Advertisements
Advertisements
Question
Using valence bond theory, predict the hybridization and magnetic character of the following:
[CoF6]3– [Atomic number of Co = 27]
Advertisements
Solution
[CoF6]3– orbitals of Co2+ ion:
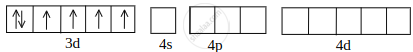
Co2+ undergoing sp3d2 hybridisation:
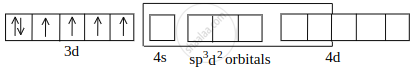
[CoF6]3– outer orbital or high-spin complex:
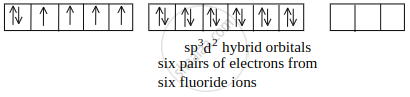
Hybridization = sp3d2
Magnetic Nature = Paramagnetic
RELATED QUESTIONS
Discuss the nature of bonding in the following coordination entity on the basis of valence bond theory:
[Fe(CN)6]4−
[Cr(NH3)6]3+ is paramagnetic while [Ni(CN)4]2− is diamagnetic. Explain why?
Discuss the nature of bonding in the following coordination entity on the basis of valence bond theory:
[FeF6]3−
Discuss the nature of bonding in the following coordination entity on the basis of valence bond theory:
[CoF6]3−
Using valence bond theory, explain the following in relation to the complexes given below:
\[\ce{[Co(NH3)6]^{3+}}\]
(i) Type of hybridisation.
(ii) Inner or outer orbital complex.
(iii) Magnetic behaviour.
(iv) Spin only magnetic moment value.
Write the hybridization and shape of the following complexes:
[Ni(CN)4]2−
What is the no. of possible isomers for the octahedral complex [Co(NH3)2(C2O4)2]?
In Fe(CO)5, the Fe – C bond possesses
Valence bond theory is based on the assumption that the bonds formed between the metal ions and ligands are ______
[Ni(CO)4] has tetrahedral geometry while [Ni(CN)4]2− has square planar, yet both exhibit diamagnetism. Explain.
[Atomic number: Ni = 28]
