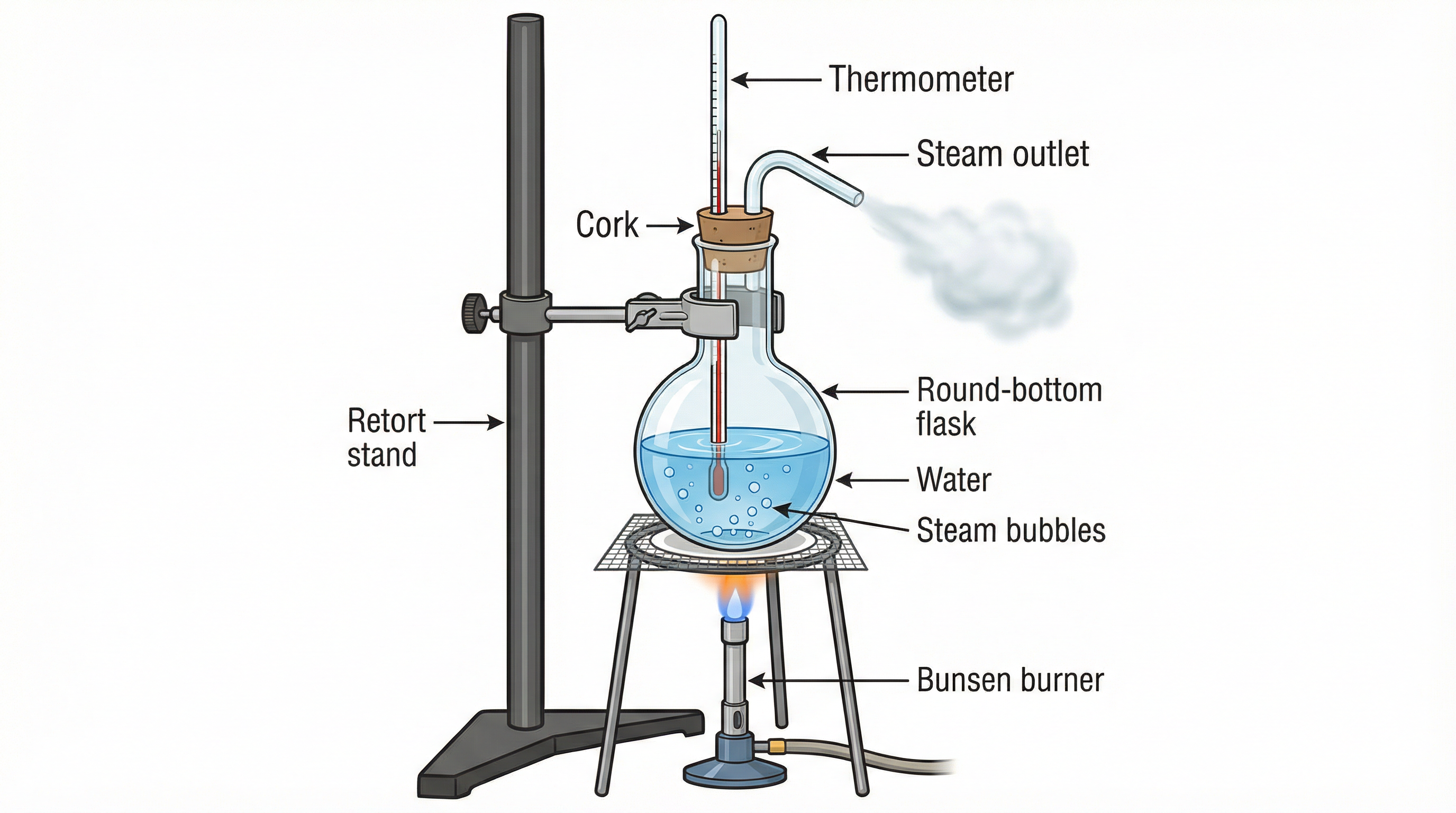
- Set up: Fill a round-bottom flask more than half with water. Insert a thermometer and a steam outlet tube through the cork, then place the flask on a burner.
- Initial Heating: Heat the water and observe small bubbles forming.
Observation: These bubbles are dissolved air escaping, not steam. - Steam Formation: As the temperature increases, steam bubbles form at the bottom.
Observation: They collapse before reaching the surface because the upper water is still cooler. - Boiling Stage: When the water reaches 100°C, steam bubbles reach the surface.
Observation: Steam inside the flask is invisible but forms a fog when it comes out and cools. - Key Test: Close the steam outlet for a few seconds.
Observation: Boiling stops even though the temperature is still 100°C because the pressure inside increases.
Conclusion:
The boiling point increases with increasing external pressure.
Part B: Decreased Pressure Lowers Boiling Point
Procedure & Observations
- Cool down: Remove the burner. Allow the water to cool to approximately 80°C.
- Seal the flask: Remove the thermometer and steam outlet tube. Seal the flask with an airtight cork.
- Invert: Turn the flask upside down and mount it on a retort stand.
- Pour ice-cold water over the outer surface of the inverted flask.
Observation: Water inside the flask begins to boil vigorously — even though it is only at approximately 80°C!
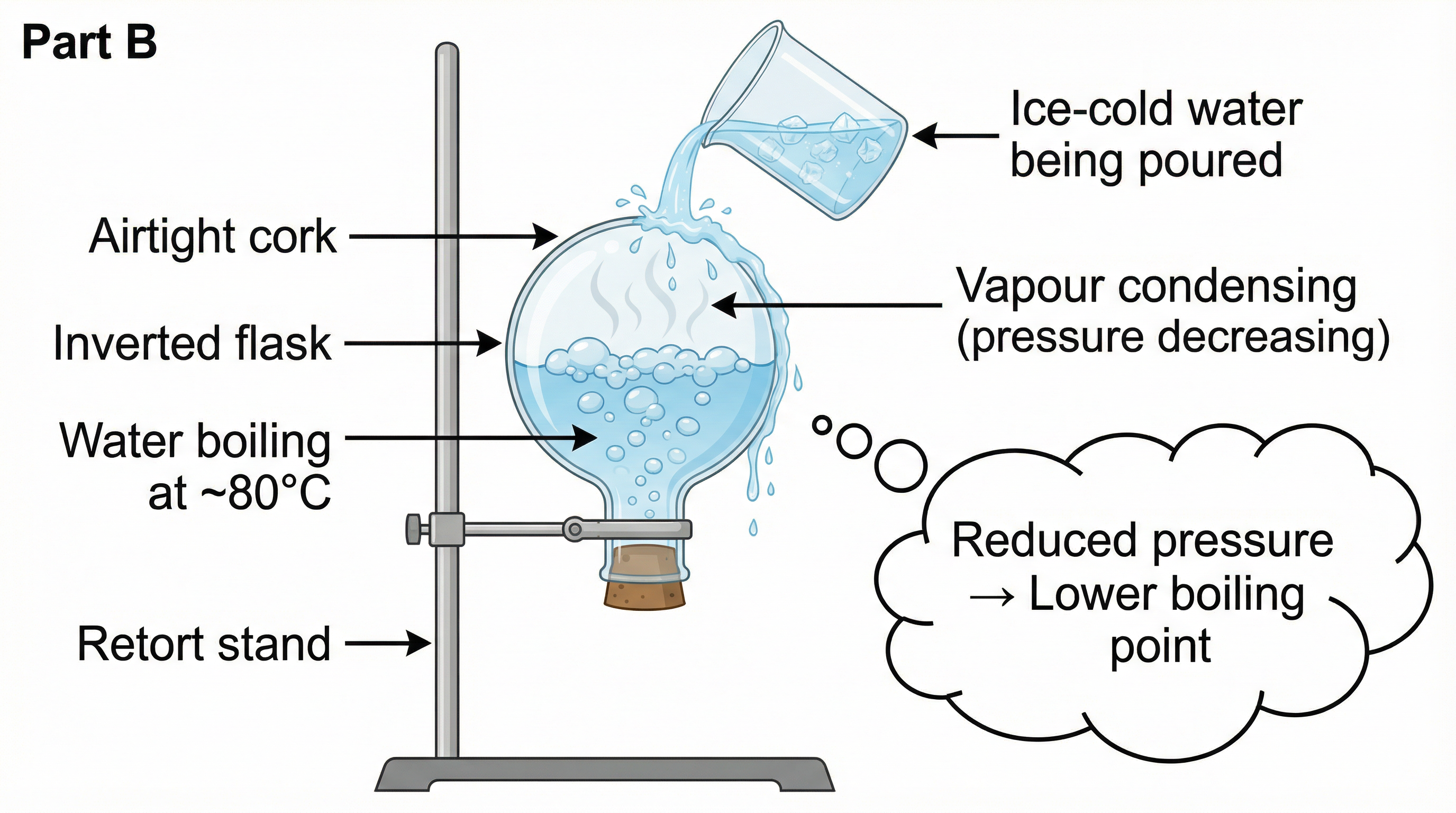
Fig.: Inverted flask experiment: Ice-cold water condenses vapour, reducing pressure, causing water to boil at ~80°C