Topics
Gravitation
- Concept of Gravitation
- Force and Motion
- Centripetal Force
- Kepler’s Laws
- Law of Orbit or Kepler's First Law
- Law of Areas or Kepler's Second Law
- Law of Periods or Kepler's Third Law
- Newton's Universal Law of Gravitation
- Uniform Circular Motion (UCM)
- Earth’s Gravitational Force
- Earth’s Gravitational Acceleration
- Mass and Weight
- Gravitational Waves
- Free Fall
- Gravitational Potential Energy
- Escape Velocity
- Weightlessness in Space
Periodic Classification of Elements
- Classification of Elements
- Dobereiner’s Triads
- Newland's Law of Octaves
- Mendeleev’s Periodic Table
- Insights into Mendeleev’s Periodic Table
- Modern Periodic Law
- The Modern Periodic Table
- Structure of the Modern Periodic Table
- Modern Periodic Table and Electronic Configuration of Elements
- Groups and Electronic Configuration
- Periods and Electronic Configuration
- Periodic Trends in the Modern Periodic Table
- Atomic Size
- Metallic and Non-metallic Characters
- Gradation in Halogen Family
Chemical Reactions and Equations
- Chemical Reaction
- Chemical Equations
- Balancing Chemical Equation
- Types of Chemical Reactions > Combination Reaction
- Types of Chemical Reactions > Decomposition Reaction
- Types of Chemical Reactions > Single Displacement Reaction
- Types of Chemical Reactions > Double Displacement Reaction
- Endothermic and Exothermic Processes
- Rate of Chemical Reaction
- Factors Affecting the Rate of a Chemical Reaction
- Chemical Properties of Carbon Compounds > Oxidation
- Chemical Properties of Carbon Compounds > Reduction
- Corrosion of Metals
- Rancidity
Effects of Electric Current
- Electric Circuit
- Heating Effect of Electric Current
- Magnetic Effect of Electric Current
- Right-hand Thumb Rule
- Applications of Biot-Savart's Law > Magnetic Field at the Centre of a Circular Loop
- Applications of Ampere’s Circuital Law > Magnetic Field of a Long Straight Solenoid
- Force on a Current Carrying Conductor in a Magnetic Field
- Fleming’s Left Hand Rule
- Electric Motor
- Electromagnetic Induction
- Galvanometer
- Faraday's Laws of Electromagnetic Induction
- Fleming’s Right Hand Rule
- Alternating current (AC) and Direct Current (DC)
- Electric Generator
Heat
Refraction of Light
Lenses
- Concept of Lenses
- Images Formed by Convex Lenses
- Images Formed by Concave Lenses
- Sign Convention
- Lens Formula
- Magnification
- Power of a Lens
- Combination of Lenses
- Defects of Vision and Their Corrections > Myopia
- Defects of Vision and Their Corrections > Hypermetropia
- Defects of Vision and Their Corrections > Presbyopia
- Apparent Size of an Object
- Use of Concave Lenses
- Use of Convex Lenses
- Persistence of Vision
Metallurgy
- Physical Properties of Metals
- Physical Properties of Non-metal
- Chemical Properties of Metal
- Reactions of Metals
- Reactivity Series of Metals
- Chemical Properties of Non-metal
- Ionic Compounds
- Metallurgy
- Basic Principles of Metallurgy > Concentration of Ores
- Basic Principles of Metallurgy > Extraction of Metals
- Basic Principles of Metallurgy > Refining of Metals
- Corrosion of Metals
- Prevention of Corrosion
Carbon Compounds
- Bonds in Carbon Compounds
- Carbon: A Versatile Element
- Hydrocarbons
- Straight chains, Branched chains, and Rings of Carbon atoms
- Functional Groups in Carbon Compounds
- Homologous Series
- Nomenclature
- Chemical Properties of Carbon Compounds > Combustion
- Chemical Properties of Carbon Compounds > Oxidation
- Chemical Properties of Carbon Compounds > Addition Reaction
- Chemical Properties of Carbon Compounds > Substitution Reaction
- Ethanol
- Ethanoic Acid
- Macromolecules and Polymers
Space Missions
School of Elements
The Magic of Chemical Reactions
- Chemical Equations
- Types of Chemical Reactions > Combination Reaction
- Types of Chemical Reactions > Decomposition Reaction
- Types of Chemical Reactions > Single Displacement Reaction
- Types of Chemical Reactions > Double Displacement Reaction
- Chemical Properties of Carbon Compounds > Oxidation
- Types of Double Displacement: Neutralization Reaction
The Acid Base Chemistry
- Properties of Acids > Physical Properties
- The pH Scale
- Acids, Bases and Their Reactivity
- Acid or a Base in a Water Solution
- Preparation and Uses of Baking Soda
- Preparation and Uses of Bleaching Powder
- Preparation and Uses of Washing Soda
- Preparation and Uses of Plaster of Paris
- Chemicals from Common Salt - Soap as a Salt
The Electric Spark
All about Electromagnetism
- Magnetic force
- Bar Magnet and Solenoid Analogy
- Right-hand Thumb Rule
- Applications of Biot-Savart's Law > Magnetic Field at the Centre of a Circular Loop
- Applications of Ampere’s Circuital Law > Magnetic Field of a Long Straight Solenoid
- Force on a Current Carrying Conductor in a Magnetic Field
- Electric Motor
- Electromagnetic Induction
- A.C. Generator
- Simple D.C. Motor
- Household Electrical Circuits
Wonders of Light 1
Wonders of Light 2
Striving for better Environment 1
- Abatement of Pollution
- Sustainable Use of Resources
- Introduction
- Definition: Latent Heat
- Definition: Latent Heat of Fusion
- Definition: Latent Heat of Vaporization
- Formula: Latent Heat
- Experiment
- Reference Data Table
- Example
- Key Points: Latent Heat
Introduction
The word latent comes from Latin latere, meaning "to lie hidden." When a substance changes its physical state — solid to liquid, or liquid to gas — it absorbs or releases a certain quantity of heat energy. This energy transfer occurs without any change in temperature. Because a thermometer shows no change, the heat appears to be "hidden."
Maharashtra State Board: Class 11
CISCE: Class 10
Definition: Latent Heat
The heat energy absorbed (or liberated) in change of phase is not externally manifested by any rise or fall in temperature, it is called the latent heat.
OR
Latent heat is the quantity of heat energy required to change the state of unit mass of a substance from one phase to another, at constant temperature and constant pressure.
OR
The quantity of heat absorbed or given out by unit mass of a substance during change of state of the substance at a constant temperature is called the latent heat of the substance.
Definition: Latent Heat of Fusion
The quantity of heat required to convert unit mass of a substance from its solid state to the liquid state, at its melting point, without any change in its temperature, is called its latent heat of fusion (Lf).
OR
The heat energy absorbed at constant temperature during the transformation of solid into liquid is called the latent heat of fusion. The amount of heat energy absorbed at constant temperature by unit mass of a solid to convert into liquid phase is called the specific latent heat of fusion.
Definition: Latent Heat of Vaporization
The quantity of heat required to convert unit mass of a substance from its liquid state to vapour state, at its boiling point without any change in its temperature is called its latent heat of vapourization (Lv).
Maharashtra State Board: Class 11
Formula: Latent Heat
Q = m × L
where,
Q = Heat energy absorbed or released during phase change
m = Mass of the substance undergoing phase change
L = Specific Latent Heat (characteristic of the substance & process)
SI Units = J kg⁻¹
Experiment
Procedure
- Place pieces of ice in a glass beaker. Insert a thermometer bulb into the ice and record the initial temperature.
- Mount the beaker on a stand and apply heat uniformly using a spirit burner.
- Record the temperature using the thermometer at regular 1-minute intervals.
- Stir the ice-water mixture continuously as melting begins to maintain thermal equilibrium.
- Continue heating even after all ice has melted — do not stop until all water converts to steam.
- Plot a graph of temperature (y-axis) versus time (x-axis) to visualise the phase transitions.
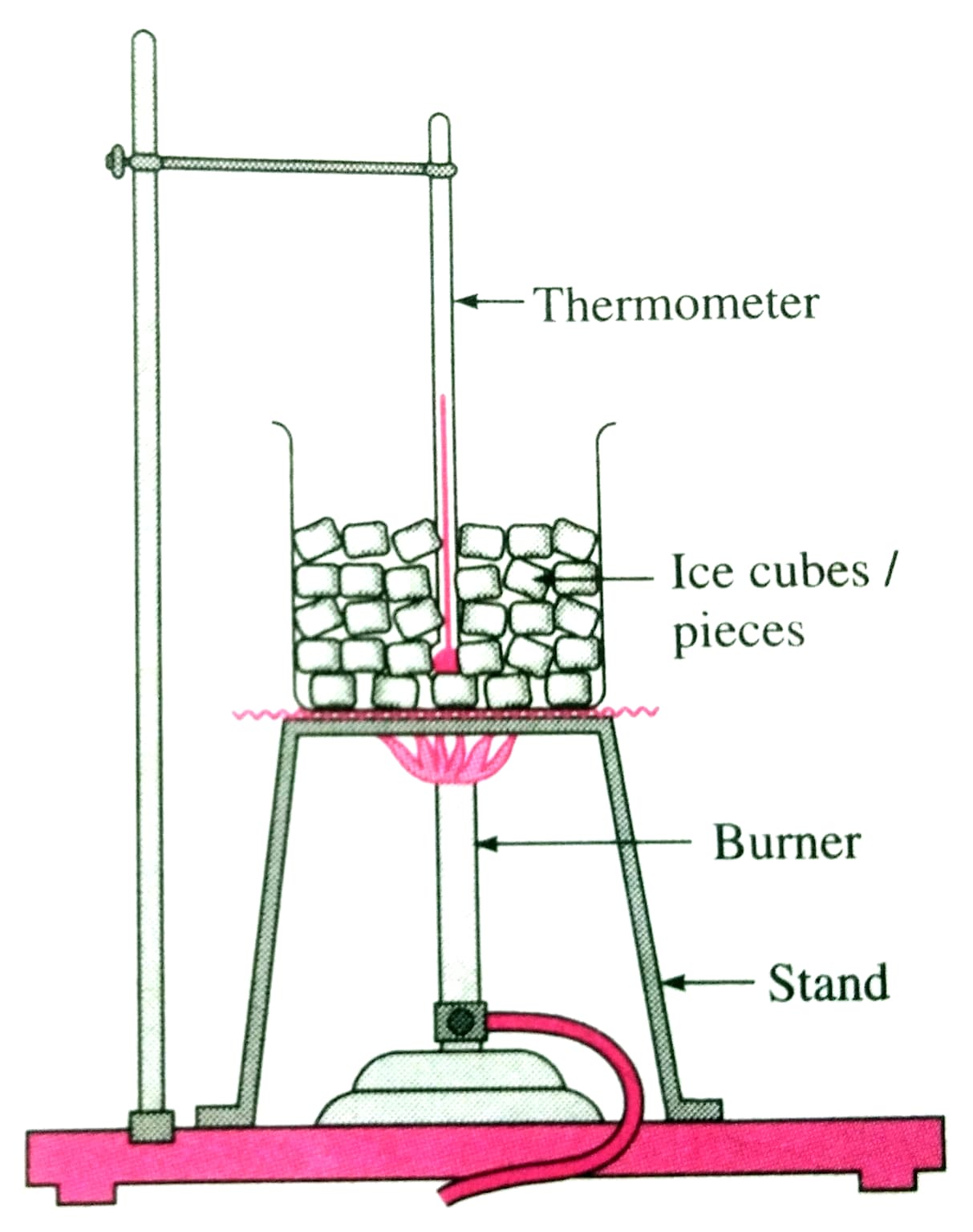
Fig.: Experimental setup: ice in a beaker, thermometer, and burner on a stand
Observations Table:
| Phase | Temperature Range | Temp. Change? | What Is Happening? |
|---|---|---|---|
| Pre-A | Below 0°C → 0°C | Yes ↑ | Ice is warming up; the kinetic energy of ice molecules increases. |
| A→B | 0°C (constant) | No — Flat | Ice melting. Heat breaks rigid bonds. Latent Heat of Fusion at work. |
| B→C | 0°C → 100°C | Yes ↑ | Liquid water warming; kinetic energy rises steadily. |
| C→D | 100°C (constant) | No — Flat | Water boiling to steam. Heat completely separates molecules. Latent Heat of Vaporisation at work. |
| D→E | Above 100°C | Yes ↑ | Steam (gas phase) heating further; the kinetic energy of gas molecules rises. |
The Heating Curve
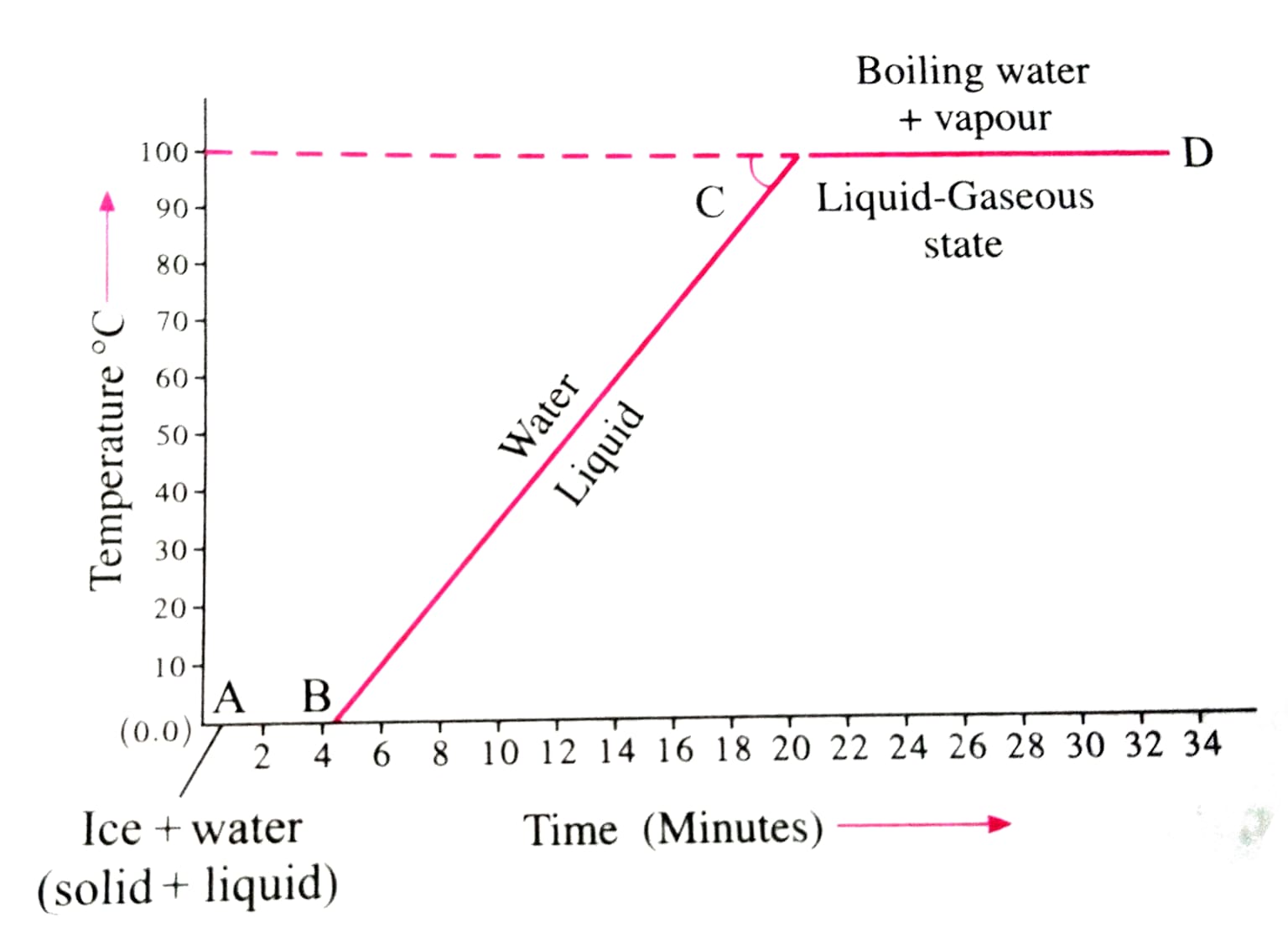
Fig. 3 — Temperature vs. Time graph for heating ice → water → steam (at 1 atm). The flat segments A→B and C→D are where latent heat is released.
Reference Data Table
All values at one standard atmospheric pressure (1 atm). Values vary at different pressures.
| Substance | Melting Point (°C) | Lf (×10⁵ J/kg) | Boiling Point (°C) | Lv (×10⁵ J/kg) |
|---|---|---|---|---|
| Water/Ice | 0 | 3.33 | 100 | 22.6 |
| Gold | 1063 | 0.645 | 2660 | 15.8 |
| Lead | 328 | 0.25 | 1744 | 8.67 |
| Ethyl Alcohol | −114 | 1.0 | 78 | 8.5 |
| Mercury | −39 | 0.12 | 357 | 2.7 |
| Nitrogen | −210 | 0.26 | −196 | 2.0 |
| Oxygen | −219 | 0.14 | −183 | 2.1 |
Example
Problem: When 0.1 kg of ice at 0°C is mixed with 0.32 kg of water at 35°C in a container, the resulting temperature of the mixture is 7.8°C. Calculate the latent heat of fusion of ice.
Given: swater = 4186 J kg⁻¹ K⁻¹
Given Data:
Mass of ice (mice) = 0.1 kg
Mass of water (mw) = 0.32 kg
Temp. of ice (Tice) = 0°C
Temp. of water (Tw) = 35°C
Final temp. (TF) = 7.8°C
Specific heats = 4186 J kg⁻¹K⁻¹
The warm water cools from 35°C to 7.8°C, losing heat to the ice.
Qlost = mw × s × (Tw − TF) = 0.32 × 4186 × (35 − 7.8) = 0.32 × 4186 × 27.2 = 36,434.9 J
Ice gains heat in two stages: (a) to melt at 0°C, then (b) to warm the meltwater to 7.8°C.
Heat lost by water = Heat gained by ice (melting + warming)
Key Points: Latent Heat
- Formula: Q = mL. Specific latent heat L has SI unit J kg⁻¹.
- Temperature stays constant during any phase change. Heat energy goes into breaking or forming intermolecular bonds, not into raising kinetic energy.
- Latent Heat of Fusion (water): Lf = 3.33 × 10⁵ J kg⁻¹ = 80 cal/g. Heat needed to melt 1 kg of ice at 0°C.
- Latent Heat of Vaporisation (water): Lv = 22.6 × 10⁵ J kg⁻¹ = 540 cal/g. Heat is needed to convert 1 kg of water to steam at 100°C.
- Lv ≫ Lf because vaporisation requires complete molecular separation and work against atmospheric pressure during expansion.
- All latent heat values depend on atmospheric pressure. Standard values quoted at 1 atm. Increasing pressure raises the boiling point (pressure cooker effect).
