Advertisements
Advertisements
प्रश्न
Discuss the structures of the hydrides of group 16 elements.
सविस्तर उत्तर
Advertisements
उत्तर
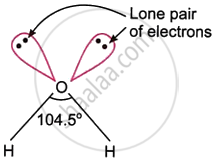
- The core group of 16 atoms in the ground state undergoes sp3 hybridisation to generate all of these angular-shaped hydrides. For example, one 2s and three 2p atomic orbitals of the ground state of the oxygen atom \[\ce{(1s^2 2s^2 2p^2_x 2p^1_y 2p^1_x)}\] mix up and redistribute their energies to form four sp3 hybrid orbitals that are orientated towards the four corners of a regular tetrahedron at an angle of 109°28' during the formation of the H2O molecule.
- Two \[\ce{O - H}\] σ-bonds are formed when the two half-filled sp3 hybrid orbitals overlap with the s atomic orbitals of two hydrogen atoms; the other two sp3 hybrid orbitals, each of which carries a single pair of electrons, do not form a bond. Thus, an angled H2O molecule is produced.
- Two lone pairs and two bond pairs of electrons encircle the oxygen atom in the H2O molecule. The expected \[\ce{H - O - H}\] angle of 109°28' decreases to 104.5°, the observed \[\ce{H - O - H}\] angle in H2O, because the lone pair-bond pair repulsion is stronger than the bond pair-bond pair repulsion.
- The figure shows the structure of H2O. The other hydrides of the group possess similar structures and are formed in the same way as discussed above for H2O.
- The group’s other hydrides are created similarly to how H2O was described above and have comparable structures. However, as these hydrides transition from H2O to H2Te, the \[\ce{H - M - H}\] bond angle reduces.
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
