Advertisements
Advertisements
प्रश्न
Draw structures of the following:
XeF4
Advertisements
उत्तर
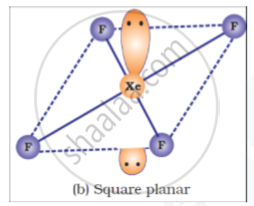
APPEARS IN
संबंधित प्रश्न
Which noble gas has the lowest boiling point?
Draw the structure of the following:
XeO3
test q 1234
Balance the following equation:
\[\ce{XeF6 + H2O -> XeO2F2 + HF}\]
Complete the following reactions:
XeF6+2H2O ----->
Fill in the blanks by choosing the appropriate word/words from the brackets
(square pyramidal, electrical, 74; 26, sp3d2, sp3d, chemical, 68, 32, tetrahedral, yellow, white, iodoform, Lucas)
The geometry of XeOF4 molecule is ______ and the hybridisation of Xenon atom in the molecule is ________.
The number of lone pairs of electrons present in ClF5:
The atomic radii of Zr and Hf are almost identical. This is because of
Which one of the following reactions of xenon compounds is not feasible?
\[\ce{XeF4 + H2O - \underline{}\underline{}\underline{}\underline{} + \underline{}\underline{}\underline{}\underline{}}\]
