Advertisements
Advertisements
प्रश्न
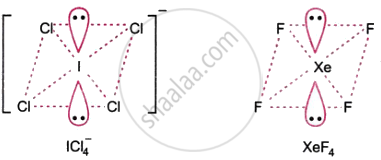
Give the formula and describe the structure of a noble gas species which is isostructural with:
\[\ce{ICI^-_4}\]
Advertisements
उत्तर
ICl4 is isostructural with XeF4 as both are square planar.
संबंधित प्रश्न
Account for the following:
Helium is used in diving apparatus.
Complete the equation : XeF2+PF5 →
Balance the following equation:
\[\ce{XeF6 + H2O -> XeO2F2 + HF}\]
Why has it been difficult to study the chemistry of radon?
What inspired N. Bartlett for carrying out reaction between Xe and PtF6?
Which one of the following does not exist?
Give the formula and describe the structure of a noble gas species which is isostructural with:
\[\ce{BrO^-_3}\]
Answer the following.
List the uses of Neon and argon gases.
Complete the following reactions:
XeF6+2H2O ----->
Draw the structures of the following
XeF6
Fill in the blanks by choosing the appropriate word/words from the brackets
(square pyramidal, electrical, 74; 26, sp3d2, sp3d, chemical, 68, 32, tetrahedral, yellow, white, iodoform, Lucas)
The geometry of XeOF4 molecule is ______ and the hybridisation of Xenon atom in the molecule is ________.
Draw structure and name the shape of bromine trifluoride.
Explain the trend in the following atomic properties of group 16 elements:
Electron gain enthalpy
Which of the following fluorides does not exist?
In which of the following pairs, the two species are isostructural:
Which of the following reactions is an example of redox reaction?
Noble gases are named because of their inertness towards reactivity. Identify an incorrect statement about them.
Discuss the trends in electronegativity and atomic radii for elements of group 16 17, 18.
The element expected to form the largest ion to achieve the nearest noble gas configuration is:
