Advertisements
Advertisements
Questions
Write the structures of the following molecule:
XeOF4
Draw the structure of the XeOF4.
Give the structure of XeOF4.
Chemical Equations/Structures
Advertisements
Solution
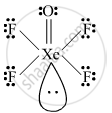
shaalaa.com
Is there an error in this question or solution?
APPEARS IN
RELATED QUESTIONS
Complete the equation : XeF2+PF5 →
test q 1234
Which one of the following does not exist?
Give the formula and describe the structure of a noble gas species which is isostructural with:
\[\ce{BrO^-_3}\]
Complete the following reactions:
XeF6+2H2O ----->
Draw the structure of XeF4.
Which of the following fluorides does not exist?
Which of the following reactions is an example of redox reaction?
Which one of the following reactions of xenon compounds is not feasible?
\[\ce{XeF6 + H2O ->[Partial][Hydrolysis] \underline{}\underline{}\underline{}\underline{} + \underline{}\underline{}\underline{}\underline{}}\]
