Advertisements
Advertisements
प्रश्न
Write the structures of the following molecule:
XeOF4
Draw the structure of the XeOF4.
Give the structure of XeOF4.
रासायनिक समीकरणे/रचना
Advertisements
उत्तर
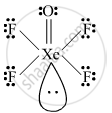
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
APPEARS IN
संबंधित प्रश्न
Account for the following:
Helium is used in diving apparatus.
Draw the structures of the following molecules: XeF2
Balance the following equation:
\[\ce{XeF6 + H2O -> XeO2F2 + HF}\]
Answer the following.
List the uses of Neon and argon gases.
Write electronic configuration and two uses of neon. (Z = 10)
Write the electronic configuration of the following element:
Krypton (Z = 36)
Sulfur dioxide reacts with sodium hydroxide solution to form _______.
The atomic radii of Zr and Hf are almost identical. This is because of
The order of increasing sizes of atomic radii among the elements O, S, Se and As is:
Write two uses of neon.
