: Ni = 28]
- (A), (B) and (C) Are Three Non-cyclic Functional Isomers of a Carbonyl Compound with Molecular Formula C4h8o. Isomers (A) and (C) Give Positive Tollens' Test Whereas Isomer (B) Does Not Give Tollens' Test but Gives Positive Iodoform Test. Isomers (A) and (B) on Reduction with Zn(Hg)/Conc. Hcl Give the Same Product (D)
- Using Iupac Norms Write the Formulae For Dichloridobis(Ethane-1,2-diamine)Cobalt(Iii)
- Using Iupac Norms Write the Formulae for the Following: Potassium Trioxalatoaluminate(Iii)
- Write the Structures of the Main Products in the Following Reactions : -1
Concepts [17]
- Concept of Coordination Compounds
- Werner’s Theory of Coordination Compounds
- Important Terms Pertaining to Coordination Compounds
- Nomenclature of Coordination Compounds
- Isomerism in Coordination Compounds
- Stereoisomerism
- Structural Isomerism
- Bonding in Coordination Compounds
- Valence Bond Theory (VBT)
- Magnetic Properties of Coordination Compounds
- Importance and Limitations of Valence Bond Theory
- Crystal Field Theory (CFT)
- Limitations of Crystal Field Theory
- Colour in Coordination Compounds
- Bonding in Metal Carbonyls
- Importance and Applications of Coordination Compounds
- Overview of Coordination Compounds
