Advertisements
Advertisements
प्रश्न
Discuss the important allotropic forms of sulphur.
Advertisements
उत्तर
Sulphur exhibits the phenomenon of allotropy and exists in various allotropic forms. The important allotropic forms of sulphur are given below.
1. Rhombic sulphur: The most common and stable type of sulphur is this one. Another name for it is an octahedral or sulphur. It has a specific gravity of 2.06 g cm−3 and is pale yellow in colour. 385.8 K is its melting point. It dissolves in carbon disulphide but not in water. At room temperature, it remains stable.
On standing, all other forms of sulphur gradually change into this form. It is made by evaporating the roll sulphur solution in carbon disulphide once rhombic sulphur crystals are formed.
At room temperature, rhombic sulphur is discovered to be composed of Sa units. As seen in the figure, the eight sulphur atoms that make up a molecule form a puckered ring.
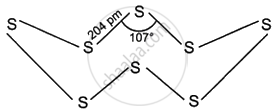
2. Monoclinic sulphur: It is also called P-sulphur or prismatic sulphur. Sulphur is melted in a dish and allowed to cool until a crust forms. After piercing this crust with a needle, the liquid underneath is extracted. The translucent crystals of monoclinic sulphur are produced upon crust removal.
Only above 369 K is monoclinic sulphur stable; below this temperature, it transforms into rhombic sulphur. Its m.p. is 392.2 K, and its specific gravity is 1.98 g cm−3. It dissolves in carbon disulphide but not in water. S8 molecules, which have a puckered ring structure resembling that of rhombic sulphur as seen in the top figure, are another kind of monoclinic sulphur. Nonetheless, the symmetry of monoclinic sulphur crystals is different from that of rhombic sulphur crystals.
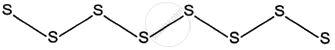
3. Plastic sulphur: A different term for it is γ-sulphur. After obtaining plastic sulphur, it is made by mixing boiling sulphur with cold water. It is a soft amorphous mass that resembles rubber. It has a 1.95 g cm−3 specific gravity. Its melting point is not very high. It does not dissolve in carbon disulphide or water.
It is unstable and, when upright, takes on a rhombic shape. The structure of plastic sulphur is open chain. In order to create open chain molecules, the sulphur atoms are joined together so that each one is connected to two more sulphur atoms via covalent bonds.
4. Milk of sulphur: A mixture of calcium pentasulphide (CaS5) and calcium thiosulphate (CaS2O3) is produced when sulphur flowers are cooked with lime milk. When hydrochloric acid is added to this combination, a white, amorphous precipitate known as milk of sulphur is produced.
\[\ce{\underset{Milk of lime}{3Ca(OH)2} + \underset{sulphur}{\underset{Flowers of}{12S}} -> \underset{pentasulphide}{\underset{Calcium}{2CaS5}} + \underset{thiosulphate}{\underset{Calcium}{CaS2O3}} + 3H2O}\]
\[\ce{2CaS5 + CaS2O3 + 6HCl -> 3CaCl2 + 3H2O + \underset{sulphur}{\underset{Milk of}{12S}}}\]
Carbon disulphide dissolves milk of sulphur. After standing for an extended period of time, it usually returns to the rhombic variety. The medical field makes extensive use of it.
5. Colloidal sulphur: Hydrogen sulphide can be passed via nitric acid to produce a colloidal form of sulphur, or sodium thiosulphate solution can be treated with diluted hydrochloric acid.
\[\ce{H2S + 2HNO3 -> 2H2O + 2NO2 + \underset{sulphur}{\underset{Colloidal}{S}}}\]
\[\ce{Na2S2O3 + 2HCl -> 2NaCl + 2SO2 + H2O + \underset{sulphur}{\underset{Colloidal}{S}}}\]
It may also be obtained be treating hydrogen sulphide with sulphur dioxide.
\[\ce{2H2S + SO2 -> 2H2O + \underset{sulphur}{\underset{Colloidal}{3S}}}\]
Colloidal sulphur changes into ordinary sulphur on heating or on keeping for a long time.
