Advertisements
Advertisements
प्रश्न
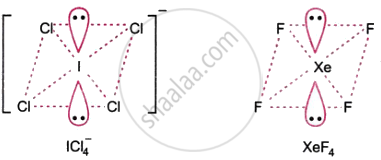
Give the formula and describe the structure of a noble gas species which is isostructural with:
\[\ce{ICI^-_4}\]
Advertisements
उत्तर
ICl4 is isostructural with XeF4 as both are square planar.
संबंधित प्रश्न
Draw the structure of the following molecule:
XeF4
Which noble gas is used in filling balloons for meteorological observations?
Complete the following equations : XeF4 + O2F2→
Balance the following equation:
\[\ce{XeF6 + H2O -> XeO2F2 + HF}\]
How are xenon fluorides XeF2, XeF4 and XeF6 obtained?
How are XeO3 and XeOF4 prepared?
Give the formula and describe the structure of a noble gas species which is isostructural with:
\[\ce{IBr^-_2}\]
Complete the given chemical equations:
`2XeF_2 + 2H_2O->`
Draw the structures of the following
XeF6
Fill in the blanks by choosing the appropriate word/words from the brackets
(square pyramidal, electrical, 74; 26, sp3d2, sp3d, chemical, 68, 32, tetrahedral, yellow, white, iodoform, Lucas)
The geometry of XeOF4 molecule is ______ and the hybridisation of Xenon atom in the molecule is ________.
The number of lone pairs of electrons present in ClF5:
In which of the following pairs, the two species are isostructural:
Substance having the lowest boiling point ______.
On partial hydrolysis, XeF6 gives ______.
Noble gases are named because of their inertness towards reactivity. Identify an incorrect statement about them.
Match List - I with List - II:
| List - I | List - II | ||
| (Species) | (Number of lone pairs of electrons on the central atom) |
||
| (A) | XeF2 | (i) | 0 |
| (B) | XeO2F2 | (ii) | 1 |
| (C) | XeO3F2 | (iii) | 2 |
| (D) | XeF4 | (iv) | 3 |
Choose the most appropriate answer from the options given below:
\[\ce{XeF4 + H2O - \underline{}\underline{}\underline{}\underline{} + \underline{}\underline{}\underline{}\underline{}}\]
The element expected to form the largest ion to achieve the nearest noble gas configuration is:
