Advertisements
Advertisements
Question
Draw structures of the following:
XeF4
Diagram
One Line Answer
Advertisements
Solution
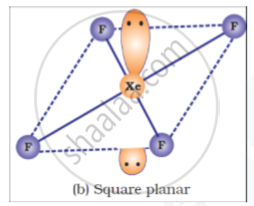
shaalaa.com
Is there an error in this question or solution?
APPEARS IN
RELATED QUESTIONS
Account for the following:
Helium is used in diving apparatus.
Write the structures of the following molecule:
XeOF4
Complete the following equations : XeF4 + O2F2→
How are xenon fluorides XeF2, XeF4 and XeF6 obtained?
Answer the following.
List the uses of Neon and argon gases.
Draw structure and name the shape of bromine trifluoride.
Match the compounds given in Column I with the hybridisation and shape given in Column II and mark the correct option.
| Column I | Column II |
| (A) XeF6 | (1) sp3d3 – distorted octahedral |
| (B) XeO3 | (2) sp3d2 – square planar |
| (C) XeOF4 | (3) sp3 – pyramidal |
| (D) XeF4 | (4) sp3d2 – square pyramidal |
The order of increasing sizes of atomic radii among the elements O, S, Se and As is:
Match List - I with List - II:
| List - I | List - II | ||
| (Species) | (Number of lone pairs of electrons on the central atom) |
||
| (A) | XeF2 | (i) | 0 |
| (B) | XeO2F2 | (ii) | 1 |
| (C) | XeO3F2 | (iii) | 2 |
| (D) | XeF4 | (iv) | 3 |
Choose the most appropriate answer from the options given below:
Which one of the following reactions of xenon compounds is not feasible?
