- Alloys are homogeneous mixtures of metals (or metals with non-metals) with different properties than pure metals—often harder, less conductive, and with lower melting points.
- 24-carat gold is soft, so 22-carat gold (an alloy with copper or silver) is used in jewellery to improve strength.
- Solder, an alloy with a low melting point, is used to join electrical wires.
- Corrosion prevention methods include painting, galvanising, anodising, electroplating, and alloying.
- Galvanising, tinning, anodisation, and electroplating all involve protective coatings to prevent corrosion, while alloying enhances durability and resistance.
Topics
Chemical Reactions and Equations
- Chemical Reactions in Daily Life
- Chemical Equations
- Balancing Chemical Equation
- Types of Chemical Reactions > Combination Reaction
- Types of Chemical Reactions > Decomposition Reaction
- Types of Chemical Reactions > Single Displacement Reaction
- Types of Chemical Reactions > Double Displacement Reaction
- Chemical Properties of Carbon Compounds > Oxidation
- The Effects of Oxidation Reactions in Everyday Life
Acids, Bases and Salts
- Acids and Bases in Daily Life
- Acids and Bases in the Laboratory
- Acids and Bases React with Metals
- Reaction of Metal Carbonates with Acids
- Acids and Bases Reaction with each other
- Reaction of Metallic Oxides with Acids
- Reaction of a Non-metallic Oxide with Base
- Common Properties of Acids and Bases
- The pH Scale
- Importance of pH in Everyday Life
- Salts > Family of Salts
- Salts > pH of Salts
- Salts > Chemicals from Common Salt
- Salts > Water in Salt Crystals
Metals and Non-metals
Carbon and its Compounds
- Importance of Carbon
- The Covalent Bond
- Allotropes of Carbon > Diamond
- Allotropes of Carbon > Graphite
- Allotropes of Carbon > Fullerene
- Carbon: A Versatile Element
- Organic Compounds
- Classification of Hydrocarbons
- Carbon Compounds: Chains, Branches, Rings
- Homologous Series
- Nomenclature
- Chemical Properties of Carbon Compounds > Combustion
- Ethanol
- Ethanoic Acid
- Soaps and Detergents
Life Processes
- Life Processes in Living Organisms
- Nutrition
- Autotrophic Nutrition
- Heterotrophic Nutrition
- Nutrition in Human Beings
- Dental Caries
- Production of ATP
- Blood Circulatory System
- Blood Vessels Entering and Leaving The Heart
- Valves of the Heart
- Tissue Fluid (Or Intercellular Fluid)
- Transportation in Plants
- Transportation of Water
- Excretion
- Excretion in Human Beings
- Kidney and Its Internal Structure
- Structure of a Kidney Tubule (Nephrons)
- Dialysis and Artificial Kidney
- Excretion in Plants
- Organ and Body Donation
Control and Co-ordination
How do Organisms Reproduce?
Heredity
Light – Reflection and Refraction
- Light and Its Straight-Line Propagation
- Reflection of Light
- Spherical Mirrors
- Image Formation by Spherical Mirrors
- Representation of Images Formed by Spherical Mirrors
- Image Formation by Concave Mirror
- Image Formation by a Convex Mirror
- Sign Convention for Reflection by Spherical Mirrors
- Ray Optics - Mirror Formula
- Refraction of Light
- Refraction through a Rectangular Glass Slab
- The Refractive Index
- Refraction by Spherical Lenses
- Image Formation by Lenses
- Image Formation in Lenses Using Ray Diagrams
- Sign Convention for Spherical Lenses
- Lens Formula
- Power of a Lens
The Human Eye and the Colourful World
Electricity
Magnetic Effects of Electric Current
- Magnetic Effect of Electric Current
- Applications of Biot-Savart's Law > Magnetic Field due to a Finite Straight Current-Carrying Wire
- Magnetic Field Due to a Current-Carrying Conductor
- Right-hand Thumb Rule
- Applications of Biot-Savart's Law > Magnetic Field at the Centre of a Circular Loop
- Applications of Ampere’s Circuital Law > Magnetic Field of a Long Straight Solenoid
- Force on a Current Carrying Conductor in a Magnetic Field
- Fleming’s Left Hand Rule
- Magnetism in Medicine
- Domestic Electric Circuits
Our Environment
- Key Points: Prevention of Corrosion
Maharashtra State Board: Class 10
Coating with a Protective Layer
Corrosion is a natural process that deteriorates metals due to environmental factors like moisture, oxygen, and chemicals. To protect metals, especially iron, various methods are used to prevent rusting and extend the lifespan of metal objects. The rate of corrosion can be reduced by isolating metals from direct contact with air and moisture.
A layer of paint, oil, grease, varnish, or enamel is applied to block air and moisture from reaching the metal surface.
- Painting and Varnishing: Metals like iron gates, railings, and furniture are coated with paint or varnish to prevent rust. However, if the paint gets scratched, rusting starts underneath.
- Greasing and Oiling: It is used for moving parts of machines, gears, and bicycle chains to prevent rusting by reducing moisture contact.
- Enamelling: A glassy layer fused onto metals to provide long-term corrosion resistance.
Limitations:
- If the coating gets scratched or damaged, rusting can begin underneath.
- It requires regular maintenance and reapplication over time.
Maharashtra State Board: Class 10
Metal Plating (Galvanisation, Tinning, Electroplating)
A non-corrodible metal is coated onto corrodible metals like iron or steel to prevent corrosion.
1. Galvanisation: A thin layer of zinc is applied to iron or steel to prevent rusting. Examples include shiny iron nails and pins. Zinc corrodes before iron because it is more electropositive, but once the zinc layer wears off, iron starts rusting.
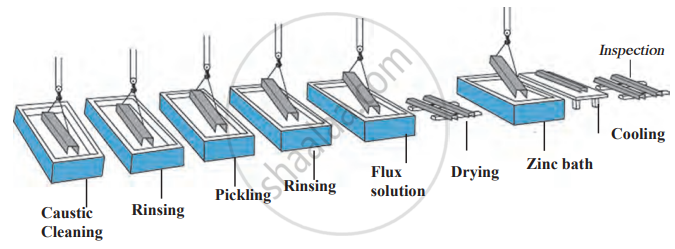
Galvanising Process
2. Tinning: Molten tin is coated on brass or copper utensils (also called kalhaee) to prevent corrosion. This protects against the greenish poisonous layer that forms on copper and brass, especially when in contact with acidic foods like buttermilk or curry.
3. Electroplating: A less reactive metal (silver, gold, or chromium) is deposited onto a more reactive metal using electrolysis. Examples include silver-plated spoons, gold-plated jewellery, and chromium-plated taps.
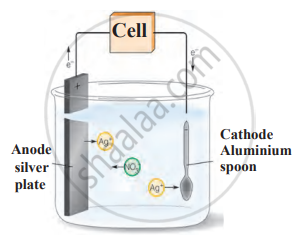
Electroplating
Limitations:
- Over time, the protective layer wears off, exposing the metal to corrosion.
- The plating process can be expensive.
Maharashtra State Board: Class 10
Anodisation (Formation of Oxide Layer)
In anodisation, metals like aluminium and copper are coated with a thin and strong oxide layer using electrolysis.
- The oxide layer is uniform, strong, and durable, preventing further oxidation.
- Protection can be increased by making the oxide layer thicker.
Example: Anodised aluminium cookware forms a protective Al₂O₃ layer, preventing oxidation and corrosion.
Limitations: Only applicable to certain metals like aluminium and copper.
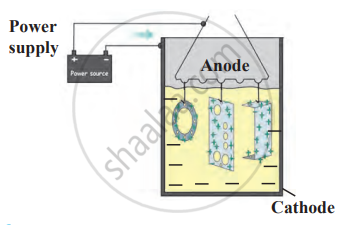
Anodization
Maharashtra State Board: Class 10
Alloying (Corrosion-Resistant Alloys)
Metals are mixed with other metals or nonmetals to create alloys, which are stronger and more resistant to corrosion.
- It provides permanent protection against corrosion.
- Improves the strength and durability of metals.
Examples,
- Bronze (90% copper + 10% tin): Used for statues and sculptures, resistant to sun and rain.
- Stainless Steel (74% iron + 18% chromium + 8% carbon): Does not rust or stain, making it ideal for utensils, construction materials, and surgical instruments.
- Coins made from alloys: Modern coins are made from corrosion-resistant alloys to prevent wear and tear.
Limitations: Expensive and difficult to separate metals once alloyed.

Coins made from various alloys
CBSE: Class 10
Maharashtra State Board: Class 10
Maharashtra State Board: Class 10
