Advertisements
Advertisements
प्रश्न
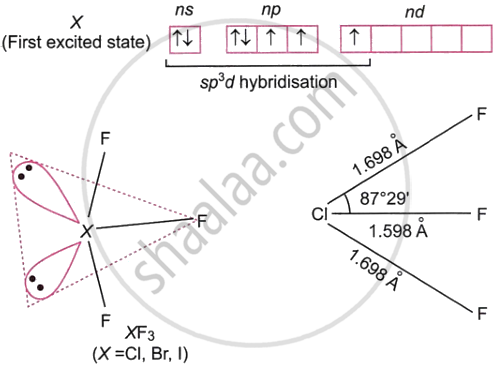
Discuss the formation and structure of XF3 (X = Cl, Br, I) molecule.
लघु उत्तर
Advertisements
उत्तर
Type XY3 interhalogen compounds have a T-shaped structure and are created when the central atom X undergoes sp3d hybridisation in its first excited state. For example, the figure shows how an XF3 molecule is formed by sp3d hybridisation as well as its structure.
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
