Advertisements
Advertisements
Questions
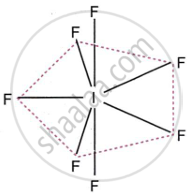
What is the hybridisation of iodine in IF7? Give its structure.
Write the structure of the following interhalogen compound:
lF7
Also mention the state of hybridisation of Cl atom present in it.
Chemical Equations/Structures
Very Short Answer
Advertisements
Solution
Hybridisation of iodine in IF7 is sp3d3. The structure of IF7 is pentagonal bipyramidal.
shaalaa.com
Is there an error in this question or solution?
