Advertisements
Advertisements
प्रश्न
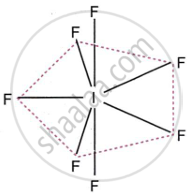
What is the hybridisation of iodine in IF7? Give its structure.
Write the structure of the following interhalogen compound:
lF7
Also mention the state of hybridisation of Cl atom present in it.
रासायनिक समीकरणे/रचना
अति संक्षिप्त उत्तर
Advertisements
उत्तर
Hybridisation of iodine in IF7 is sp3d3. The structure of IF7 is pentagonal bipyramidal.
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
