Advertisements
Advertisements
Question
What type of isomerism is exhibited by the complex [Co(en)3]3+?
(en = ethane-1,2-diamine)
Advertisements
Solution 1
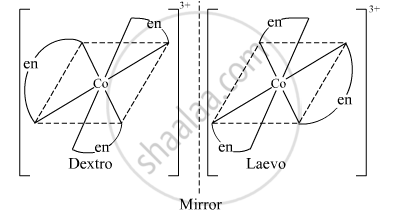
The complex [Co(en)3]3+ exhibits optical isomerism. Its optical isomers are shown below.
Solution 2
Optical isomerism is exhibited by the complex [Co (en)3]3+.
APPEARS IN
RELATED QUESTIONS
Draw the geometrical isomers of complex \[\ce{[Co(en)2Cl2]+}\].
Indicate the type of isomerism exhibited by the following complex and draw the structure for the isomer:
[Co(NH3)5(NO2)](NO3)2
Indicate the type of isomerism exhibited by the following complex and draw the structure for the isomer:
[Pt(NH3)(H2O)Cl2]
Draw the structure of optical isomers of [Cr(NH3)2Cl2(en)]+.
Draw all the isomers (geometrical and optical) of [Co(NH3)Cl(en)2]2+.
Name the type of isomerism that the compound with molecular formula C3H6O2 exhibits. Represent the isomers.
Write the IUPAC name of [Co(en)2Cl2]+ ion.
Name the type of isomerism shown by the following pair of compounds:
[Cr(NH3)5Br]SO4 and [Cr(NH3)5SO4]Br
Indicate the types of isomerism exhibited by the following complexes and draw the structures for these isomers:
[Pt(NH3)(H2O)Cl2 ]
Indicate the types of isomerism exhibited by the following complex and draw the structure for these isomer: \[\ce{[Pt(NH3)(H2O)Cl2]}\]
