Advertisements
Advertisements
प्रश्न
Draw structures of the following:
XeF4
आकृती
एका वाक्यात उत्तर
Advertisements
उत्तर
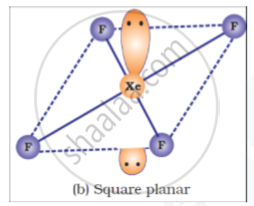
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
APPEARS IN
संबंधित प्रश्न
Draw the structures of the following molecules: XeF2
How are xenon fluorides XeF2, XeF4 and XeF6 obtained?
Give the formula and describe the structure of a noble gas species which is isostructural with:
\[\ce{ICI^-_4}\]
Give the formula and describe the structure of a noble gas species which is isostructural with:
\[\ce{IBr^-_2}\]
Write electronic configuration and two uses of neon. (Z = 10)
Draw the structure of XeF4.
Partial hydrolysis of XeF4 gives ____________.
Match List - I with List - II:
| List - I | List - II | ||
| (Species) | (Number of lone pairs of electrons on the central atom) |
||
| (A) | XeF2 | (i) | 0 |
| (B) | XeO2F2 | (ii) | 1 |
| (C) | XeO3F2 | (iii) | 2 |
| (D) | XeF4 | (iv) | 3 |
Choose the most appropriate answer from the options given below:
Write two uses of neon.
The element expected to form the largest ion to achieve the nearest noble gas configuration is:
