Advertisements
Chapters
1: Solid State
2: Solutions
3: Electrochemistry
4: Chemical Kinetics
5: Surface Chemistry
Inorganic Chemistry
6: General Principles and Processes of Isolation of Elements
▶ 7: p-Block Elements
8: d-and ƒ-Block Elements
9: Coordination Compounds
Organic Chemistry
10: Haloalkanes and Haloarenes
Chapter 11: Alcohols, Phenols and Ethers
Chapter 12: Aldehydes, Ketones and Carboxylic Acids
Chapter 13: Organic Compounds containing Nitrogen
Chapter 14: Biomolecules
15: Polymers
Chapter 16: Chemistry in Everyday Life
![Nootan solutions for केमिस्ट्रि पार्ट १ अँड २ [इंग्रजी] इयत्ता १२ आयएससी chapter 7 - p-Block Elements Nootan solutions for केमिस्ट्रि पार्ट १ अँड २ [इंग्रजी] इयत्ता १२ आयएससी chapter 7 - p-Block Elements - Shaalaa.com](/images/chemistry-part-1-and-2-english-class-12-isc_6:a6407550bf9a46188b122e0d354548f8.png)
Advertisements
Solutions for Chapter 7: p-Block Elements
Below listed, you can find solutions for Chapter 7 of CISCE Nootan for केमिस्ट्रि पार्ट १ अँड २ [इंग्रजी] इयत्ता १२ आयएससी.
Nootan solutions for केमिस्ट्रि पार्ट १ अँड २ [इंग्रजी] इयत्ता १२ आयएससी 7 p-Block Elements REVIEW EXERCISES [Pages 384 - 441]
Why is dinitrogen unreactive at ordinary temperature?
What happens when dinitrogen is passed over calcium carbide at 1300 K?
What happens when magnesium nitride is treated with water?
What happens when ammonia is treated with excess of chlorine?
What happens when nitric acid is poured over saw dust?
What happens when nitric acid is treated with gold?
Why is solubility of ammonia in water very high?
Explain why a precipitate of silver chloride dissolves in NH4OH.
Explain, why zinc sulphate does not give a precipitate with excess of NH4OH.
Explain, why nitric acid makes wool yellow.
Why does oxygen not show oxidation states of +4 and +6, whereas sulphur does so?
What type of hybridisation is involved for oxygen in H2O? Give reason for your answer.
Give the structure of sulphur tetrafluoride.
Discuss briefly the properties of group 16 elements.
Define catenation and illustrate with example.
Arrange H2S, H2O, H2Se, H2Te in the order of decreasing thermal stability.
Explain the hybridisation in SF6 molecule.
What is the shape of SF6 molecule?
What is the oxidation state of sulphur in the following?
Sulphurous acid
What is oxidation state of sulphur in the following?
Sulphuric acid
What is the oxidation state of S in the following oxoacid?
Dithionic acid
What is the oxidation state of S in the following oxoacid?
Peroxodisulphuric acid
What type of hybridisation does explain the trigonal bipyramidal shape of SF4?
Describe the properties of O, S, Se, Te and Po (group 16 elements) with reference to metallic/non-metallic character.
Describe the properties of O, S, Se, Te and Po (group 16 elements) with reference to catenation.
Describe the properties of O, S, Se, Te and Po (group 16 elements) with reference to thermal stability of hydrides.
Describe the properties of O, S, Se, Te and Po (group 16 elements) with reference to oxidation states.
Describe the properties of O, S, Se, Te and Po (group 16 elements) with reference to allotropy.
Draw the structure of peroxymonosulphuric acid.
Which has the larger bond angle H2S or H2O and why?
Explain the following observation:
Among the hydrides of elements of group 16, water shows unusual physical properties.
Give reasons for the following:
Sulphur in vapour state shows paramagnetic behaviour.
Account for the following:
Tendency to show −2 oxidation state diminishes from sulphur to poloniuum in group 16.
Account for the following:
NO2 readily forms a dimer whereas ClO2 does not.
Assign appropriate reason for the following observation.
SF6 is not easily hydrolysed whereas SF4 is easily hydrolysed.
Assign appropriate reason for the following observation.
Sulphur in vapour state exhibits some paramagnetic behaviour.
Give reason for the following statement:
SF6 is not easily hydrolysed though thermodynamically it should be.
Give reason for the following fact:
Sulphur disappears when boiled with an aqueous solution of sodium sulphite.
Give reason for the following fact:
H2SO4 is a diprotic acid.
Assign a reason for the following:
Sulphur hexafluoride is used as a gaseous electrical insulator.
SF6 is known but SCl6 is not. Why?
Assign a reason for the following:
SF6 is not easily hydrolysed.
Discuss the structure:
SO2
Discuss the structure:
SO3
What is allotropy?
Why is ozone regarded as an allotrope of oxygen?
Why is ozone used as a disinfectant?
What is tailing of mercury?
How is ozone formed in the upper atmosphere?
How is ozone useful for us?
What happens when ozone is passed in an acidified solution of ferrous sulphate?
What happens when moist sulphur is treated with ozone?
What happens when ozone is passed in an aqueous solution of potassium iodide?
Out of oxygen and ozone, which is a better oxidising agent and why?
How is ozone prepared?
Discuss the structure of ozone.
Describe the important oxidation reactions of ozone.
What happens when ozone is treated with ethylene?
What happens when ozone reacts with potassium iodide?
What happens when black lead sulphide is treated with ozone?
What happens when ozone is treated with an aqueous solution of potassium manganate?
Why is it that molecular nitrogen is not particularly reactive?
Among the hydrides of group 15, predict the hydride having highest thermal stability.
Among the hydrides of group 15, predict the hydride having largest bond angle.
Among the hydrides of group 15, predict the hydride having lowest boiling point.
Among the hydrides of group 15, predict the hydride having maximum reducing power.
Among the hydrides of group 15, predict the hydride having least basic character.
Write the names and structures of oxoacids of phosphorus in which phosphorus exists in +1 oxidation state.
Write the names and structures of oxoacids of phosphorus in which phosphorus exists in +3 oxidation state.
Write the names and structures of oxoacids of phosphorus in which phosphorus exists in +5 oxidation state.
Write the names and structures of oxoacids of phosphorus in which phosphorus exists in +7 oxidation state.
Complete the following chemical equation:
\[\ce{Ca3P2 + H2O ->}\]
Complete and balance the following reaction:
\[\ce{N2O4 + NO ->[250 K]}\]
Complete and balance the following chemical equation:
\[\ce{Pb(NO3)2 ->[Heat]}\]
Complete and balance the following reaction:
\[\ce{NH3 + NaOCl ->}\]
Write the state of hybridisation of nitrogen in \[\ce{NO^-_3}\] ion.
Write the structure of pyrophosphoric acid.
Nitrogen exists as diatomic molecule and phosphorus as P4. Why?
Explain giving reason the following:
SF6 is known but SH6 is not known.
Arrange the following in the decreasing order of property indicated:
H2O, H2S, H2Se, H2Te (boiling point)
Arrange the following in the decreasing order of property indicated:
NH3, PH3, AsH3, SbH3 (basic strength)
Why are all the P-F bonds in PF5 molecule not of the same length?
Why does oxygen not show oxidation states of +4 and +6, whereas sulphur does so?
What type of hybridisation is involved for oxygen in H2O? Give reason for your answer.
Give the structure of sulphur tetrafluoride.
Discuss briefly the properties of group 16 elements.
Define catenation and illustrate with example.
Arrange H2S, H2O, H2Se, H2Te in the order of decreasing thermal stability.
Explain the hybridisation in SF6 molecule.
What is the shape of SF6 molecule?
What is the oxidation state of sulphur in the following?
Sulphurous acid
What is oxidation state of sulphur in the following?
Sulphuric acid
What is the oxidation state of S in the following oxoacid?
Dithionic acid
What is the oxidation state of S in the following oxoacid?
Peroxodisulphuric acid
Account for the following:
NH3 has higher boiling point than PH3.
Account for the following:
H3PO3 is a diprotic acid.
Account for the following:
OF2 should be called oxygen difluoride and not fluorine oxide.
What type of hybridisation does explain the trigonal bipyramidal shape of SF4?
Describe the properties of O, S, Se, Te and Po (group 16 elements) with reference to metallic/non-metallic character.
Describe the properties of O, S, Se, Te and Po (group 16 elements) with reference to catenation.
Describe the properties of O, S, Se, Te and Po (group 16 elements) with reference to thermal stability of hydrides.
Describe the properties of O, S, Se, Te and Po (group 16 elements) with reference to oxidation states.
Describe the properties of O, S, Se, Te and Po (group 16 elements) with reference to allotropy.
PH3 forms bubbles when passed slowly in water but NH3 dissolves. Explain why?
Draw the structure of peroxymonosulphuric acid.
State the type of hybrid orbitals associated with P in PCl5.
State the type of hybrid orbital associated with S in SF6.
What is the nature of bonds in NO2 molecule?
Which has the larger bond angle H2S or H2O and why?
Describe the manufacturing of H2SO4 by the contact process.
Discuss the structure of PCl3.
Oxides of nitrogen have open chain structure while those of phosphorus have closed chain or cage structures. Why is it so? Illustrate with one structural example for each type of oxides.
Which type of hybridization is present in the ammonia molecule?
What is the expected bond angle in NH3?
Explain the following observation:
Among the hydrides of elements of group 16, water shows unusual physical properties.
Explain the following observation:
Unlike phosphorus, nitrogen shows little tendency for catenation.
Explain the following observation:
Unlike phosphorus, nitrogen shows little tendency for catenation.
Explain the following observation:
Sulphur in vapour state exhibits paramagnetic behaviour.
Account for the following:
Tendency to show −2 oxidation state diminishes from sulphur to poloniuum in group 16.
Account for the following:
NO2 readily forms a dimer whereas ClO2 does not.
Account for the following:
PH3 is a weaker base than NH3.
Account for the following:
SF6 exists but OF6 does not.
Account for the following:
Sulphur exhibits tendency for catenation but oxygen does not.
Assign appropriate reason for the following observation:
Phosphinic acid behaves as a monoprotic acid.
Assign appropriate reason for the following observation.
SF6 is not easily hydrolysed whereas SF4 is easily hydrolysed.
Give reason for the following:
Red phosphorus is less reactive than white phosphorus.
Assign appropriate reason for the following observation.
Sulphur in vapour state exhibits some paramagnetic behaviour.
Give chemical reaction in support the following:
All the bonds in PCl5 molecule are not equivalent.
Give chemical reaction in support of the following:
Sulphur exhibits greater tendency for catenation than selenium.
Give reason for the following statement:
SF6 is not easily hydrolysed though thermodynamically it should be.
Give reason for the following:
Nitric oxide becomes brown when released in air.
Give reason for the following:
PCl5 is ionic in the solid state.
Give reason for the following fact:
Sulphur disappears when boiled with an aqueous solution of sodium sulphite.
Give reason for the following fact:
H3PO4 is a diprotic acid.
Assign a reason for the following:
In group 15, the bond angle \[\ce{H - M - H}\] decreases in the following order: NH3 (107.8°), PH3 (93.6°), AsH3 (91.8°).
Assign a reason for the following:
Sulphur hexafluoride is used as a gaseous electrical insulator.
SF6 is known but SCl6 is not. Why?
Assign a reason for the following:
Not all bonds in a molecule of PCl5 are equivalent.
Assign a reason for the following:
SF6 is not easily hydrolysed.
Write chemical equation for the following reaction:
\[\ce{Ca3(PO4)2 + SiO2 + C ->}\]
Write the structures of the following molecule:
H2SO3
Draw the structure of the following:
H2SO4
Draw the structure of the following:
H2S2O7
Describe the following about halogen family elements:
Relative oxidising power
Describe the following about halogen family elements:
Relative acid strength of hydrides.
Describe the following about halogen family elements:
Oxoacids and their relative oxidising ability
What are interhalogen compounds?
How are interhalogen compounds classified?
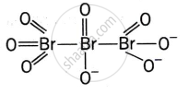
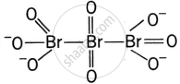
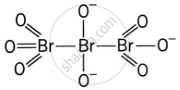
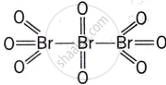
Give the structure of IF7.
Discuss briefly the properties of group 17 elements (halogen family).
State two examples of compounds in which halogens exhibit higher oxidation state.
Arrange the following in the order of property indicated for the given set:
HF, HCl, HBr, HI - increasing acid strength.
Arrange the following according to the property mentioned against the given:
HClO4, HIO4, HBrO4 in the order of increasing oxidising ability.
Account for the following:
HI in aqueous solution is a stronger acid than hydrochloric acid.
Account for the following:
Noble gases exhibit low chemical activity.
Name a compound in which chlorine displays an oxidation state of +7.
Write the balanced equation for the following reaction:
\[\ce{BrO^-_3 + F2 + OH- ->}\]
Which one among the following is the strongest oxidising agent?
\[\ce{ClO^-_4, BrO^-_4, IO^-_4}\]
Why is HF the weakest acid among hydrohalo acids inspite of the fact that fluorine is most electronegative?
Arrange the following in decreasing order of property indicated:
F2, Cl2, Br2, I2 (bond energy)
Arrange the following in the increasing order of the property mentioned:
MF, MCl, MBr, MI (ionic character)
Write an example of a neutral molecule which is isoelectronic to ClO−.
Explain, why is the boiling point of hydrogen fluoride higher than that of hydrogen chloride?
Give two examples of pseudohalogens.
What are noble gases and why are they called so?
Write the names and electronic configurations of all the noble gases.
Write the structures of the following molecule:
XeOF4
Complete the equation:
\[\ce{XeF4 + H2O ->}\]
Name the molecular geometry of XeOF4.
Complete and balance the following equation:
\[\ce{XeF6 + KF ->}\]
Explain, why does neon not form compounds analogous to those formed by xenon?
Name the molecular geometry of XeO3.
What is the state of hybridisation of Xe in XeF2?
What is the state of hybridisation of Xe in XeF6?
What is the state of hybridisation of Xe in XeOF6?
What is the state of hybridisation of Xe in XeO3?
What is the state of hybridisation of Xe in XeF4?
Complete the following equation:
\[\ce{XeF2 + H2O ->}\]
Complete the equation:
\[\ce{XeF4 + H2O ->}\]
Complete the following reaction.
\[\ce{XeF6 + H2O ->}\]
Complete and balance the following reaction:
\[\ce{XeF6 + SiO2 ->}\]
Explain the shape of XeF4 on the basis of VSEPR theory.
What products are formed when XeF4 is hydrolysed?
Why are interhalogen compounds more reactive than the related elemental halogens?
State reason for the following:
Fluorine provides the largest variety of interhalogen compounds amongst halogens.
Why are fluorine and oxygen compounds more aptly called oxygen fluorides?
Fluorine has lower electron affinity than chlorine and yet it is a stronger oxidising agent than chlorine. Explain.
Of HI and HCl which has a weaker covalent bond and what effect has it on their acid strengths?
NaOCl solution becomes unstable on warming. What happens to it?
Draw the structure of the following molecule:
XeF4
Draw the structure of SF4 molecule.
Give appropriate reason for each of the following observation:
Of the noble gases, only xenon is known to form chemical compounds.
Give appropriate reason for the following observation:
Despite its lower electron affinity, fluorine is a stronger oxidising agent than chlorine.
Why is HF not stored in plain glass bottles?
Give appropriate reason for the following observation:
Only higher members of group 18 of the periodic table are expected to form compounds.
Fluorine has lower electron affinity than chlorine and yet it is a stronger oxidising agent than chlorine. Explain.
Account for the following:
NO2 readily forms a dimer whereas ClO2 does not.
Draw the structure of \[\ce{SiF^{2-}_6}\].
Draw the structure of the following molecule:
XeF4
Write the structures of the following molecule:
XeOF4
Give chemical evidence for the following:
Fluorine is a stronger oxidising agent than chlorine.
Assign appropriate reason for the following:
Hydrogen fluoride is a much weaker acid than HCl in aqueous solution.
Assign appropriate reason for the following:
The bond energy of F2 is less than that of Cl2.
Assign appropriate reason for the following:
Bleaching of flowers by Cl2 is permanent while by SO2 is temporary.
Complete the following equation:
\[\ce{XeF2 + H2O ->}\]
Write balanced chemical equation for the following reaction:
\[\ce{XeF4 + SbF5 ->}\]
Write balanced chemical equation for the following reaction:
\[\ce{U + ClF3 ->}\]
Balance the following equation:
\[\ce{XeF6 + H2O -> XeO2F2 + HF}\]
Complete the following reaction equation:
\[\ce{CaOCl2 + HCl ->}\]
Write the structures of the following:
IF5
Write the structure of the following:
XeOF3
Draw the structure of the following:
XeO3
Answer the following.
Draw structures of XeF2.
Write the structural formula for IF3.
Draw the structure of hypochlorous acid.
Draw the structure of chlorous acid.
Write the structures of the following molecule:
XeOF4
Draw the structure of the following:
XeO3
Nootan solutions for केमिस्ट्रि पार्ट १ अँड २ [इंग्रजी] इयत्ता १२ आयएससी 7 p-Block Elements VERY SHORT ANSWER TYPE QUESTIONS [Page 446]
Among groups 14 and 15, the elements of which group are less metallic?
How does the metallic character vary in group 15?
What type of multiple bonds are present in N2 molecule?
What is the molecular formula of a white phosphorus molecule?
What is the shape of a white phosphorus molecule?
Name the shape of the hydrides of group 15 elements.
Arrange the hydrides (MH3) of group 15 elements in the decreasing order of thermal stability.
Arrange the hydrides (MH3) of group 15 elements in the decreasing order of basic character.
Arrange the hydrides (MH3) of group 15 elements in the decreasing order of reducing character.
Name the shape of PCl3 molecule.
Name the shapes of PCl5 molecule.
Among N2O3, P4O6 and As4O5, which is most acidic?
Write the formulae of the oxoacids of phosphorus in which phosphorus is present in +5 oxidation state.
What is the basicity of phosphorus acid (H3PO3)?
What are chalcogens?
Write the valence shell electronic configuration of group 16 elements.
Write the molecular formula of sulphur.
Which has a higher electron affinity, O or S?
Write the formula of a compound in which oxygen exists in +2 oxidation state.
Can oxygen exist in +6 oxidation state?
What type of hybridisation is usually involved in the compounds containing sulphur in +6 oxidation state?
Name the metal(s) which belong(s) to group 16.
Write the formulae of the allotropes of oxygen.
What type of hybridisation is involved in the formation of a H2O molecule?
Arrange H2O, H2S, H2Se and H2Te in the decreasing order of volatility.
Arrange H2O, H2S, H2Se and H2Te in the decreasing order of acidic character.
Does water act as a reducing agent?
What is the shape of SF6 molecule?
Draw the structure of SO3 molecule.
Why are the elements of group 17 called ‘halogens’?
Name a halogen which exists as a solid at room temperature.
The elements of which group do have the smallest atomic radii in respective periods?
How does the following property vary on moving down group 17?
Density
How does the following property vary on moving down group 17?
Electron affinity
How does the following property vary on moving down group 17?
Ionisation energy
Among the halogens, which has the highest electron affinity?
Can fluorine exhibit positive oxidation states?
Can iodine displace chlorine from NaCl solution?
Which hydrogen halide does exist in the liquid state at 290 K?
Arrange halogen acids in the decreasing order of thermal stability.
Arrange halogen acids in the decreasing order of reducing power.
Arrange the halogen acids in the decreasing order of acidic strength.
What is the name of the compound OF2?
What is the general formula of perhalic acids?
Among HCIO, HBrO and HIO, which is most acidic?
Arrange oxoacids of chlorine in the decreasing order of their acidic strength.
In an interhalogen compound XYn, which halogen (X or Y) does exist in −1 oxidation state?
Among Cl2, Br2 and BrCl, which is most reactive?
What is the shape of ClF3 molecule?
What type of hybridisation is involved in the formation of IF5 molecule?
Name a pseudo halide ion and the corresponding pseudo halogen.
What is the value of Cp/Cv for a noble gas?
Among halogens and noble gases, which have higher atomic radii?
Are noble gases soluble in water?
What type of interaction is responsible for the solubility of a noble gas in water?
Name the chemist who prepared the first compound of xenon.
Write the structure of the first compound of xenon.
What is the geometry of XeF2 molecule?
What type of hybridisation is involved in the formation of XeF2 molecule?
What is the shape of the molecule of following xenon compound?
XeF4
What is the shape of the molecule of following xenon compound?
XeF6
What is the shape of the molecule of following xenon compound?
XeO3
What is the shape of the molecule of following xenon compound?
XeOF4
Name the type of hybridisation involved in the formation of following xenon compound.
XeF4
What is the state of hybridisation of Xe in XeF6?
Name the type of hybridisation involved in the formation of following xenon compound.
XeOF4
What is the state of hybridisation of Xe in XeO3?
Write the formula of xenic acid.
Which property is involved in the separation of noble gases by Dewar’s method?
What is the oxidation state of phosphorus in H3PO3?
What is the oxidation state of phosphorus in the following:
PCl3
What is the oxidation state of phosphorus in the following:
Ca3P2
What is the oxidation state of phosphorus in the following:
Na3PO4
What is the oxidation state of phosphorus in the following:
POF3
What is the oxidation state of S in the following compound?
PbS
What is the oxidation state of S in the following compound?
SO2
What is the oxidation state of S in the following compound?
SF6
What is the oxidation state of S in the following compound?
Na2S2O3
What is the oxidation state of S in the following compound?
H2SO3
Find the oxidation state of the halogen in the following compound:
Cl2O
Find the oxidation state of the halogen in the following compound:
ClO2
Find the oxidation state of the halogen in the following compound:
KBrO3
Find the oxidation state of the halogen in the following compound:
NaClO4
Find the total number of lone pairs of electrons in N2O3.
Nootan solutions for केमिस्ट्रि पार्ट १ अँड २ [इंग्रजी] इयत्ता १२ आयएससी 7 p-Block Elements SHORT ANSWER TYPE QUESTIONS [Pages 447 - 449]
Nitrogen exists as a diatomic molecule, while the other elements of group 15 exist as polyatomic molecules. Explain.
Why are the atomic radii of group 15 elements smaller than those of the corresponding elements of group 14?
Why are the melting points of antimony and bismuth lower than the expected values?
Why are the ionisation energies of group 15 elements much higher than those of the corresponding elements of group 14?
How does the metallic character vary on moving down the group 15? Give suitable explanation for the observed order.
The trinegative ions (M3−) are formed only by N and P among group 15 elements. Explain.
Why does the stability of +5 oxidation state decrease on moving down the group 15?
PCl5 exists but NCl5 does not. Explain.
Write the structure of white phosphorus.
Write the structure of red phosphorus.
Giving suitable reasons, arrange the hydrides (MH3) of group 15 elements in the decreasing order of \[\ce{H - M - H}\] bond angle.
Discuss the structure of NH3 and explain why does it possess a shorter bond angle than the expected one.
Why does PH3 possess a smaller bond angle than that for NH3?
Why does the basic strength of the hydrides of group 15 elements decrease on moving down the group?
Among NH3, PH3, AsH3, SbH3 and BiH3, which does possess the highest reducing power and why?
Why does ammonia have a higher boiling point than that of phosphine?
Nitrogen does not form pentahalide.
Discuss the structure of PCl5.
Complete and balance the following equation:
\[\ce{N2H4 + Cu^{2+} ->}\]
Complete and balance the following equation:
\[\ce{N2H4 + KIO3 ->}\]
Complete and balance the following equation:
\[\ce{PCl3 + H2O ->}\]
Complete and balance the following equation:
\[\ce{BiCl3 + H2O ->}\]
Complete and balance the following chemical equation:
\[\ce{Pb(NO3)2 ->[Heat]}\]
Complete and balance the following equation:
\[\ce{P2O5 + HNO3 ->}\]
Name the oxides of group 15 elements having the group element in +3 oxidation states.
Name the oxides of group 15 elements having the group element in +4 oxidation state.
Write the structures of different oxides of nitrogen.
Why do the oxides of phosphorus and other heavier elements of group 15 have cage structures?
Write the structure of phosphorus trioxide.
Write the structure of phosphorus pentoxide.
Write the structures of different oxides of nitrogen.
Write the formula, structure and basicity of the following oxoacid:
Hypophosphorus acid
Write the formula, structure and basicity of the following oxoacid:
Cyclometaphosphoric acid
Write the formula, structure and basicity of the following oxoacid:
Orthophosphoric acid
Write the formula, structure and basicity of the following oxoacid:
Pyrophosphoric acid
Why does orthophosphoric acid form three types of salts? Explain its ionisation behaviour.
How is orthophosphoric acid prepared from rock phosphate?
Mention important uses of orthophosphoric acid.
Why does nitrogen differ from other elements of its group in its chemical behaviour?
Why are group 16 elements called chalcogens?
Why is polonium called so?
Why does oxygen exist as a gas, whereas other elements of the group are solids at room temperature?
Write the structure of a sulphur molecule.
Why are the melting and boiling points of polonium lower than those of selenium?
Why is the first ionisation energy of oxygen lower than that of nitrogen?
Why is it difficult to ionise group 16 elements?
The electron affinity of oxygen is lower than that of sulphur. Explain.
Why does the tendency of group 16 elements to exist in −2 oxidation state decrease on moving down the group?
Oxygen usually exists in −2 oxidation state but it exhibits +2 state in OF2. Explain.
Why do sulphur and other heavier elements of group 16 exhibit higher oxidation states?
Explain how sulphur exists in the +2 oxidation state.
Explain how sulphur exists in the +4 oxidation state.
Explain how sulphur exists in the +6 oxidation state.
How does the following property vary on moving down the group 16?
Atomic radii
How does the following property vary on moving down the group 16?
Metallic character
How does the following property vary on moving down the group 16?
Catenation
Discuss the important allotropic forms of sulphur.
Discuss the structure of H2O molecule.
Why does \[\ce{H - M - H}\] bond angle in the hydrides of group 16 elements decrease on going down the group?
Water is a liquid while all other hydrides of group 16 elements are gases at room temperature. Explain.
Giving suitable reasons, arrange the hydrides of group 16 elements in the decreasing order of their thermal stability.
Giving suitable reasons, arrange the hydrides of group 16 elements in the increasing order of their acidic character.
Why does water not act as a reducing agent?
Why should the compound F2O not be called as fluorine oxide? Write the correct representation and name of the compound.
How many types of halides are formed by sulphur? Write their general formulae and the oxidation states of sulphur in them.
Why is SF6 used as a gaseous insulator in high voltage generators?
Discuss the structure of SF6.
Discuss the structure:
SO2
Discuss the structure:
SO3
Write the structure of sulphur trioxide in the solid state.
Write the structure of selenium trioxide in the solid state.
Write the molecular formula and structure of sulphuric acid.
Write the formula and structure of the following oxoacid of sulphur:
Sulphurous acid
Write the formula and structure of the following oxoacid of sulphur:
Peroxodisulphuric acid
Write the molecular formula and structure of dithionic acid.
Why are the elements of group 17 called ‘halogens’?
Why does the state of agglomeration (the tendency of molecules to come closer) increase on moving down the group 17?
Why are the halogen atoms smallest in their respective periods?
Why does the density of halogens increase on moving down the group?
Why do melting and boiling points of halogens increase on moving down the group?
Why do halogens have very high ionisation energies?
Why do ionisation energies decrease in going from F to I in group 17?
Why do halogens possess very high values of electronegativity?
Arrange the halogens in the order of decreasing electronegativity.
Why do halogens have very high values of electron affinity?
The electron affinity of fluorine is unexpectedly lower than that of chlorine. Comment on the statement and explain.
Giving suitable reasons, arrange halogens in the decreasing order of electron affinity.
Why does iodine exhibit a slight metallic character?
Why does fluorine exhibit only −1 oxidation state in all of its compounds?
Taking the example of chlorine, show that all halogens (except fluorine) can exhibit +l, +3, +5 and +7 oxidation states.
Why does fluorine not exhibit higher oxidation states?
Explain the pale yellow colour of fluorine.
Explain violet colour of iodine.
How would you account for the very high reactivity of halogens?
Why do halogens act as oxidising agents?
A halogen of low atomic number can oxidise halide ions of higher atomic numbers. Comment on the statement and explain with suitable examples.
The following reaction is feasible or not feasible and why?
\[\ce{F2 + 2NaBr -> 2NaF + Br2}\]
The following reaction is feasible or not feasible and why?
\[\ce{Cl2 + 2NaF -> 2NaCl + F2}\]
The following reaction is feasible or not feasible and why?
\[\ce{Br2 + 2NaCl -> 2NaBr + Cl2}\]
The following reaction is feasible or not feasible and why?
\[\ce{I2 + 2NaCl -> 2NaI + Cl2}\]
The following reaction is feasible or not feasible and why?
\[\ce{Br2 + 2NaI -> 2NaBr + I2}\]
Giving suitable reasons, arrange metal halides \[\ce{(M - X)}\] in the decreasing order of their ionic character.
Hydrogen fluoride is a low boiling liquid, while all other hydrogen halides are gases at room temperature. Comment and explain.
Why does HF not show reducing properties, whereas other halogen halides do?
Why does the reducing power of hydrogen halides increase with increase in the atomic number of halogen?
Giving suitable reasons, arrange hydrogen halides in the increasing order of their acidic strength.
Write the structure of Cl2O.
Write the structure of ClO2.
Write the structure of Cl2O7.
What types of oxoacids are formed by halogens? Mention the oxidation states of halogens in them.
Write the formula and structure of hypofluorous acid.
Draw the structure of hypochlorous acid.
Complete and balance the following equation:
\[\ce{NaOH + Br2 ->[Cold]}\]
Complete and balance the following equation:
\[\ce{Na2O2 + ClO2 ->}\]
Complete and balance the following equation:
\[\ce{NaOH + Cl2 ->[Hot]}\]
Complete and balance the following equation:
\[\ce{NaClO3 + H2O ->[Electrolysis]}\]
What is bleaching powder?
How is bleaching powder prepared from slaked lime?
State two important uses of bleaching powder.
Giving suitable explanation, arrange hypohalous acids of chlorine, bromine and iodine in the increasing order of their acid strength.
Giving suitable explanation, arrange oxoacids of chlorine in the increasing order of their acid strength.
What are interhalogen compounds?
How are interhalogen compounds classified?
Discuss the formation and structure of XF3 (X = Cl, Br, I) molecule.
Write the structure of the following interhalogen compound:
ClF5
Also mention the state of hybridisation of Cl atom present in it.
What is the hybridisation of iodine in IF7? Give its structure.
Why does fluorine exhibit an anomalous behaviour as compared to the behaviour of other halogens?
What is basic iodine?
What are the evidences which support the existence of basic iodine?
What are pseudo halides and pseudo halogens and why are they called so? Give some examples.
The elements of group 18 should rightly be called as noble gases instead of inert gases. Comment on the statement and explain.
How would you justify the inclusion of group 18 in between group 17 and group 1 in the periodic table?
Write the names and electronic configurations of all the noble gases.
Why do noble gases exist as monoatomic molecules?
The atomic radii of noble gases are larger than those of the corresponding halogens. Explain.
Why are the melting and boiling points of noble gases much lower as compared to those of other substances of comparable molecular masses?
Why do noble gases have very high ionisation energies?
Giving suitable explanation, arrange noble gases in the increasing order of their solubility in water.
What type of interactions are responsible for the solubility of a noble gas in water? Explain.
Why does the solubility of noble gases in water increase with increase in the atomic number?
Among He, Ne, Ar, Kr and Xe, which gas is most easily liquefiable and why?
The electron gain enthalpy values of noble gases are large positive. Explain.
What type of compounds of noble gases were obtained prior to 1962?
When and how did the chemistry of noble gases begin?
Name the chemist who prepared the first compound of a noble gas.
Write the names and formulae of some important compounds of xenon.
Write the names and formulae of some important compounds of krypton.
Give the method of preparation of XeF2.
Give the methods of preparation of the following compound:
XeOF4
Give the methods of preparation of the following compound:
XeO3
Complete the following equation:
\[\ce{XeF2 + H2O ->}\]
Complete the equation:
\[\ce{XeF4 + H2O ->}\]
Complete the following reaction.
\[\ce{XeF6 + H2O ->}\]
Complete and balance the following equation:
\[\ce{XeF6 + NaF ->}\]
Complete and balance the following equation:
\[\ce{XeOF4 + H2O ->}\]
Complete and balance the following equation:
\[\ce{XeO3 + H2O ->}\]
Write the structures of fluorides of xenon.
Write the structure of XeOF4. Mention the state of hybridisation of xenon atom in it.
Write the structure of XeO3. Mention the state of hybridisation of xenon atom in it.
How would you account for the square pyramidal structure of XeOF4 molecule? Explain the formation of the molecule.
Explain why aluminium, though an electropositive metal, finds extensive use as a structural material.
Discuss the pattern of variation in the oxidation state of the following:
Al to Tl
Discuss the pattern of variation in the oxidation state of the following:
Si to Pb
Discuss the pattern of variation in the oxidation states of the following:
P to Bi
What is the importance of ultrapure elemental silicon?
How is ultrapure elemental silicon obtained?
How far do you agree with the phenomenon concept of inert pair effect? Justify your answer.
Compare the structures of white phosphorus, P4O6 and P4O10.
Nootan solutions for केमिस्ट्रि पार्ट १ अँड २ [इंग्रजी] इयत्ता १२ आयएससी 7 p-Block Elements LONG ANSWER TYPE QUESTIONS [Pages 449 - 450]
Discuss the following property and its variation in the group 15 elements:
Atomic radii
How does the metallic character vary in group 15?
Discuss the following property and its variation in the group 15 elements:
Oxidation states
Discuss the following property and its variation in the group 15 elements:
Allotropy
Discuss the following property and its variation in the group 15 elements:
Catenation
What is the inert pair effect?
How does the inert pair effect affect the properties of group 15 elements?
Discuss the preparation of the hydrides of group 15 elements.
Discuss the structures of the hydrides of group 15 elements.
Discuss the important characteristics of the hydrides of group 15 elements.
What types of halides are formed by group 15 elements? Discuss their structures and important characteristics.
How are the different oxides of nitrogen prepared?
Discuss general characteristics of different oxides of nitrogen.
Write the structures of different oxides of nitrogen.
Give a brief account of the oxoacids of nitrogen and write their structures.
Give a brief account of the oxoacids of phosphorus and write their structures.
Mention some important methods of preparation of orthophosphoric acid.
Mention some properties of orthophosphoric acid.
In what ways does nitrogen differ from the other elements of group 15?
What are the causes for the anomalous behaviour of nitrogen?
Why are group 16 elements called chalcogens?
Discuss the important characteristics of group 16 elements.
Compare the elements of group 15 with those of group 16 in relation to the following property:
Atomic radii
Compare the elements of group 15 with those of group 16 in relation to the following property:
Ionisation energy
Compare the elements of group 15 with those of group 16 in relation to the following property:
Electronegativity
Compare the elements of group 15 with those of group 16 in relation to the following property:
Electron affinity
Compare the elements of group 15 with those of group 16 in relation to the following property:
Oxidation states
With suitable explanation, give a brief account of the oxidation states exhibited by the elements of group 16.
With suitable explanation, give a brief account of the allotropy exhibited by the elements of group 16.
Discuss the structures of the hydrides of group 16 elements.
Discuss the important characteristics of the hydrides of group 16 elements.
Give a brief account of the halides of group 16 elements.
Give a brief account of the oxides of group 16 elements.
Draw the structure of SF4 molecule.
Discuss the structure of SF6.
Discuss the structure:
SO2
Discuss the structure:
SO3
Give a brief account of the oxoacids of sulphur.
Give a brief account of the oxoacids of selenium.
Give a brief account of the oxoacids of tellurium.
Write the formula and structure of the following oxoacid of sulphur:
Sulphurous acid
Draw the structure of peroxymonosulphuric acid.
Write the molecular formula and structure of thiosulphuric acid.
Write the molecular formula and structure of dithionic acid.
Discuss the anomalous behaviour of oxygen in relation to the properties of group 16 elements.
What are halogens and why are they called so?
Give the important physical characteristics of halogens.
Give the important chemical characteristics of halogens.
With suitable explanations, give a brief account of the oxidation states exhibited by halogens.
Why are halogens coloured?
Discuss the following property and its variation with reference to the elements of group 17:
Reactivity
Discuss the following property and its variation with reference to the elements of group 17:
Oxidising nature
Discuss the following property and its variation with reference to the elements of group 17:
Acidic nature of oxoacids
How are hydrogen halides prepared?
Discuss the structures of halides.
Mention some of the important characteristics with suitable explanation of hydrogen halides.
What type of oxoacids are formed by halogens?
Give a brief account of the preparation of the oxoacids of chlorine.
Give a brief account of the important properties of the oxoacids of chlorine.
Give a brief account of the acidic nature of the oxoacids of chlorine.
What are interhalogen compounds?
How are interhalogen compounds classified?
Write any two characteristics of interhalogen compounds.
Give a brief account of the structures of interhalogen compounds.
Write any two characteristics of interhalogen compounds.
Give a brief account of the structures of interhalogen compounds.
In what ways does fluorine differ from other halogens?
Discuss four points of anomalous behavior of fluorine.
What are noble gases and why are they called so?
Mention the important physical characteristics of noble gases.
Discuss the following property with reference to noble gases:
Chemical reactivity
Discuss the following property with reference to noble gases:
Atomicity
Discuss the following property with reference to noble gases:
Atomic radii
Discuss the following property with reference to noble gases:
Melting point
Discuss the following property with reference to noble gases:
Boiling point
Discuss the following property with reference to noble gases:
Ionisation energy
Discuss the following property with reference to noble gases:
Solubility in water
Discuss the following property with reference to noble gases:
Liquefaction tendency
Give a brief account of the chemistry of noble gases, with a special reference to the fluorides and oxyfluorides of xenon.
Give the methods of preparation of XeF4.
Give the properties of the following compound:
XeF4
Draw the structure of the following molecule:
XeF4
Give the methods of preparation of the following compound:
XeOF4
Give the properties of the following compound:
XeOF4
Write the structures of the following molecule:
XeOF4
Give the methods of preparation of the following compound:
XeF6
Give the properties of the following compound:
XeF6
Draw structures of XeF6.
Give the methods of preparation of the following compound:
XeO3
Give the properties of the following compound:
XeO3
Draw the structure of XeO3
What are the important ores of aluminium?
How is the metal extracted from the bauxite ore?
How does tin occur in nature?
Give a brief account of the extraction of tin from its ores.
Mention the important alloys of tin.
What are the important ores of lead?
How is the metal extracted from lead ores?
Mention some important properties of lead.
How is phosphine prepared in the laboratory?
Discuss important properties of phosphine.
Discuss the uses of phosphine.
Discuss the structure of phosphine.
Give the preparation of the following:
PCl3
Give the properties of the following:
PCl3
Give the uses of the following:
PCl3
Give the preparation of the following:
PCl5
Give the properties of the following:
PCl5
Give the uses of the following:
PCl5
How does sulphur occur in nature?
Give a brief account of the extraction of sulphur.
What is the effect of heat on sulphur?
How does phosphorus occur in nature?
Describe the preparation of white phosphorus.
Describe the preparation of red phosphorus.
Describe the manufacturing of H2SO4 by the contact process.
Give a brief account of the preparation of the oxoacids of chlorine.
Give a brief account of the important properties of the oxoacids of chlorine.
Give the important uses of oxoacids of chlorine.
How are noble gases isolated from air?
Describe the important uses of noble gases isolated from air.
Using VSEPR theory, predict the probable structure of \[\ce{SO^{2-}_3}\].
Using VSEPR theory, predict the probable structure of \[\ce{IF^-_6}\].
Using VSEPR theory, predict the probable structure of XeF2.
Using VSEPR theory, predict the probable structure of \[\ce{ClO^-_4}\].
Using VSEPR theory, predict the probable structure of \[\ce{ICl^-_4}\].
Using VSEPR theory, predict the probable structure of \[\ce{IBr^-_2}\].
Describe the shape of the following species:
SiF4
Describe the shape of the following species:
\[\ce{SiF^{2-}_6}\]
Describe the shape of the following species:
PF5
Nootan solutions for केमिस्ट्रि पार्ट १ अँड २ [इंग्रजी] इयत्ता १२ आयएससी 7 p-Block Elements INTEGER TYPE QUESTION [Page 450]
The reaction of Xe and O2F2 gives a Xe compound P. The number of moles of HF produced by the complete hydrolysis of 1 mol of P is ______. (nearest integer)
Nootan solutions for केमिस्ट्रि पार्ट १ अँड २ [इंग्रजी] इयत्ता १२ आयएससी 7 p-Block Elements OBJECTIVE (MULTIPLE CHOICE) TYPE QUESTIONS [Pages 450 - 455]
NH3 has much higher boiling point than PH3, because ______.
NH3 has a much higher molecular mass.
NH3 forms hydrogen bonds.
NH3 contains ionic bonds while PH3 contains covalent bonds.
NH3 undergoes umbrella inversion.
Which of the following oxides of nitrogen is the anhydride of nitrous acid?
NO
N2O3
N2O4
N2O5
PCl5 exists but NCl5 does not because ______.
nitrogen has no vacant d-orbitals.
NCl5 is unstable.
N2 is inert.
None of the above.
Which hydride is most stable?
AsH3
SbH3
N2O4
NH3
The largest bond angle is in ______.
NH3
PH3
AsH3
BiH3
NH3 molecule can enter into complex formation through ______.
ionic bond
covalent bond
coordinate bond
electron-deficient bond
NF3 is less polar than NH3 because ______.
F is more reactive than H.
NH3 forms associated molecules.
The resultant of bond polarity is less.
The resultant of the individual polarities is opposed by the polarity of lone pair.
The oxoacid of phosphorus in which phosphorus has the lowest oxidation state is ______.
hypophosphorus acid
orthophosphoric acid
pyrophosphoric acid
metaphosphoric acid
Which of the following nitrogen halides is most stable?
NI3
NBr3
NCl3
NF3
A white precipitate is obtained on hydrolysis of ______.
PCl5
NCl3
BiCl3
AsCl3
Structure of SF4 is ______.
octahedral
bipyramidal
square planar
tetrahedral
Which one has the highest bond energy?
O-O
S-S
Se-Se
Te-Te
H2S is more acidic than H2O although sulphur is less electronegative than oxygen. This is because ______.
water is a highly associated compound.
H-S bond is weaker than H-O bond.
the molecular weight of H2S is more than that of H2O.
H2S is a gas while H2O is a liquid.
Which has sp2 hybridisation?
CO2
N2O
SO2
CO
The maximum covalency of sulphur is ______.
2
4
6
8
Oxygen exhibits −1 oxidation state in ______.
OF2
H2O
H2O2
HCIO
The hybridisation state of S in SF6 is ______.
sp3d
sp3d2
d2sp3
sp3d3
Which one of the following has lowest boiling point?
H2O
H2S
H2Se
H2Te
Ozone is ______.
a compound of oxygen.
an allotrope of oxygen.
an isotope of oxygen.
an isobar of oxygen.
Which one is the strongest reducing agent?
HF
HCl
HBr
HI
Which one is the strongest oxidising agent?
HCIO
HCIO2
HCIO3
HCIO4
Among the following, the pseudo halide is ______.
CN−
ICl
\[\ce{I^-_3}\]
IF5
Fluorine can exist in the oxidation states ______.
−1 only
−1 and +1 only
−1, +1 and +3 only
−1, +1, +3, +5 and +7
The most powerful oxidising agent is ______.
fluorine
chlorine
bromine
iodine
Iodine can form the ions ______.
I−
I+
I3+
All of these
The following acids have been arranged in the order of decreasing acid strength.
CIOH (I) BrOH (II) IOH (III)
Identify the correct order.
I > II > III
II > I > III
III > II > I
I > III > II
If one litre of air is passed repeatedly on hot copper and hot magnesium till no further decrease in volume takes place, the volume of residual gas would be about ______.
200 mL
100 mL
10 mL
zero
Which one of the following fluorides does not exist?
HeF4
XeF4
CF4
SF6
The structure of XeF6 is ______.
distorted octahedral
pyramidal
tetrahedral
trigonal bipyramidal
The first noble gas compound obtained was ______.
XeF2
XeF4
XePtF5
XeOF4
XeF6 on complete hydrolysis gives ______.
Xe
XeO2
XeO3
XeO4
Geometry of XeOF4 molecule is ______.
square planar
square pyramidal
triangular pyramidal
octahedral
The ionic radii (in Å) of N3−, O2− and F− are respectively ______.
1.36, 1.40 and 1.71
1.36, 1.71 and 1.40
1.71, 1.40 and 1.36
1.71, 1.36 and 1.40
Which among the following is the most reactive?
Cl2
Br2
I2
ICl
Match the catalysts to the correct processes:
| Catalysts | Process | ||
| (A) | TiCl4 | (i) | Wacker process |
| (B) | PdCl2 | (ii) | Ziegler-Natta polymerization |
| (C) | CuCl2 | (iii) | Contact process |
| (D) | V2O5 | (iv) | Deacon’s process |
(A) - (ii), (B) - (iii), (C) - (iv), (D) - (i)
(A) - (iii), (B) - (i), (C) - (ii), (D) - (iv)
(A) - (iii), (B) - (ii), (C) - (iv), (D) - (i)
(A) - (ii), (B) - (i), (C) - (iv), (D) - (iii)
Which one has the highest boiling point?
He
Ne
Kr
Xe
Assertion: Nitrogen and oxygen are the main components in the atmosphere but these do not react to form oxides of nitrogen.
Reason: The reaction between nitrogen and oxygen requires high temperature.
The assertion is incorrect, but the reason is correct.
Both the assertion and reason are incorrect.
Both assertion and reason are correct, and the reason is the correct explanation for the assertion.
Both assertion and reason are correct, but the reason is not the correct explanation for the assertion.
The species in which the N atom is in a state of sp hybridization is ______.
\[\ce{NO^+_2}\]
\[\ce{NO^-_2}\]
\[\ce{NO^-_2}\]
NO2
The reaction of zinc with dilute and concentrated nitric acid, respectively, produces ______.
N2O and NO2
NO2 and NO
NO and N2O
NO2 and N2O
The pair in which phosphorous atoms have a formal oxidation state of +3 is ______.
orthophosphorous and pyrophosphorous acids
pyrophosphorous and hypophosphoric acids
orthophosphorous and hypophosphoric acids
pyrophosphorous and pyrophosphoric acids
The reagent(s) that can selectively precipitate S2− from a mixture of S2− and \[\ce{SO^{2-}_4}\] in aqueous solution is (are) ______.
CuCl2
BaCl2
Pb(OOCCH3)2
Na2[Fe(CN)5NO]
Consider the molecules CH4, NH3 and H2O. Which of the given statement is false?
The H-C-H bond angle in CH4, the H-N-H bond angle in NH3, and the H-O-H bond angle in H2O are all greater than 90°.
The H-O-H bond angle in H2O is larger than the H-C-H bond angle in CH4.
The H-O-H bond angle in H2O is smaller than the H-N-H bond angle in NH3.
The H-C-H bond angle in CH4 is larger than the H-N-H bond angle in NH3.
Among the following the correct order of acidity is:
HClO2 < HClO < HClO3 < HClO4
HClO4 < HClO2 < HClO < HClO3
HClO3 < HClO4 < HClO2 < HClO
HClO < HClO2 < HClO3 < HClO4
When copper is heated with conc. HNO3, it produces ______.
Cu(NO3)2 and NO2
Cu(NO3)2 and NO
Cu(NO3)2, NO and NO2
Cu(NO3)2 and N2O
Which one of the following orders is correct for the bond dissociation enthalpy of halogen molecules?
Br2 > I2 > F2 > Cl2
F2 > Cl2 > Br2 > l2
I2 > Br2 > Cl2 > F2
Cl2 > Br2 > F2 > I2
The product obtained as a result of a reaction of nitrogen with CaC2 is ______.
Ca(CN)2
CaCN
CaCN3
Ca2CN
Match the compounds given in column I with the hybridisation and shape given in column II and mark the correct option.
| Column I | Column II | ||
| (A) | XeF6 | (i) | Distorted octahedral |
| (B) | XeO3 | (ii) | Square planar |
| (C) | XeOF4 | (iii) | Pyramidal |
| (D) | XeF4 | (iv) | Square pyramidal |
(A) (B) (C) (D) (i) (iii) (iv) (ii) (A) (B) (C) (D) (i) (ii) (iv) (iii) (A) (B) (C) (D) (iv) (iii) (i) (ii) (A) (B) (C) (D) (iv) (i) (ii) (iii)
Which is the correct statement for the given acids?
Phosphinic acid is a diprotic acid while phosphonic acid is a monoprotic acid.
Phosphinic acid is a monoprotic acid while phosphonic acid is a diprotic acid.
Both are triprotic acids.
Both are diprotic acids.
The species, having bond angles of 120° is ______.
PH3
CIF3
NCl3
BCl3
In which of the pair of ions, both species contain S-S bond?
\[\ce{S4O^{2-}_6, S2O^{2-}_3}\]
\[\ce{S2O^{2-}_7, S2O^{2-}_8}\]
\[\ce{S4O^{2-}_6, S2O^{2-}_7}\]
\[\ce{S2O^{2-}_7, S2O^{2-}_3}\]
Match the interhalogen compounds of column I with the geometry in column II and assign the correct code.
| Column I | Column II | ||
| (1) | XX' | (i) | T-shape |
| (2) | \[\ce{XX^'_3}\] | (ii) | Pentagonal bipyramidal |
| (3) | \[\ce{XX^'_5}\] | (iii) | Linear |
| (4) | \[\ce{XX^'_7}\] | (iv) | Square-pyramidal |
| (v) | Tetrahedral |
(1) (2) (3) (4) (iii) (iv) (i) (ii) (1) (2) (3) (4) (iii) (i) (iv) (ii) (1) (2) (3) (4) (v) (iv) (iii) (ii) (1) (2) (3) (4) (iv) (iii) (ii) (i)
Name the gas that can readily decolourises acidified KMnO4 solution.
CO2
SO2
NO2
P2O5
Which of the following species is not paramagnetic?
O2
B2
NO
CO
The products obtained when chlorine gas reacts with cold and dilute aqueous NaOH are ______.
ClO− and \[\ce{ClO^-_3}\]
\[\ce{ClO^-_2}\] and \[\ce{ClO^-_3}\]
Cl− and ClO−
Cl− and \[\ce{ClO^-_2}\]
The compound that does not produce nitrogen gas by the thermal decomposition is:
Ba(N3)2
(NH4)2Cr2O7
NH4NO2
(NH4)2SO4
When Metal ‘M’ is treated with NaOH, a white gelatinous precipitate ‘X’ is obtained, which is soluble in excess of NaOH. Compound ‘X’ when heated strongly gives an oxide which is used in chromatography as an adsorbent. The metal ‘M’ is ______.
Zn
Ca
Al
Fe
The correct order of N-compounds in its decreasing order of oxidation states is ______.
HNO3, NH4Cl, NO, N2
HNO3, NO, NH4Cl, N2
HNO3, NO, N2, NH4Cl
NH4Cl, N2, NO, HNO3
Which of the following statements is not true for halogens?
All but fluorine show positive oxidation states.
All are oxidizing agents.
All form monobasic oxyacids.
Chlorine has the highest electron gain enthalpy.
In the structure of CIF3, the number of lone pair of electrons on central atom ‘Cl’ is ______.
four
two
one
three
Which oxide of nitrogen is not a common pollutant introduced into the atmosphere both due to natural and human activity?
N2O
NO2
N2O5
NO
The oxoacid of sulphur that does not contain bond between sulphur atoms is ______.
H2S2O7
H2S2O3
H2S4O6
H2S2O4
The noble gas that does not occur in the atmosphere is:
Ne
He
Kr
Ra
The number of pentagons in C60 and trigons (triangles) in white phosphorus, respectively, are:
12 and 3
20 and 3
20 and 4
12 and 4
An organic compound ‘A’ is oxidized with Na2O2 followed by boiling with HNO3. The resultant solution is then treated with ammonium molybdate to yield a yellow precipitate. Based on above observation, the element present in the given compound is ______.
fluorine
phosphorus
nitrogen
sulphur
The group number, number of valence electrons, and valency of an element with atomic number 15, respectively, are ______.
16, 6 and 3
15, 6 and 12
16, 5 and 2
15, 5 and 3
Which of the following is paramagnetic?
O2
N2
H2
Li2
Identify the correct formula of 'oleum' from the following:
H2S2O8
H2S2O7
H2SO3
H2SO4
Which of the following cannot act both as Bronsted acid and as Bronsted base?
\[\ce{HSO^-_4}\]
\[\ce{HCO^-_3}\]
NH3
HCl
The correct structure of tribromooctaoxide is:
The number of moles of hydrogen molecules required to produce 20 moles of ammonia through Haber’s process is ______.
10
20
30
40
Identify the incorrect statement related to PCl5 from the following:
Three equatorial P-Cl bonds make an angle of 120° with each other.
Two axial P-Cl bonds make an angle of 180° with each other.
Axial P-Cl bonds are longer than equatorial P-Cl bonds.
PCl5 molecule is non-reactive.
Match the xenon compounds in Column-I with its structure in Column-II and assign the correct code:
| Column-I | Column-II | ||
| (1) | XeF4 | (i) | Pyramidal |
| (2) | XeF6 | (ii) | Square planar |
| (3) | XeOF4 | (iii) | Distorted octahedral |
| (4) | XeO3 | (iv) | Square pyramidal |
(1) (2) (3) (4) (i) (ii) (iii) (iv) (1) (2) (3) (4) (ii) (iii) (iv) (i) (1) (2) (3) (4) (ii) (iii) (i) (iv) (1) (2) (3) (4) (iii) (iv) (i) (ii)
Which is the correct thermal stability order for H2E (E = 0, S, Se, Te and Po)?
H2S < H2O < H2Se < H2Te < H2Po
H2O < H2S < H2Se < H2Te < H2Po
H2Po < H2Te < H2Se < H2S < H2O
H2Se < H2Te < H2Po < H2O < H2S
Match the following.
| Column I | Column II |
| A) Pure nitrogen | (i) Chlorine |
| B) Haber process | (ii) Sulphuric acid |
| C) Contact process | (iii) Ammonia |
| D) Deacons Process | (iv) sodium azide (or) Barium azide |
Which of the following is the correct option?
A B C D (i) (ii) (iii) (iv) A B C D (ii) (iv) (i) (iii) A B C D (iii) (iv) (ii) (i) A B C D (iv) (iii) (ii) (i)
Which of the following oxoacid of sulphur has -O-O- linkage?
H2SO3, sulphurous acid
H2SO4, sulphuric acid
H2S2O8, peroxodisulphuric acid
H2S2O7, pyrosulphuric acid
Match the following:
| Oxide | Nature | ||
| (1) | CO | (i) | Basic |
| (2) | BaO | (ii) | Neutral |
| (3) | Al2O3 | (iii) | Acid |
| (4) | Cl2O7 | (iv) | Amphoteric |
Which of the following is correct option?
(1) (2) (3) (4) (i) (ii) (iii) (iv) (1) (2) (3) (4) (ii) (i) (iv) (iii) (1) (2) (3) (4) (iii) (iv) (i) (ii) (1) (2) (3) (4) (iv) (iii) (ii) (i)
While titrating dilute HCl solution with aqueous NaOH, which of the following will not be required?
Clamp and phenolphthalein
Surette and porcelain tile
Bunsen burner and measuring cylinder
Pipette and distilled water
The statement that is not true about ozone is:
In the stratosphere, CFCs release chlorine free radicals (Cl) which reacts with O3 to give chlorine dioxide radicals.
In the stratosphere, it forms as protective shield against UV radiation.
It is a toxic gas and its reaction with NO gives NO2.
In the atmosphere, it is depleted by CFCs.
In a molecule of pyrophosphoric acid, the number of P-OH, P=O and P-O-P bonds/moiety(ies) respectively are ______.
4, 2 and 1
3, 3 and 3
4, 2 and 0
2, 4 and 1
If the boiling point of H2O is 373 K, the boiling point of H2S will be ______.
equal to 373 K.
greater than 300 K but less than 373 K.
less than 300 K.
more than 373 K.
Aqua regia is used for dissolving noble metals (Au, Pt, etc.). The gas evolved in this process is ______.
N2
NO
N2O5
N2O3
The reacition in which the hybridisation of the underlined atom is affected is:
\[\ce{H2\underline{\text{S}}O4 + NaCl ->[420 K]}\]
\[\ce{\underline{\text{N}}H3 ->[H+]}\]
\[\ce{H3\underline{\text{P}}O2 ->[Disproportionation]}\]
\[\ce{XeF4 + SbF5 ->}\]
The structure of PCl5 in the solid state is ______.
tetrahedral [PCl4]+ and octahedral [PCl6]−
trigonal bipyramidal
square planar [PCl4]+ and octahedral [PCl6]−
square pyramidal
Noble gases are named because of their inertness towards reactivity. Identify an incorrect statement about them.
Noble gases have large positive values of electron gain enthalpy.
Noble gases are sparingly soluble in water.
Noble gases have very high melting and boiling points.
Noble gases have weak dispersion forces.
Match List-I with List-II.
| List-I | List-II | ||
| (A) | PCl5 | (i) | Square pyramidal |
| (B) | SF6 | (ii) | Trigonal planar |
| (C) | BrF5 | (iii) | Octahedral |
| (D) | BF3 | (iv) | Trigonal bipyramidal |
Choose the correct answer from the options given below.
(A) - (iv), (B) - (iii), (C) - (ii), (D) - (i)
(A) - (iv), (B) - (iii), (C) - (i), (D) - (ii)
(A) - (ii), (B) - (iii), (C) - (iv), (D) - (i)
(A) - (iii), (B) - (i), (C) - (iv), (D) - (ii)
In which one of the following arrangements the given sequence is not strictly according to the properties indicated against it?
CO2 < SiO2 < SnO2 < PbO2 : Increasing oxidizing power
HF < HCl < HBr < HI : Increasing acidic strength
H2O < H2S < H2Se < H2Te : Increasing pKa values
NH3 < PH3 < AsH3 < SbH3 : Increasing acidic character
Statement I: Acid strength increases in the order given as HF << HCl << HBr << HI.
Statement II: As the size of the elements F, Cl, Br, and I increase down the group, the bond strength of HF, HCl, HBr and HI decreases and so the acid strength increases.
In the light of the above statements, choose the correct answer from the options given below.
Statement I is incorrect but Statement II is true.
Both Statement I and Statement II are true.
Both Statement I and Statement II are false
Statement I is correct but statement II is false.
Match List-I with List-II.
| List-I (Name of oxo acid) |
List-II Oxidation state of ‘P’ |
||
| (A) | Hypophosphorous acid | (i) | +5 |
| (B) | Orthophosphoric acid | (ii) | +4 |
| (C) | Hypophosphoric acid | (iii) | +3 |
| (D) | Orthophosphorous acid | (iv) | +2 |
| (v) | +1 |
Choose the correct answer from the options given below
A - (v), B - (i), C - (ii), D - (iii)
A - (iv), B - (i), C - (ii), D - (iii)
A - (iv), B - (v), C - (ii), D - (iii)
A - (v), B - (iv), C - (ii), D - (iii)
A group 15 element, which is a metal and forms a hydride with strongest reducing power among group 15 hydrides. The element is ______.
Bi
As
p
Sb
The set that represents the pair of neutral oxides of nitrogen is ______.
NO and N2O
N2O and N2O3
N2O and NO2
NO and NO2
The oxidation states of nitrogen in NO, NO2, N2O and \[\ce{NO^-_3}\] are in the order of:
\[\ce{NO^-_3}\] > NO2 > NO > N2O
NO2 > \[\ce{NO^-_3}\] > NO > N2O
N2O > NO2 > NO > \[\ce{NO^-_3}\]
NO > NO2 > N2O > \[\ce{NO^-_3}\]
Fex2 and Fey3 are known when x and y are ______.
x = F, Cl, Br, I and y = F, Cl, Br
x = F, Cl, Br and y = F, Cl, Br, I
x = Cl, Br, I and y = F, Cl, Br, I
x = F, Cl, Br, I and y = F, Cl, Br, I
The correct statement(s) related to oxoacids of phosphorous is (are):
Upon heating H3PO3 undergoes disproportionation reaction to produce H3PO4 and PH3.
White H3PO3 can act as reducing agent, H3PO4 can not.
H3PO3 is a monobasic acid.
The H atom of P-H bond in H3PO3 is not ionizable in water.
The number of ioinisable hydrogens present in the product obtained from a reaction of phosphorus trichloride and phosphoric acid is ______.
3
0
2
1
Statement I: The boiling points of the following hydrides of group 16 elements increases in the order:
H2O < H2S < H2Se < H2Te
Statement II: The boiling points of these hydrides increase with increase in molar mass.
In the light of above statements, choose the most appropriate answer from the options given below:
Both statement I and statement II are incorrect.
Statement I is correct but statement II is incorrect.
Statement I is incorrect but statement II is correct.
Both statement I and statement II are correct.
Amongst the following which one will have maximum lone pair-lone pair electron repulsions?
IF5
SF4
XeF2
ClF3
Heating white phosphorus with conc. NaOH solution gives mainly ______.
Na3P and H2O
H3PO and NaH
P(OH)3 and NaH2PO4
PH3 and NaH2PO2
Match List-I with List-II by matching the gas evolved during each reaction.
| List-I | List-II | ||
| (A) | \[\ce{(NH4)2Cr2O7 ->[\Delta]}\] | (i) | H2 |
| (B) | \[\ce{KMnO4 + HCl ->}\] | (ii) | N2 |
| (C) | \[\ce{Al + NaOH + H2O ->}\] | (iii) | O2 |
| (D) | \[\ce{NaNO3 ->[\Delta]}\] | (iv) | Cl2 |
Choose the correct answer from the options given below:
A - (ii), B - (iii), C - (i), D - (iv)
A - (iii), B - (i), C - (iv), D - (ii)
A - (ii), B - (iv), C - (i), D - (iii)
A - (iii), B - (iv), C - (i), D - (ii)
White phosphorus reacts with thionyl chloride to give ______.
PCl5, SO2 and S2Cl2
PCl3, SO2 and S2Cl2
PCl3, SO2 and Cl2
PCl5, SO2 and Cl2
Concentrated HNO3 reacts with iodine to give ______.
HI, NO2 and H2O
HIO2, N2O and H2O
HIO3, NO2 and H2O
HIO4, N2O and H2O
The treatment of galena with HNO3 produces a gas that is ______.
Paramagnetic
Bent in geometry
an acidic oxide
Colourless
The compound(s) which react(s) with NH3 to give boron nitride (BN) is (are):
B
B2H6
B2O3
HBF4
The correct option(s) related to the extraction of iron from its ore in the blast furnace operating in the temperature range 900-1500 K is (are):
Limestone is used to remove silicate impurity.
Pig iron obtained from blast furnace contains about 4% carbon.
Coke (C) converts CO2 to CO.
Exhaust gases consist of NO2 and CO.
Nootan solutions for केमिस्ट्रि पार्ट १ अँड २ [इंग्रजी] इयत्ता १२ आयएससी 7 p-Block Elements FILL IN THE BLANKS TYPE QUESTIONS [Pages 456 - 457]
The elements of group 15 possess a ______ np subshell.
On moving down the group 15, metallic character ______.
Nitrogen exhibits all oxidation states from ______ to ______.
PH3 molecule is formed by ______ hybridisation and has H-P-H bond angle equal to ______.
Among the hydrides of group 15 elements, ______ is most stable and most basic.
Among the hydrides of group 15 elements, the strongest reducing agent is ______.
Hydrazine burns in oxygen to give ______ and ______.
The trihalides of group 15 elements are predominantly ______ and have ______ structures.
PCl5 is formed by ______ hybridisation and is ______ in shape.
Sb4O6 is ______ in nature, while Bi2O3 is ______.
Hyponitrous acid has the formula ______ and its basicity is ______.
The oxidation states of phosphorus in hypophosphorus acid and pyrophosphoric acid are ______ and ______ respectively.
Orthophosphoric acid is a ______ basic acid and forms ______ types of salts.
The density of group 16 elements ______ on moving down the group.
The first ionisation energy of S is ______ than that of P.
The electron affinity of oxygen is ______ than that of S.
The allotropic forms of sulphur can be grouped into ______ classes.
The low volatility of water as compared to the other hydrides of group 16 elements is due to the ______ of H2O molecules through ______.
SF4 molecule involves ______ hybridisation and is ______ in shape.
The electron affinity of fluorine is ______ than that of chlorine.
Fluorine molecules absorb ______ light and appear ______, whereas iodine molecules absorb ______ light and show ______ colour.
The high reactivity of halogens is due to their ______ dissociation energy and ______ electron affinity.
Bromine can displace only ______ from its salt solutions.
Among the hydrogen halides, the strongest reducing agent is ______.
The acid strength of different oxoacids of a halogen increases with ______ in the oxidation number of the halogen.
In a interhalogen compound XYn, X is always ______ halogen and exists in a ______ oxidation state.
Noble gases are soluble in water due to ______ interaction.
The electron affinities of noble gases are nearly equal to ______ because they possess stable ______ electronic configurations.
The first chemical compound of a noble gas prepared in the laboratory was ______. It was prepared by ______ in ______.
XeF2 is a colourless ______ and is stable when ______ and ______.
XeF6 is prepared by heating a mixture of Xe and F2 in the ratio ______ at ______ K under a pressure of ______ atm.
XeOF4 involves ______ hybridisation and is ______ in shape.
Nootan solutions for केमिस्ट्रि पार्ट १ अँड २ [इंग्रजी] इयत्ता १२ आयएससी 7 p-Block Elements ASSERTION-REASON TYPE QUESTIONS [Page 457]
Assertion: The oxoacids of phosphorus containing P-H bonds are strong reducing agents.
Reason: The P-H bonds present in these acids dissociate to give H+ ions.
If both Assertion and Reason are CORRECT and Reason is the CORRECT explanation of the Assertion.
If both Assertion and Reason are CORRECT but Reason is not the CORRECT explanation of the Assertion.
If Assertion is CORRECT but Reason is INCORRECT.
If Assertion is INCORRECT but Reason is CORRECT.
Assertion: Nitric acid turns yellow on long standing.
Reason: It slowly decomposes to give NO2 which gets dissolved in it.
If both Assertion and Reason are CORRECT and Reason is the CORRECT explanation of the Assertion.
If both Assertion and Reason are CORRECT but Reason is not the CORRECT explanation of the Assertion.
If Assertion is CORRECT but Reason is INCORRECT.
If Assertion is INCORRECT but Reason is CORRECT.
Assertion: The O-O bond length in ozone is identical with that in molecular oxygen.
Reason: The ozone molecule is a resonance hybrid of two canonical structures.
If both Assertion and Reason are CORRECT and Reason is the CORRECT explanation of the Assertion.
If both Assertion and Reason are CORRECT but Reason is not the CORRECT explanation of the Assertion.
If Assertion is CORRECT but Reason is INCORRECT.
If Assertion is INCORRECT but Reason is CORRECT.
Assertion: The shape of XeOF2 molecule is T-shaped.
Reason: It is formed by sp3d2 hybridisation of Xe atom.
If both Assertion and Reason are CORRECT and Reason is the CORRECT explanation of the Assertion.
If both Assertion and Reason are CORRECT but Reason is not the CORRECT explanation of the Assertion.
If Assertion is CORRECT but Reason is INCORRECT.
If Assertion is INCORRECT but Reason is CORRECT.
Assertion: HI can not be prepared by the action of cone. H2SO4 on Kl.
Reason: HI is more volatile than H2SO4.
If both Assertion and Reason are CORRECT and Reason is the CORRECT explanation of the Assertion.
If both Assertion and Reason are CORRECT but Reason is not the CORRECT explanation of the Assertion.
If Assertion is CORRECT but Reason is INCORRECT.
If Assertion is INCORRECT but Reason is CORRECT.
Assertion: Higher noble gases are soluble in water.
Reason: Noble gases are monoatomic in nature.
If both Assertion and Reason are CORRECT and Reason is the CORRECT explanation of the Assertion.
If both Assertion and Reason are CORRECT but Reason is not the CORRECT explanation of the Assertion.
If Assertion is CORRECT but Reason is INCORRECT.
If Assertion is INCORRECT but Reason is CORRECT.
Assertion: ICl is more reactive than I2.
Reason: I-Cl bond is weaker than I-I bond.
If both Assertion and Reason are CORRECT and Reason is the CORRECT explanation of the Assertion.
If both Assertion and Reason are CORRECT but Reason is not the CORRECT explanation of the Assertion.
If Assertion is CORRECT but Reason is INCORRECT.
If Assertion is INCORRECT but Reason is CORRECT.
Nootan solutions for केमिस्ट्रि पार्ट १ अँड २ [इंग्रजी] इयत्ता १२ आयएससी 7 p-Block Elements 'NCERT TEXT-BOOK' Exercises [Pages 457 - 460]
Discuss the general characteristics of Group 15 elements with reference to their electronic configuration.
Discuss the general characteristics of Group 15 elements with reference to their oxidation state.
Discuss the general characteristics of Group 15 elements with reference to their atomic size.
Discuss the general characteristics of Group 15 elements with reference to their ionisation enthalpy.
Discuss the general characteristics of Group 15 elements with reference to their electronegativity.
Why does the reactivity of nitrogen differ from phosphorus?
Discuss the trends in chemical reactivity of group 15 elements.
Why does NH3 form hydrogen bond but PH3 does not?
How is nitrogen prepared in the laboratory? Write the chemical equations of the reactions involved.
How is ammonia manufactured industrially?
Illustrate how copper metal can give different products on reaction with HNO3.
Write the resonance structures for NO2.
Give the resonating structure of N2O5.
The HNH angle value is higher than HPH, HAsH and HSbH angles. Why?
Why does R3P=O exist but R3N=O does not (R = alkyl group)?
Explain why NH3 is basic while BiH3 is only feebly basic.
Nitrogen exists as diatomic molecule and phosphorus as P4. Why?
Write main differences between the properties of white phosphorus and red phosphorus.
Why does nitrogen show catenation properties less than phosphorus?
Give the disproportionation reaction of H3PO3
Can PCl5 act as an oxidising as well as a reducing agent? Justify.
Justify the placement of O, S, Se, Te and Po in the same group of the periodic table in terms of electronic configuration, oxidation state and hydride formation.
Why is dioxygen a gas but sulphur a solid?
Knowing the electron gain enthalpy values for \[\ce{O -> O-}\] and \[\ce{O -> O^{2-}}\] as −141 and 702 kJ mol−1 respectively, how can you account for the formation of a large number of oxides having O2− species and not O−?
(Hint: Consider lattice energy factor in the formation of compounds).
Which aerosols deplete ozone?
Describe the manufacturing of H2SO4 by the contact process.
How is SO2 an air pollutant?
Why are halogens strong oxidising agents?
Account for the following:
Fluorine forms only one oxoacid HOF.
Explain why inspite of nearly the same electronegativity, oxygen forms hydrogen bonding while chlorine does not.
Give two uses of ClO2.
Why are halogens coloured?
Write the reactions of F2 with water.
Write the reactions of Cl2 with water.
How can you prepare Cl2 from HCl ? Write reaction only.
How can you prepare HCl from Cl2?
What inspired N. Bartlett for carrying out reaction between Xe and PtF6?
What is the oxidation state of phosphorus in H3PO3?
What is the oxidation state of phosphorus in the following:
PCl3
What is the oxidation state of phosphorus in the following:
Ca3P2
What is the oxidation state of phosphorus in the following:
Na3PO4
What is the oxidation state of phosphorus in the following:
POF3
Write balanced equations for NaCl is heated with sulphuric acid in the presence of MnO2
Write balanced equations for chlorine gas is passed into a solution of NaI in water.
How are xenon fluorides XeF2, XeF4 and XeF6 obtained?
With what neutral molecule is ClO− isoelectronic? Is that molecule a Lewis base?
Give the methods of preparation of the following compound:
XeO3
Give the methods of preparation of the following compound:
XeOF4
Arrange the following in the order of property indicated for the given set:
F2, Cl2, Br2, I2 - increasing bond dissociation enthalpy.
Arrange the following in the order of property indicated for the given set:
HF, HCl, HBr, HI - increasing acid strength.
Arrange the following in the increasing order of property mentioned:
NH3, PH3, AsH3, SbH3, BiH3 (Base strength)
Which one of the following does not exist?
XeOF4
NeF2
XeF2
XeF6
Give the formula and describe the structure of a noble gas species which is isostructural with:
\[\ce{ICI^-_4}\]
Give the formula and describe the structure of a noble gas species which is isostructural with:
\[\ce{IBr^-_2}\]
Give the formula and describe the structure of a noble gas species which is isostructural with:
\[\ce{BrO^-_3}\]
Why do noble gases have comparatively large atomic sizes?
Write two uses of neon.
Give the uses of argon.
Nootan solutions for केमिस्ट्रि पार्ट १ अँड २ [इंग्रजी] इयत्ता १२ आयएससी 7 p-Block Elements QUESTIONS FROM ISC EXAMINATION PAPERS [Pages 460 - 461]
Write a balanced equation for the following reaction:
Fluorine and dilute sodium hydroxide.
Give balanced chemical equations for the following:
Chlorine gas is passed through cold, dilute NaOH.
Give balanced chemical equations for the following:
Sulphur dioxide gas is passed through NaOH solution.
Give the reactions and the conditions required for the preparation of the following compounds.
XeF6
Give the reactions and the conditions required for the preparation of the following compound.
XeOF4
Give balanced equations for the following reaction:
Ozone and mercury
Give balanced equations for the following reaction:
Action of heat on a mixture of sodium chloride and concentrated sulphuric acid.
Name the inert gases used for:
Filling sodium vapour lamps.
Name the inert gases used for:
Obtaining light of different colours in neon signs.
What is the hybridisation of the chlorine atom in CIF3 molecule?
Draw the structure of the ClF3 molecule and state its geometry.
Xenon gives a series of fluorides, but helium and neon do not. Why?
(At. No.: Xe = 54, Ne = 10, He = 2.)
Explain giving reasons why the halogens are coloured and the colour deepens from fluorine to iodine.
Draw the structure of xenon hexafluoride molecule and state the hybridisation of the central atom and the structure of the molecule.
Among the following halogens, the one which does not form an oxyacid is:
Fluorine
Chlorine
Bromine
Iodine
Write a balanced chemical equation for the following reaction:
Ozone and lead sulphide
Write a balanced chemical equation for the following reaction:
Chlorine is passed through hot concentrated NaOH solution.
Write a balanced chemical equation for the following reaction:
Sulphuric acid is treated with phosphorous.
For the molecule IF7:
- Draw the structure of the molecule.
- State the hybridisation of the central atom.
- State the geometry of the molecule.
Draw the structure of xenon tetrafluoride molecule and state the hybridization of the central atom and the geometry of the molecule.
When SO2 gas is passed through the acidified K2Cr2O7 solution, the colour of the solution changes to ______.
Red
Black
Orange
Green
Give a balanced equation for the following reaction:
Phosphorous reacts with conc. sulfpuric acid.
Sulphur dioxide acts as an oxidising agent as well as a reducing agent. Give one reaction each to show its oxidising nature and its reducing nature.
Explain why high pressure is required in the manufacture of sulphur trioxide by the contact process. State the law or principle used.
Calculate the equilibrium constant (Kc) for the formation of NH3 in the following reaction:
\[\ce{N2_{(g)} + 3H2_{(g)} <=> 2NH3_{(g)}}\]
At equilibrium, the concentration of NH3, H2 and N2 are 1.2 × 10−2, 3.0 × 10−2 and 1.5 × 10−2 M respectively.
Give balanced equation for the following reaction:
Lead sulphide is heated with hydrogen peroxide.
Give a balanced equation for the following reaction:
Ozone is treated with potassium iodide solution.
Discuss the theory involved in the manufacture of sulphuric acid by the contact process.
What is the type of hybridisation of iodine in interhalogen compound IF3?
What is the type of hybridisation of iodine in interhalogen compound IF5?
What is the type of hybridisation of iodine in interhalogen compound IF7?
Draw the structure of xenon hexafluoride molecule and state the hybridisation of the central atom and the structure of the molecule.
Fill in the blanks:
The geometry of XeOF4 molecule is ______ and the hybridisation of xenon atom in the molecule is ______.
Explain why:
Nitrogen does not form pentahalides.
Explain why:
Helium is used for filling weather balloons.
ICl is more reactive than I2.
Write the structure of HClO4.
Draw the structure of the following:
H3PO3
Explain why:
Mercury loses its meniscus in contact with ozone.
Explain giving reasons why the halogens are coloured and the colour deepens from fluorine to iodine.
Explain why:
Hydride of sulphur is a gas while hydride of oxygen is a liquid.
Complete and balance the following reaction:
\[\ce{NaCl + MnO2 + H2SO4 ->}\] ______ + ______ + ______ + ______
Why is the electron affinity of fluorine less than chlorine?
Give reason for the following:
Red phosphorus is less reactive than white phosphorus.
Explain why:
Ozone acts as a powerful oxidising agent.
Draw structures of XeF6.
Give the structure of IF7.
Why are interhalogen compounds more reactive than the related elemental halogens?
Account for the following:
Sulphur exhibits tendency for catenation but oxygen does not.
Explain why:
On being slowly passed through water, PH3 forms bubbles but NH3 dissolves.
Complete and balance the following reactions:
\[\ce{P4 + H2SO4 ->}\] ______ + ______ + ______
Complete and balance the following reaction:
\[\ce{Ag + \underset{(dilute)}{HNO3} ->}\] ______ + ______ + ______
Complete and balance the following chemical equation:
\[\ce{P4 + NaOH + H2O ->[Heat][Inert atm.]}\] ______ + ______
Complete and balance the following chemical equation:
\[\ce{Cu + \underset{dil.}{HNO3} ->}\] ______ + ______ + ______
Draw the structure of xenon tetrafluoride molecule and state the hybridization of the central atom and the geometry of the molecule.
Complete the statement by selecting the correct alternative from the choices given below:
Copper metal on treatment with conc. nitric acid (HNO3) gives ______.
Cu(NO3)2 + CuO + H2O
Cu(NO3)2 + NO + H2O
Cu(NO3)2 + NO2 + H2O
Cu(NO3)2 + H2SO4 + H2O
In compounds XeF2, XeF4 and XeF6, the number of lone pair(s) on Xe atom respectively is ______.
2, 3, 1
1, 2, 3
4, 1, 2
3, 2, 1
BrF5 molecule is an interhalogen compound.
What is the structure of the given molecule?
Pentagonal bipyramidal
Square pyramidal
square planar
Tetrahedral
BrF5 molecule is an interhalogen compound.
What is the type of hybridisation shown by central atom of the above molecule?
sp3
sp
sp3d2
sp3d3
If chlorine gas is passed through hot and conc. aqueous sodium hydroxide solution, the products formed have chlorine in different oxidation states. These oxidation states are indicated as:
−1 and +5
−1 and +3
−1 and +1
+1 and +5
With reference to XeOF4 molecule, answer the following question:
What is the type of hybridisation of Xe atom in the given molecule?
sp3d
sp3d2
sp3d3
sp3
With reference to XeOF4 molecule, answer the following question:
What is the geometry of the molecule?
octahedral
square pyramidal
square planar
tetrahedral
Ozone is an allotropic form of oxygen. It acts as a powerful oxidising agent.
During the oxidation of mercury (Hg) by ozone, the sub oxide (Hg2O) formed dissolves in mercury causing it to lose its meniscus and starts sticking to the sides of glass. What is this phenomenon called?
Branching of mercury
Tailing of mercury
Breaking of meniscus
distorted meniscus
Ozone is an allotropic form of oxygen. It acts as a powerful oxidising agent.
Which one of the following compounds is formed when ozone reacts with black lead sulphide?
Blue coloured lead trioxide
White coloured lead sulphate
Green coloured lead oxide
Red coloured tri lead tetroxide
Solutions for 7: p-Block Elements
![Nootan solutions for केमिस्ट्रि पार्ट १ अँड २ [इंग्रजी] इयत्ता १२ आयएससी chapter 7 - p-Block Elements Nootan solutions for केमिस्ट्रि पार्ट १ अँड २ [इंग्रजी] इयत्ता १२ आयएससी chapter 7 - p-Block Elements - Shaalaa.com](/images/chemistry-part-1-and-2-english-class-12-isc_6:a6407550bf9a46188b122e0d354548f8.png)
Nootan solutions for केमिस्ट्रि पार्ट १ अँड २ [इंग्रजी] इयत्ता १२ आयएससी chapter 7 - p-Block Elements
Shaalaa.com has the CISCE Mathematics केमिस्ट्रि पार्ट १ अँड २ [इंग्रजी] इयत्ता १२ आयएससी CISCE solutions in a manner that help students grasp basic concepts better and faster. The detailed, step-by-step solutions will help you understand the concepts better and clarify any confusion. Nootan solutions for Mathematics केमिस्ट्रि पार्ट १ अँड २ [इंग्रजी] इयत्ता १२ आयएससी CISCE 7 (p-Block Elements) include all questions with answers and detailed explanations. This will clear students' doubts about questions and improve their application skills while preparing for board exams.
Further, we at Shaalaa.com provide such solutions so students can prepare for written exams. Nootan textbook solutions can be a core help for self-study and provide excellent self-help guidance for students.
Concepts covered in केमिस्ट्रि पार्ट १ अँड २ [इंग्रजी] इयत्ता १२ आयएससी chapter 7 p-Block Elements are .
Using Nootan केमिस्ट्रि पार्ट १ अँड २ [इंग्रजी] इयत्ता १२ आयएससी solutions p-Block Elements exercise by students is an easy way to prepare for the exams, as they involve solutions arranged chapter-wise and also page-wise. The questions involved in Nootan Solutions are essential questions that can be asked in the final exam. Maximum CISCE केमिस्ट्रि पार्ट १ अँड २ [इंग्रजी] इयत्ता १२ आयएससी students prefer Nootan Textbook Solutions to score more in exams.
Get the free view of Chapter 7, p-Block Elements केमिस्ट्रि पार्ट १ अँड २ [इंग्रजी] इयत्ता १२ आयएससी additional questions for Mathematics केमिस्ट्रि पार्ट १ अँड २ [इंग्रजी] इयत्ता १२ आयएससी CISCE, and you can use Shaalaa.com to keep it handy for your exam preparation.