Advertisements
Advertisements
Question
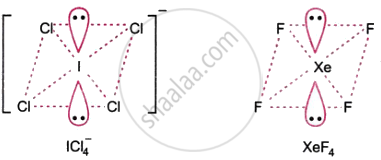
Give the formula and describe the structure of a noble gas species which is isostructural with:
\[\ce{ICI^-_4}\]
Advertisements
Solution
ICl4 is isostructural with XeF4 as both are square planar.
RELATED QUESTIONS
Draw the structure of the following molecule:
XeF4
Which noble gas has the lowest boiling point?
Which noble gas is used in filling balloons for meteorological observations?
Complete the equation : XeF2+PF5 →
Complete the following equations : XeF4 + O2F2→
Draw the structure of the following:
XeO3
How are XeO3 and XeOF4 prepared?
Which one of the following does not exist?
Give the formula and describe the structure of a noble gas species which is isostructural with:
\[\ce{BrO^-_3}\]
Complete the following reactions:
XeF6+2H2O ----->
Write the electronic configuration of the following element:
Krypton (Z = 36)
Draw the structure of XeF4.
In which of the following pairs, the two species are isostructural:
Match the compounds given in Column I with the hybridisation and shape given in Column II and mark the correct option.
| Column I | Column II |
| (A) XeF6 | (1) sp3d3 – distorted octahedral |
| (B) XeO3 | (2) sp3d2 – square planar |
| (C) XeOF4 | (3) sp3 – pyramidal |
| (D) XeF4 | (4) sp3d2 – square pyramidal |
\[\ce{XeF4 + H2O - \underline{}\underline{}\underline{}\underline{} + \underline{}\underline{}\underline{}\underline{}}\]
Discuss the trends in electronegativity and atomic radii for elements of group 16 17, 18.
Write two uses of neon.
The element expected to form the largest ion to achieve the nearest noble gas configuration is:
