Advertisements
Advertisements
प्रश्न
Give the structure of sulphur tetrafluoride.
आकृती
अति संक्षिप्त उत्तर
Advertisements
उत्तर
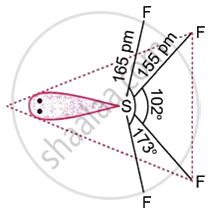
In SF4, the sulphur atom is in a state of sp3d hybridisation. As seen in the image, it has a trigonal bipyramidal structure, with a single pair of electrons occupying one of the equatorial locations.
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
