Topics
Chemical Reactions and Equations
- Chemical Reactions in Daily Life
- Chemical Equations
- Balancing Chemical Equation
- Types of Chemical Reactions > Combination Reaction
- Types of Chemical Reactions > Decomposition Reaction
- Types of Chemical Reactions > Single Displacement Reaction
- Types of Chemical Reactions > Double Displacement Reaction
- Chemical Properties of Carbon Compounds > Oxidation
- The Effects of Oxidation Reactions in Everyday Life
Acids, Bases and Salts
- Acids and Bases in Daily Life
- Acids and Bases in the Laboratory
- Acids and Bases React with Metals
- Reaction of Metal Carbonates with Acids
- Acids and Bases Reaction with each other
- Reaction of Metallic Oxides with Acids
- Reaction of a Non-metallic Oxide with Base
- Common Properties of Acids and Bases
- The pH Scale
- Importance of pH in Everyday Life
- Salts > Family of Salts
- Salts > pH of Salts
- Salts > Chemicals from Common Salt
- Salts > Water in Salt Crystals
Metals and Non-metals
Carbon and its Compounds
- Importance of Carbon
- The Covalent Bond
- Allotropes of Carbon > Diamond
- Allotropes of Carbon > Graphite
- Allotropes of Carbon > Fullerene
- Carbon: A Versatile Element
- Organic Compounds
- Classification of Hydrocarbons
- Carbon Compounds: Chains, Branches, Rings
- Homologous Series
- Nomenclature
- Chemical Properties of Carbon Compounds > Combustion
- Ethanol
- Ethanoic Acid
- Soaps and Detergents
Life Processes
- Life Processes in Living Organisms
- Nutrition
- Autotrophic Nutrition
- Heterotrophic Nutrition
- Nutrition in Human Beings
- Dental Caries
- Production of ATP
- Blood Circulatory System
- Blood Vessels Entering and Leaving The Heart
- Valves of the Heart
- Tissue Fluid (Or Intercellular Fluid)
- Transportation in Plants
- Transportation of Water
- Excretion
- Excretion in Human Beings
- Kidney and Its Internal Structure
- Structure of a Kidney Tubule (Nephrons)
- Dialysis and Artificial Kidney
- Excretion in Plants
- Organ and Body Donation
Control and Co-ordination
How do Organisms Reproduce?
Heredity
Light – Reflection and Refraction
- Light and Its Straight-Line Propagation
- Reflection of Light
- Spherical Mirrors
- Image Formation by Spherical Mirrors
- Representation of Images Formed by Spherical Mirrors
- Image Formation by Concave Mirror
- Image Formation by a Convex Mirror
- Sign Convention for Reflection by Spherical Mirrors
- Ray Optics - Mirror Formula
- Refraction of Light
- Refraction through a Rectangular Glass Slab
- The Refractive Index
- Refraction by Spherical Lenses
- Image Formation by Lenses
- Image Formation in Lenses Using Ray Diagrams
- Sign Convention for Spherical Lenses
- Lens Formula
- Power of a Lens
The Human Eye and the Colourful World
Electricity
Magnetic Effects of Electric Current
- Magnetic Effect of Electric Current
- Applications of Biot-Savart's Law > Magnetic Field due to a Finite Straight Current-Carrying Wire
- Magnetic Field Due to a Current-Carrying Conductor
- Right-hand Thumb Rule
- Applications of Biot-Savart's Law > Magnetic Field at the Centre of a Circular Loop
- Applications of Ampere’s Circuital Law > Magnetic Field of a Long Straight Solenoid
- Force on a Current Carrying Conductor in a Magnetic Field
- Fleming’s Left Hand Rule
- Magnetism in Medicine
- Domestic Electric Circuits
Our Environment
- Fullerene
- Uses of Fullerene
Maharashtra State Board: Class 9
Fullerene:
Fullerene is an allotrope of carbon that is rarely found in nature, existing in soot and interstellar space. The first discovered fullerene, Buckminsterfullerene (C60), was named after architect Richard Buckminster Fuller due to its resemblance to the geodesic dome he designed. The discovery of fullerene C60 by Harold Kroto, Robert Curl, and Richard Smalley earned them the Nobel Prize in Chemistry in 1996. Fullerenes, including C60, C70, and others, exhibit unique molecular structures and properties.

Buckytubes (Carbon nanotubes)
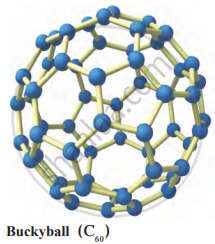
Structure of fullerene
- Fullerenes exist in the form of hollow spheres (buckyballs) or tubes (buckytubes).
- A fullerene molecule can contain between 30 to 900 carbon atoms, depending on its type.
- Buckminsterfullerene (C60) is the most well-known example, resembling a soccer ball in structure.
- Fullerenes are soluble in organic solvents such as carbon disulphide and chlorobenzene.
- They are lightweight and possess excellent thermal and chemical stability.
- Fullerenes exhibit unique electrical and optical properties, making them useful in nanotechnology and materials science.
- They are used in drug delivery systems, as they can trap and transport molecules within their structure.
- Fullerenes have potential applications in superconductors, solar cells, and lubricants due to their structural and chemical properties.
Maharashtra State Board: Class 9
Uses of Fullerene:
- Fullerenes are used as insulators due to their unique molecular structure.
- They act as catalysts in water purification systems, helping to remove impurities.
- Fullerenes exhibit superconductivity at certain temperatures, making them valuable in advanced technologies.
- They are used in drug delivery systems, as they can encapsulate and transport molecules to targeted areas.
- Fullerenes are applied in the development of solar cells to improve energy efficiency.
- They are used in the production of lubricants due to their spherical molecular structure, reducing friction.
- Fullerenes are utilised in nanotechnology for creating advanced materials and devices.
- They are employed in the development of antioxidants, as fullerenes can neutralise free radicals.
- Fullerenes have applications in sensors due to their ability to interact with different chemical and physical environments.
